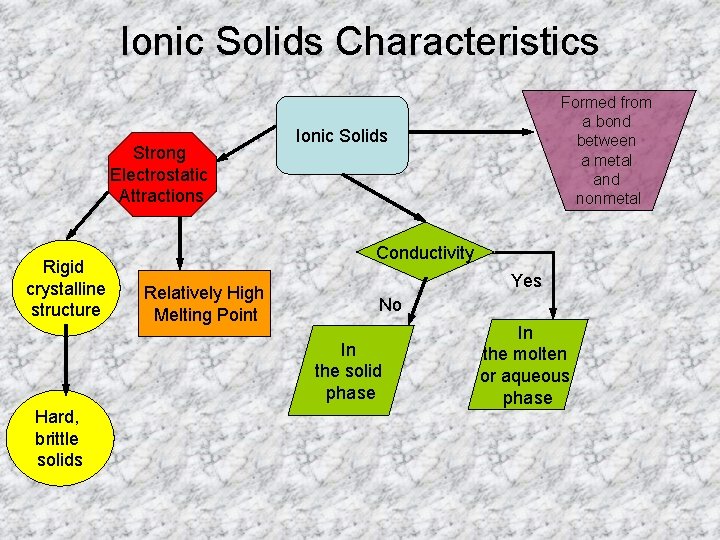
Ionic Solids Characteristics Strong Electrostatic Attractions Rigid crystalline



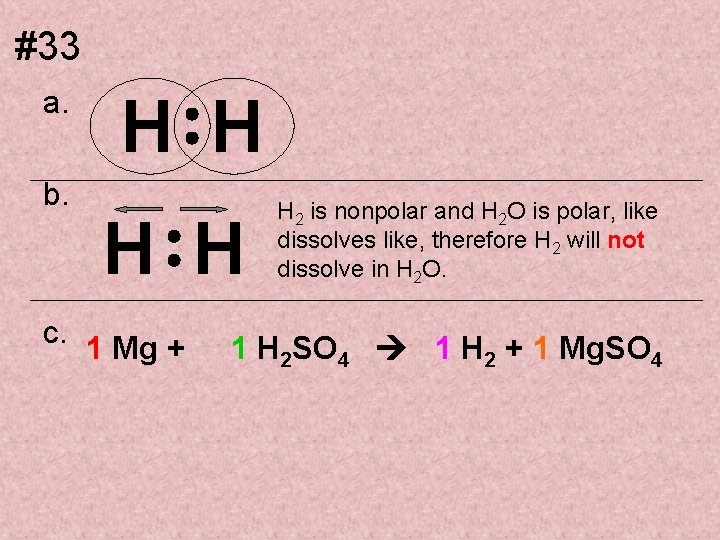





- Slides: 16

Ionic Solids Characteristics Strong Electrostatic Attractions Rigid crystalline structure Ionic Solids Conductivity Relatively High Melting Point Yes No In the solid phase Hard, brittle solids Formed from a bond between a metal and nonmetal In the molten or aqueous phase

The First Couple • # 1) Energy is released when bonds are formed. Energy is absorbed when bonds are broken. • #2) As a bond forms the PE decreases. • #3) Energy is released -> stability increases • #4) Two atoms combine to form a molecule, bond formed, energy is released

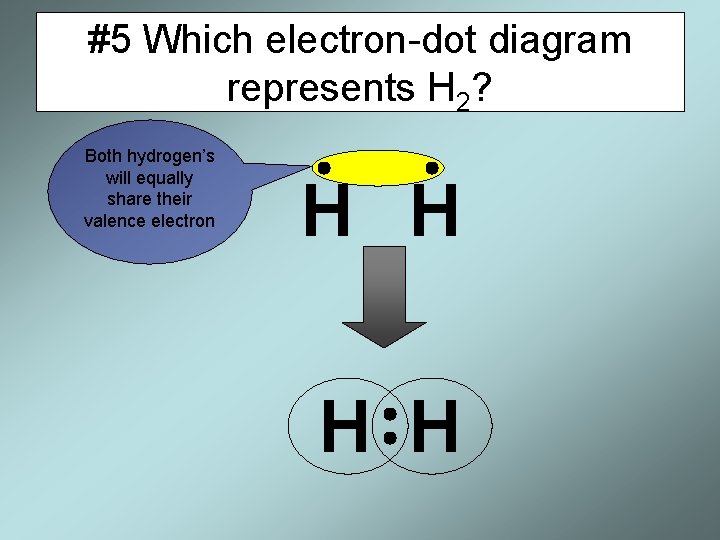
#5 Which electron-dot diagram represents H 2? Both hydrogen’s will equally share their valence electron H H

