Erythrocyte metabolism Alice Skoumalov Erythrocytes deliver oxygen to

Erythrocyte metabolism Alice Skoumalová

Erythrocytes Ø deliver oxygen to body tissues and remove carbon dioxide and protons Ø biconcave 7. 7μm Ø lack cell organelles Ø 120 days Ø women 4, 2 -5, 4 million/μl, men 4, 6 -6, 2 million/μl

The erythrocyte membrane 50% lipid bilayer (phospholipids, cholesterol) 50% proteins SDS-PAGE: separation of proteins (band 1 -7) isolation and analysis (10 main proteins) Integral: Anion exchanger protein, Glycophorin A, B, C Peripheral: Spectrin, Ankyrin, Actin
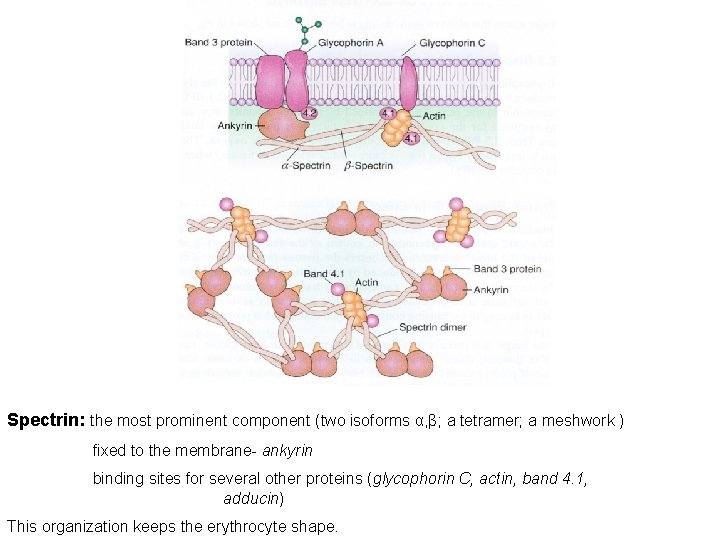
Spectrin: the most prominent component (two isoforms α, β; a tetramer; a meshwork ) fixed to the membrane- ankyrin binding sites for several other proteins (glycophorin C, actin, band 4. 1, adducin) This organization keeps the erythrocyte shape.

