Gases cont and finished Ideal vs Real Ideal
















- Slides: 17

Gases, cont. (and finished!) Ideal vs. Real Ideal Gas Law Dalton’s Law Diffusion

Chemistry Joke A Chemistry Prison Punsilicon put his neon The the window ledge. He climbed out and then krypton along the wall to meet his buddy. I hope the guard cesium

Ideal vs. Real b Ideal Gases • Follow gas laws at all conditions • Conform to the Kinetic Molecular Theory Ø Insignificant volume Ø No attraction / repulsion to each other

Ideal vs. Real b Ideal Gases don’t exist!!! • Molecules do take up space • There attractive forces between them Ø Otherwise gases would never liquefy • But…at many temperatures and pressures real gases act like ideal gases. • This makes for easier math!

Ideal vs. Real b Real Gases behave ideally… • At HIGH TEMPERATURES and LOW PRESSURES… Ø In these conditions, a gas will stay a gas. Ø At low pressures, the molecules are far apart. –We can ignore their volume. Ø At high temperatures, molecules are not together very long. –We can ignore their attractions.

Ideal vs. Real • At LOW TEMPERATURES and HIGH PRESSURES… Ø Gases experience both particle volume and attraction Ø Can be liquefied or solidified –Think of Dry Ice b We’ll continue to study ideal gases!

Ideal Gas Law b PV = n. RT n represents number of moles b R is the universal gas constant b

Ideal Gas Law b. Units for P and V have to match “R”

Ideal Gas Law Problems b Calculate the pressure in atmospheres of 0. 412 mol of He at 16°C & occupying 3. 25 L. GIVEN: WORK: P = ? atm PV = n. RT n = 0. 412 mol P(3. 25)=(0. 412)(0. 0821)(289) mol L atm/mol K T = 16°C = 289 K K L V = 3. 25 L P = 3. 01 atm R= 0. 0821 L atm/mol K

Ideal Gas Law Problems b Find the volume of 85 g of O 2 at 25°C and 104. 5 k. Pa. GIVEN: WORK: V=? 85 g 1 mol = 2. 7 mol n = 2. 7 mol 32. 00 g T = 25°C = 298 K PV = n. RT P = 1. 03 atm (1. 03)V=(2. 7) (0. 0821)(298) atm mol R = 0. 0821 V = 64 L

Dalton’s Law of Partial Pressures b For a mixture of gases in a container… b P 1 represents the “partial pressure” or the contribution by that gas. b Dalton’s Law is particularly useful in calculating the pressure of gases collected over water.

Dalton’s Law of Partial Pressures b If the gases in the first three containers are all put into the fourth, we can find the pressure in the 4 th container by adding up the pressures in the first 3:

Dalton’s Law of Partial Pressures

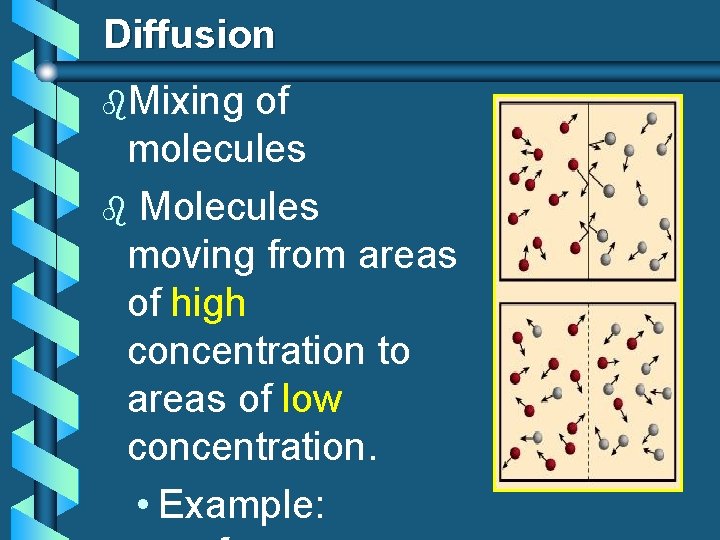
Diffusion b. Mixing of molecules b Molecules moving from areas of high concentration to areas of low concentration. • Example:

Diffusion b. With diffusion, the mass of the particle is important: • Gases of lower molar mass diffuse faster than gases of higher molar mass.

Diffusion b. Example: compare diffusion rates of helium and nitrogen. b. The molar mass of He = 4. 0 g b. The molar mass of N 2 = 28. 02 g • Therefore… b. Helium diffuses faster than nitrogen.

Chemistry Joke Q: What did the electron say to the proton to make it unhappy? A: Something negative!