#9) In which compound do the atoms have the greatest difference in electronegativity? (1) Na. Br |0. 9 – 3. 0| = 2. 1 (2) Al. Cl 3 |1. 6 – 3. 2| = 1. 6 (3) KF |0. 8 – 4. 0| = 3. 2 (4) Li. I |1. 0 – 2. 7| = 1. 7

#13) Given the reaction: M + 2 H 2 O -> M(OH)2 + H 2 The metal represented by M is most likely a metal from Group M(OH)2 The subscript of 2 on the hydroxide came from the charge on the metal (M). +2 -1 M (OH) So the Metal (M) must be from Group 2 because of its +2 oxidation state!

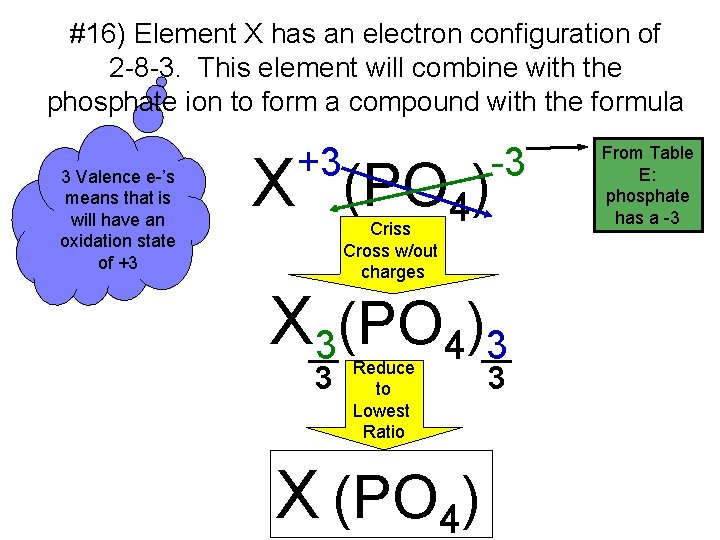
#16) Element X has an electron configuration of 2 -8 -3. This element will combine with the phosphate ion to form a compound with the formula 3 Valence e-’s means that is will have an oxidation state of +3 +3 X (PO 4) -3 Criss Cross w/out charges X 3(PO 4)3 3 Reduce to Lowest Ratio X (PO 4) 3 From Table E: phosphate has a -3
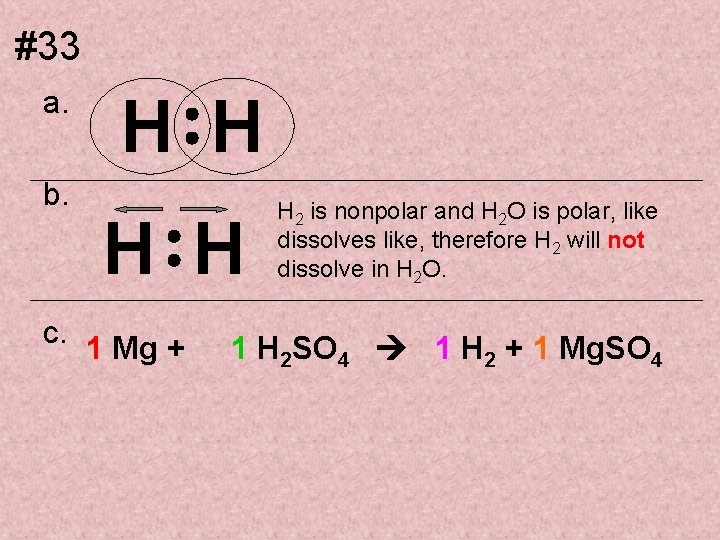
#33 a. b. H H c. 1 Mg + H 2 is nonpolar and H 2 O is polar, like dissolves like, therefore H 2 will not dissolve in H 2 O. 1 H 2 SO 4 1 H 2 + 1 Mg. SO 4