Hereditary spherocytosis Ø Ø autosomal dominant a deficiency in a spectrin amount and its abnormalities the presence of spherocytes in the blood the spleen‘s hemolysis
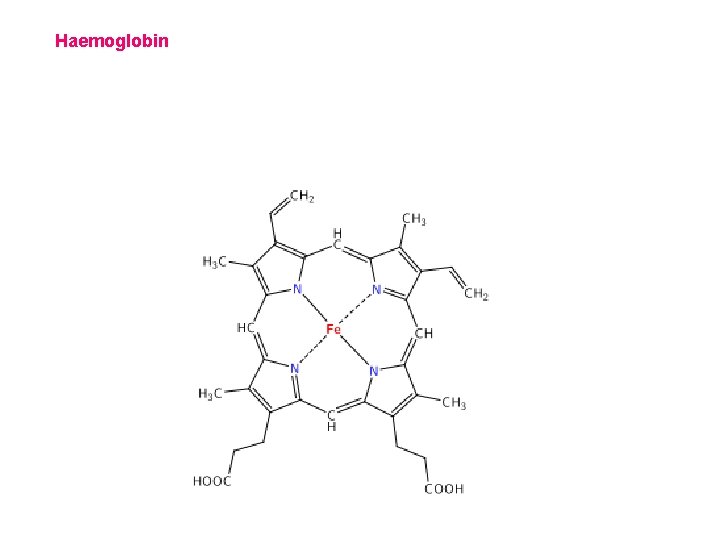
Haemoglobin
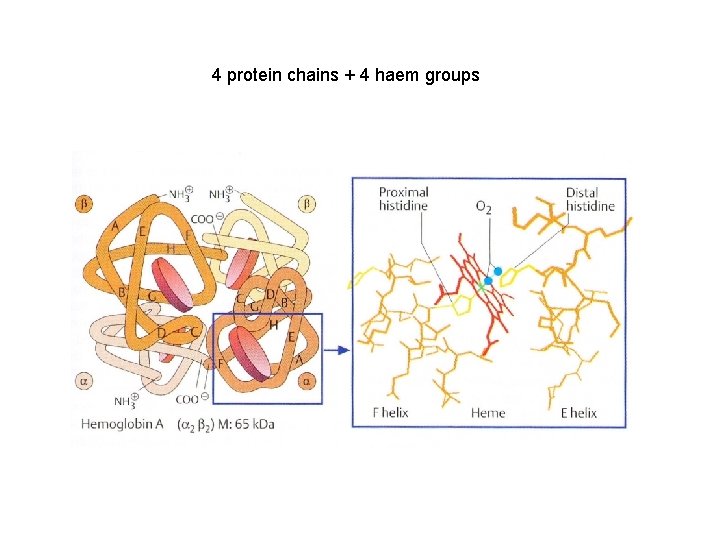
4 protein chains + 4 haem groups
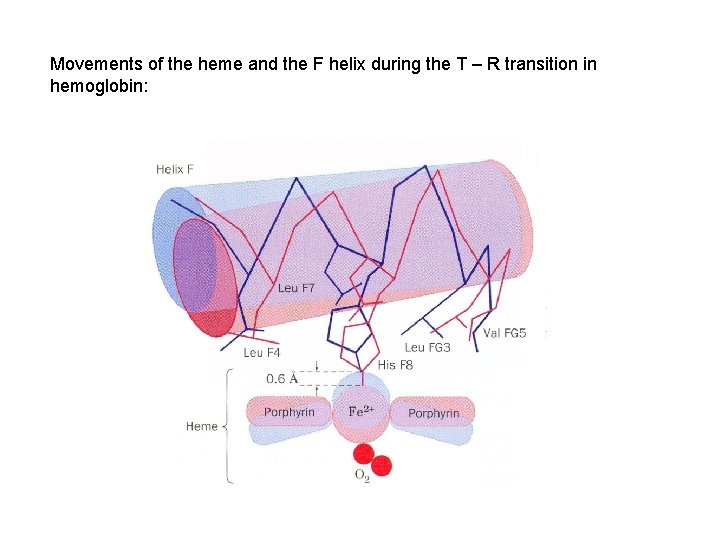
Movements of the heme and the F helix during the T – R transition in hemoglobin:

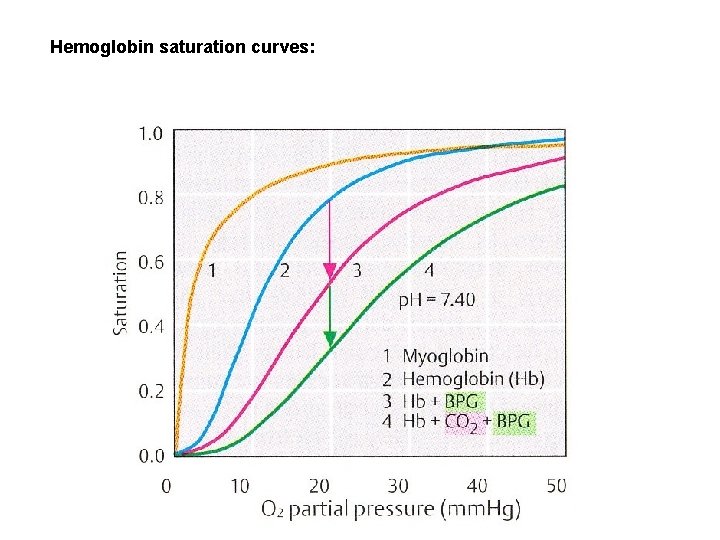
Hemoglobin saturation curves:

Hemoglobin autooxidation • • O 2 binds to Fe 2+ - an intermediate structure- an electron is delocalized between the iron ion and the O 2 the side effect - every so often a molecule of oxyhaemoglobin undergoes decomposition and release superoxide Hem - Fe 2+- O 2 Hem - Fe 3+ - O 2 • - Methemoglobin (Fe 3+) is unable to bind O 2 (methaemoglobin reductase)

Erythrocyte exceptions They lack organelles • no ATP production in oxidative phosphorylation • no ability to replace damaged lipids and proteins (low metabolic activities, with no ability to synthesize new proteins or lipids) Free radicals exposure • haemoglobin autoxidation (O 2 • - release) • a cell membrane rich in polyunsaturated fatty acids (susceptible to lipid peroxidation) • deformation in tiny capillaries; catalytic ions leakage (cause of lipid peroxidation)

Erythrocyte metabolism • Glucose as a source of energy • Glycolysis generates ATP and 2, 3 -bisphoglycerate • The pentose phosphate pathway produces NADPH • Glutathione synthesis- the antioxidant defence system


Glucose- source of energy Glucose transporter: • • • integral membrane protein (12 membrane-spanning helices) a channel for the glucose transport insulin-independent transporter Glycolysis in erythrocytes 1. Source of ATP • • Lactate- the end product Cover energy requirement 2. Generate 2, 3 -bisphoglycerate (2, 3 -BPG) • • a major reaction pathway for the consumption of glucose in erythrocytes the specific binding of 2, 3 -BPG to deoxyhemoglobin decreases the oxygen affinity of hemoglobin and facilites oxygen release in tissues

2, 3 -bisphoglycerate • Allosteric effector of haemoglobin: – binds to deoxyhaemoglobin (a central cavity capable of binding 2, 3 BPG) – decreases haemoglobin‘s O 2 affinity • Clinical aspects: – In people with high-altitude adaptation or smokers the concentration of 2, 3 -BPG in the blood is increased (low oxygen supply) – Fetal haemoglobin has low BPG affinity - the higher O 2 affinity facilitates the transfer of O 2 to the fetus via the placenta
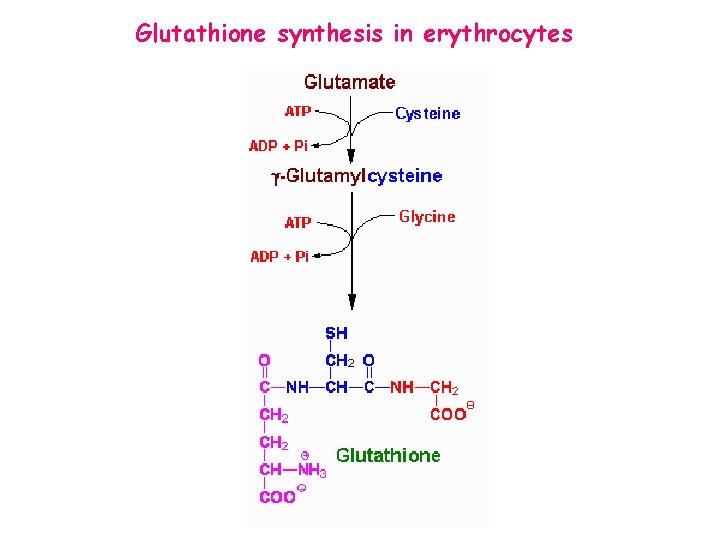
Glutathione synthesis in erythrocytes
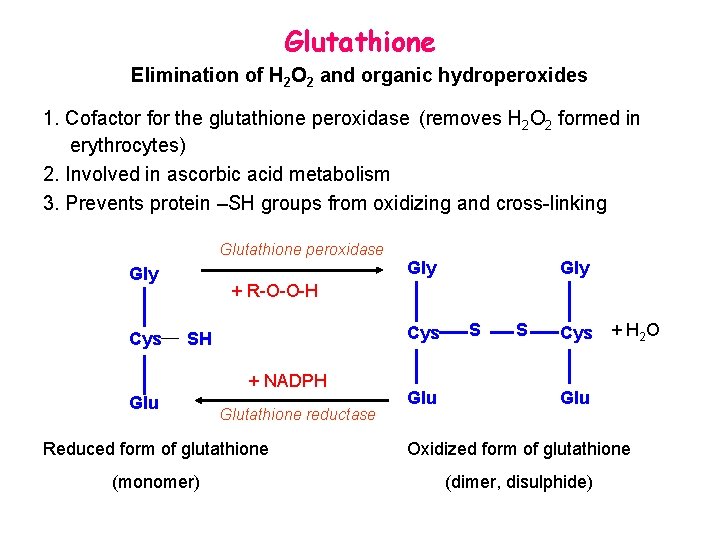
Glutathione Elimination of H 2 O 2 and organic hydroperoxides 1. Cofactor for the glutathione peroxidase (removes H 2 O 2 formed in erythrocytes) 2. Involved in ascorbic acid metabolism 3. Prevents protein –SH groups from oxidizing and cross-linking Glutathione peroxidase Gly Cys Gly + R-O-O-H Cys SH + NADPH Glutathione reductase Glu S S Cys + H 2 O Glu Reduced form of glutathione Oxidized form of glutathione (monomer) (dimer, disulphide)