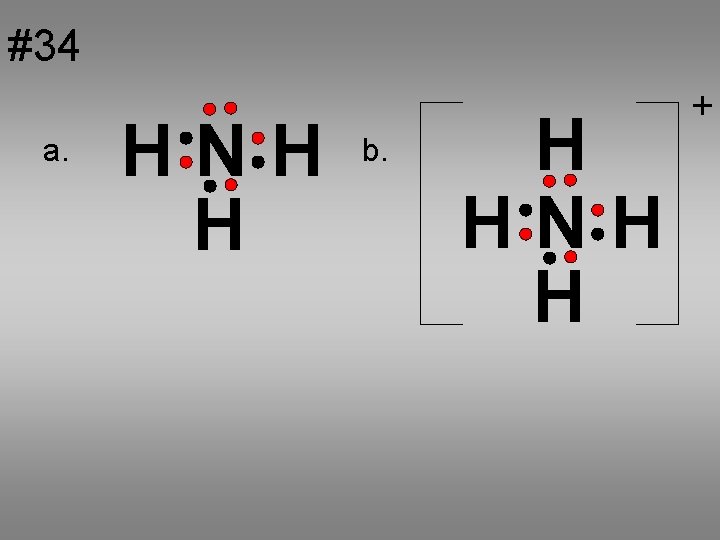
#34 a. HNH H b. H HNH H +

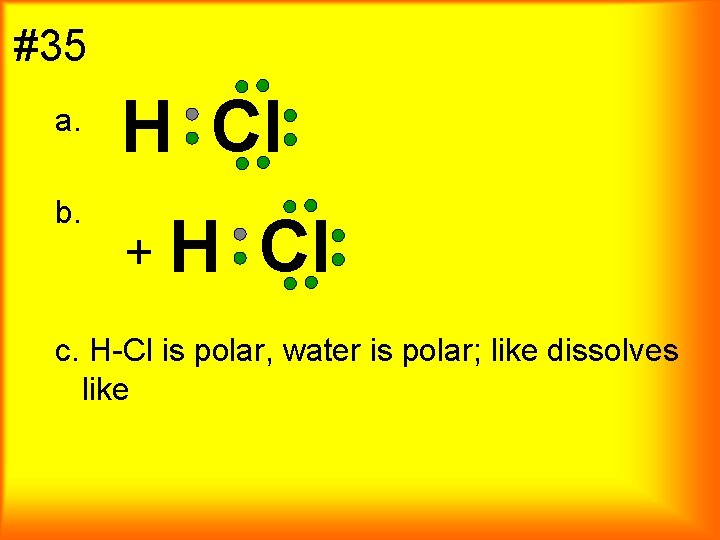
#35 a. b. H Cl + H Cl c. H-Cl is polar, water is polar; like dissolves like

#36 a. N N OR N N b. N 2 is very stable, unreactive because of triple bond (a lot of energy was released when this bond was formed).