The pentose phosphate pathway in erythrocytes • Generates NADPH - reduction of glutathione (eliminates H 2 O 2 formed in erythrocytes) Clinical apect: • Glucose-6 -phosphate dehydrogenase deficiency – Causes hemolytic anemia (decreased production of NADPH - reduced protection against oxidative stress - haemoglobin oxidation and Heinz bodies formation, membrane lipid peroxidation and hemolysis) – Hemolytic crises are evocated by drugs (primaquine, sulphonamide antibiotics) and foods (broad beans) – The most common enzyme deficiency disease in the world (100 million people)
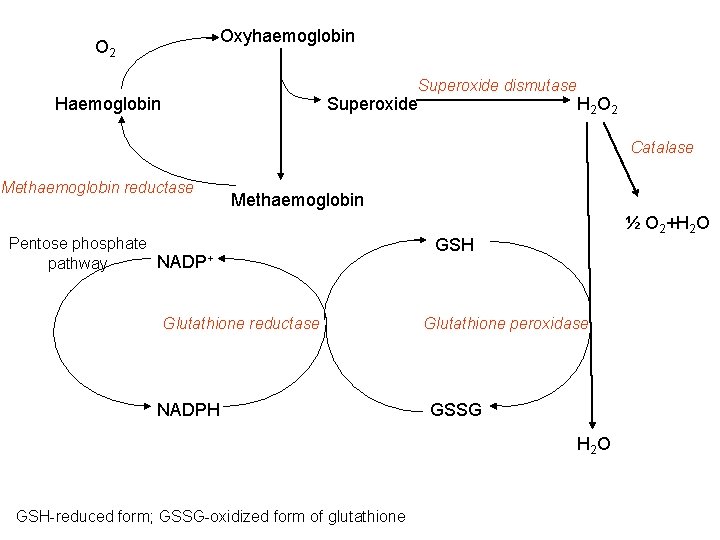
Oxyhaemoglobin O 2 Superoxide dismutase Haemoglobin Superoxide H 2 O 2 Catalase Methaemoglobin reductase Methaemoglobin Pentose phosphate NADP+ pathway Glutathione reductase NADPH ½ O 2+H 2 O GSH Glutathione peroxidase GSSG H 2 O GSH-reduced form; GSSG-oxidized form of glutathione
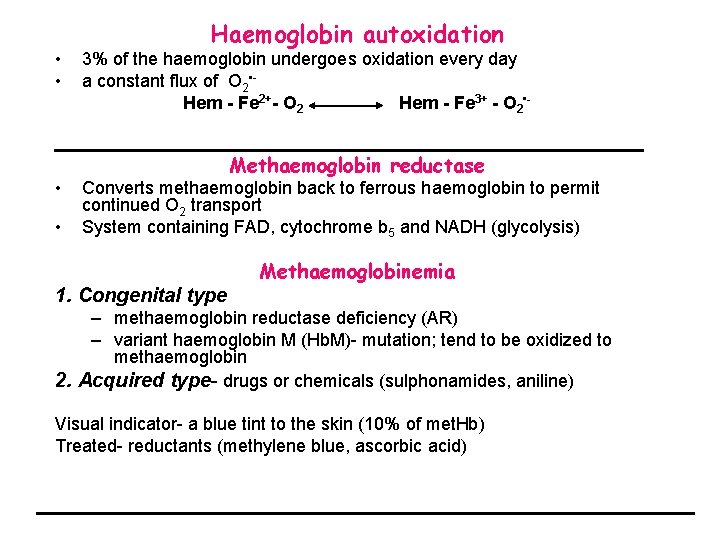
• • Haemoglobin autoxidation 3% of the haemoglobin undergoes oxidation every day a constant flux of O 2 • Hem - Fe 2+- O 2 Hem - Fe 3+ - O 2 • - Methaemoglobin reductase Converts methaemoglobin back to ferrous haemoglobin to permit continued O 2 transport System containing FAD, cytochrome b 5 and NADH (glycolysis) Methaemoglobinemia 1. Congenital type – methaemoglobin reductase deficiency (AR) – variant haemoglobin M (Hb. M)- mutation; tend to be oxidized to methaemoglobin 2. Acquired type- drugs or chemicals (sulphonamides, aniline) Visual indicator- a blue tint to the skin (10% of met. Hb) Treated- reductants (methylene blue, ascorbic acid)

Superoxide dismutase (SOD) • • a high concentration in erythrocytes accelerates the dismutation O 2 • - to H 2 O 2 remove 1. Catalase 2. Glutathione peroxidase 1. Catalase • • a ferric haem group bound to the active site catalyses decomposition of H 2 O 2 to water and oxygen: 2 H 2 O 2 2 H 2 O+O 2
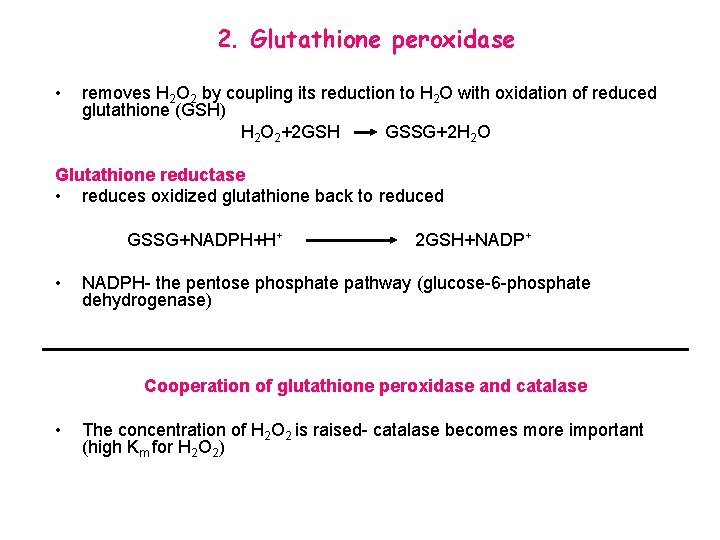
2. Glutathione peroxidase • removes H 2 O 2 by coupling its reduction to H 2 O with oxidation of reduced glutathione (GSH) H 2 O 2+2 GSH GSSG+2 H 2 O Glutathione reductase • reduces oxidized glutathione back to reduced GSSG+NADPH+H+ • 2 GSH+NADP+ NADPH- the pentose phosphate pathway (glucose-6 -phosphate dehydrogenase) Cooperation of glutathione peroxidase and catalase • The concentration of H 2 O 2 is raised- catalase becomes more important (high Km for H 2 O 2)

Biological antioxidants: Quinols and enols α-Tocopherol (vit. E) Ubiquinol (coenzyme Q) Ascorbic acid (vit. C) Carotenoids β-Carotin Lycopin Others Glutathione Bilirubin
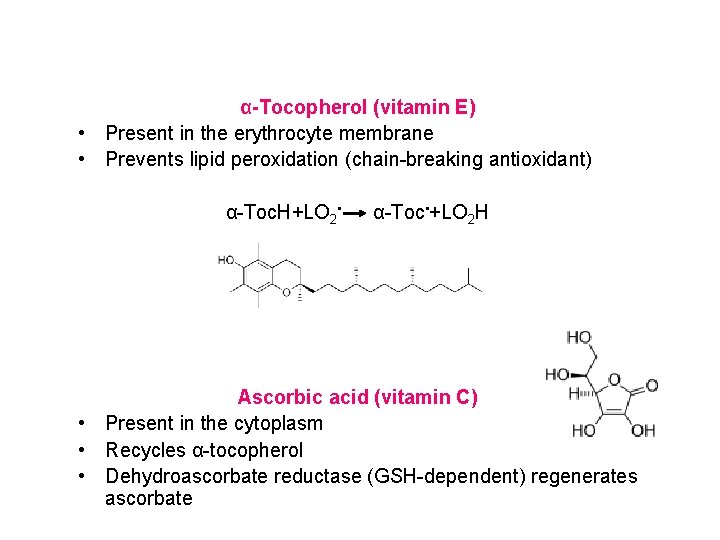
α-Tocopherol (vitamin E) • Present in the erythrocyte membrane • Prevents lipid peroxidation (chain-breaking antioxidant) α-Toc. H+LO 2 • α-Toc • +LO 2 H Ascorbic acid (vitamin C) • Present in the cytoplasm • Recycles α-tocopherol • Dehydroascorbate reductase (GSH-dependent) regenerates ascorbate