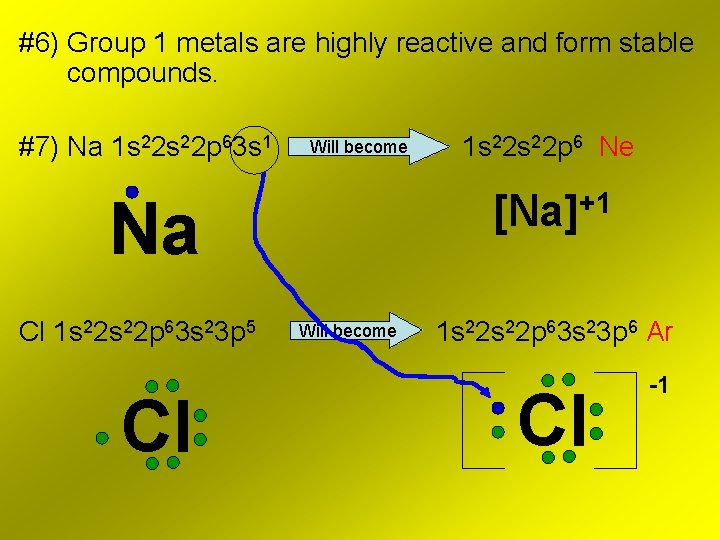
#6) Group 1 metals are highly reactive and form stable compounds. #7) Na 1 s 22 p 63 s 1 Will become [Na]+1 Na Cl 1 s 22 p 63 s 23 p 5 Cl 1 s 22 p 6 Ne Will become 1 s 22 p 63 s 23 p 6 Ar Cl -1

#8. Radon is a group 2 (metal), it wants to lose its 2 valence electrons to a nonmetal. 1) Iodine – nonmetal, wants to gain 1 e 2) Fluorine – nonmetal, wants to gain 1 e 3) sodium – metal, wants to lose 1 e 4) calcium – metal, wants to lose 2 e. Of the 2 choices that want electrons choice 2 is more reactive, larger electronegativity, than choice 1. Fluorine is the answer (choice 2).

#10) Which compound would most likely have the greatest ionic character? (1) CO |2. 6 – 3. 5| = 0. 9 (2) KF |0. 8 – 4. 0| = 3. 2 (3) Ca. O |1. 0 – 3. 5| = 2. 5 (4) Li. H |1. 0 – 2. 1| = 1. 1

#11) Given the reaction H 2 + Cl 2 → 2 HCl • Bond Breaking requires/absorbs energy. • Bond Formation releases/emits energy. The H 2 and Cl 2 bonds are broken – requires energy! The HCl bond is formed- releases energy! ü The answer is choice (4) the forming of the HCl bond releases energy

#12) Noble gases have the most stable electron configuration, choice 1 (Ne). #14) In order for the electrons to have been transferred to Oxygen, the oxygen will have to be the most electronegative element and the ionic character of the compound would be 1. 7 or greater. (1) CO 2 (2) N 2 O |2. 6 – 3. 5| = 0. 9 |3. 0 – 3. 5| = 0. 5 (2) NO 2 |3. 0 – 3. 5| = 0. 5 (4) Na 2 O |0. 9 – 3. 5| = 2. 6

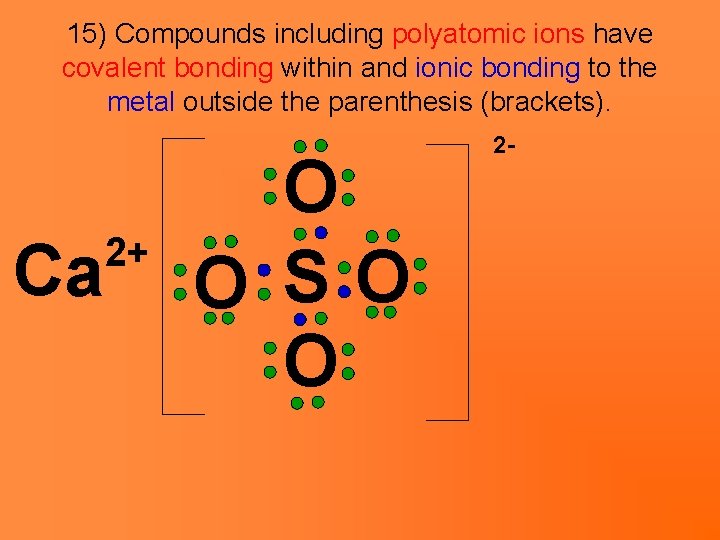
15) Compounds including polyatomic ions have covalent bonding within and ionic bonding to the metal outside the parenthesis (brackets). Ca 2+ O O SO O 2 -