Haemoglobinopathy • abnormal structure of the haemoglobin (mutation) • large number of haemoglobin mutations, a fraction has deleterious effects • sickling, change in O 2 affinity, heme loss or dissociation of tetramer • haemoglobin M and S, and thalassemias Haemoglobin M • replacement of the histidine (E 8 or F 7) in α or β-chain by the tyrosine • the iron in the heme group is in the Fe 3+ state (methaemoglobin) stabilized by the tyrosine • methaemoglobin can not bind oxygen Thalassemias • genetic defects- α or β-chains are not produced (α or β-thalassemia)
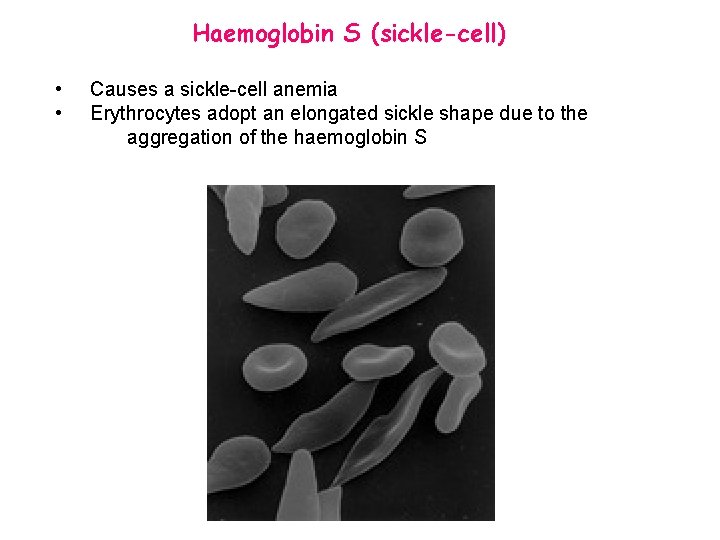
Haemoglobin S (sickle-cell) • • Causes a sickle-cell anemia Erythrocytes adopt an elongated sickle shape due to the aggregation of the haemoglobin S
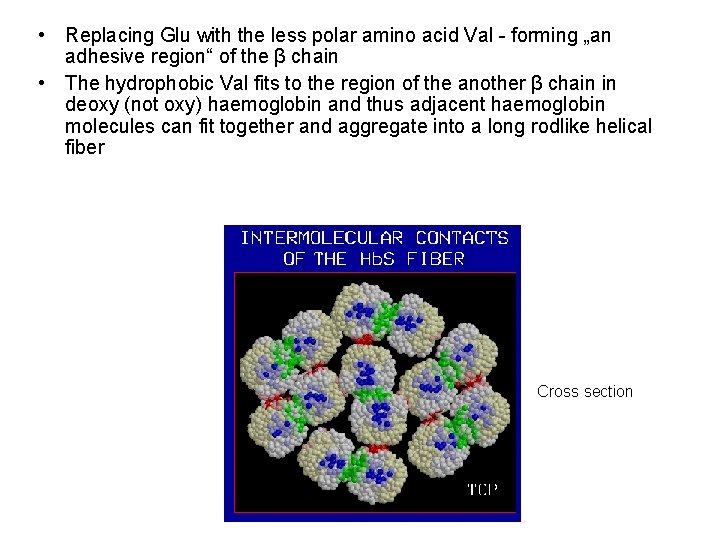
• Replacing Glu with the less polar amino acid Val - forming „an adhesive region“ of the β chain • The hydrophobic Val fits to the region of the another β chain in deoxy (not oxy) haemoglobin and thus adjacent haemoglobin molecules can fit together and aggregate into a long rodlike helical fiber Cross section
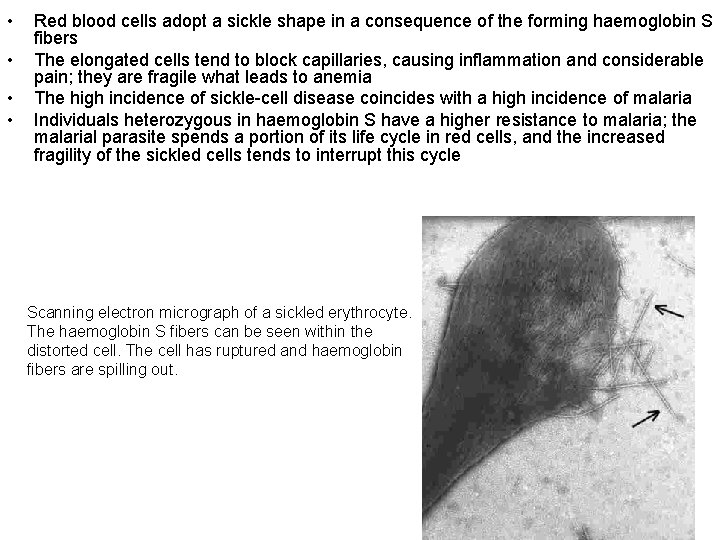
• • Red blood cells adopt a sickle shape in a consequence of the forming haemoglobin S fibers The elongated cells tend to block capillaries, causing inflammation and considerable pain; they are fragile what leads to anemia The high incidence of sickle-cell disease coincides with a high incidence of malaria Individuals heterozygous in haemoglobin S have a higher resistance to malaria; the malarial parasite spends a portion of its life cycle in red cells, and the increased fragility of the sickled cells tends to interrupt this cycle Scanning electron micrograph of a sickled erythrocyte. The haemoglobin S fibers can be seen within the distorted cell. The cell has ruptured and haemoglobin fibers are spilling out.

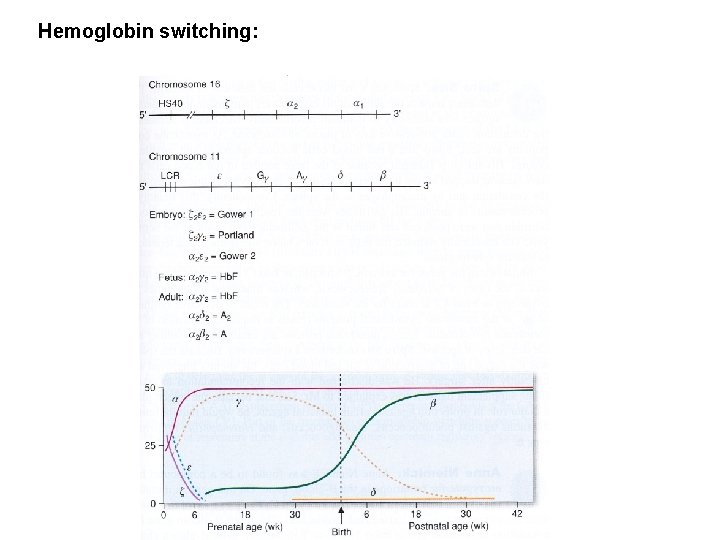
Hemoglobin switching:

Glycosylated haemoglobin (Hb. A 1) • • formed by hemoglobin's exposure to high plasma levels of glucose non-enzymatic glycolysation (glycation)- sugar bonding to a protein normal level Hb. A 1 - 5%; a buildup of Hb. A 1 - increased glucose concentration the Hb. A 1 level is proportional to average blood glucose concentration over previous weeks; in individuals with poorly controlled diabetes, increases in the quantities of these glycated hemoglobins are noted (patients monitoring) Sugar CHO Sugar CH + NH 2 N CH 2 Protein Schiff base Amadori reaction Sugar CH 2 NH CH 2 Protein Glycosylated protein
- Slides: 32