Empirical Formulas Empirical Formula Lowest whole ratio H






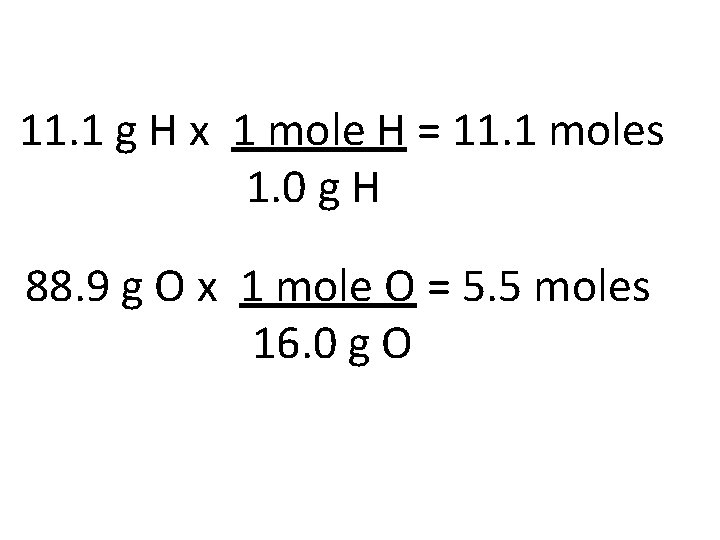




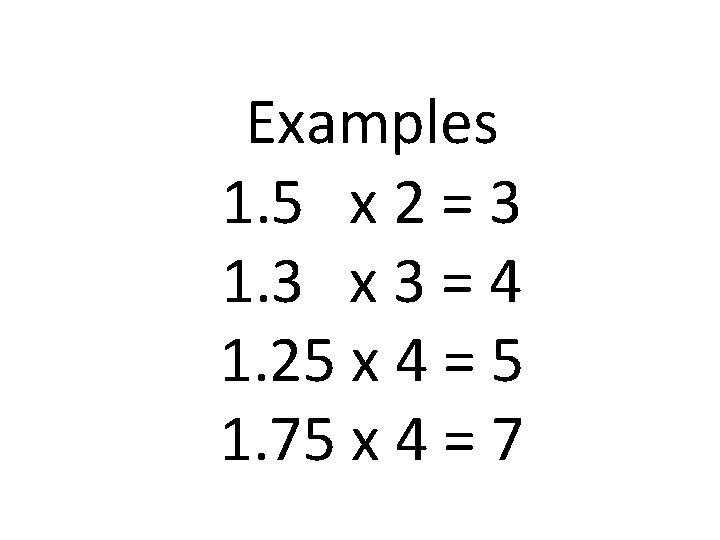
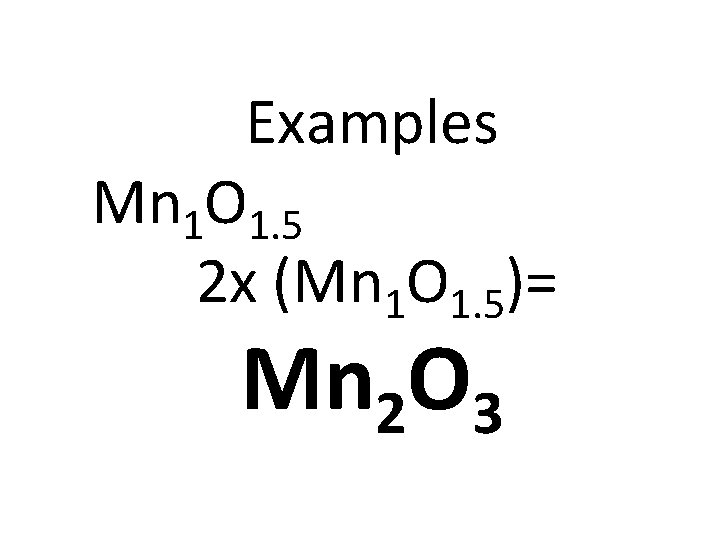




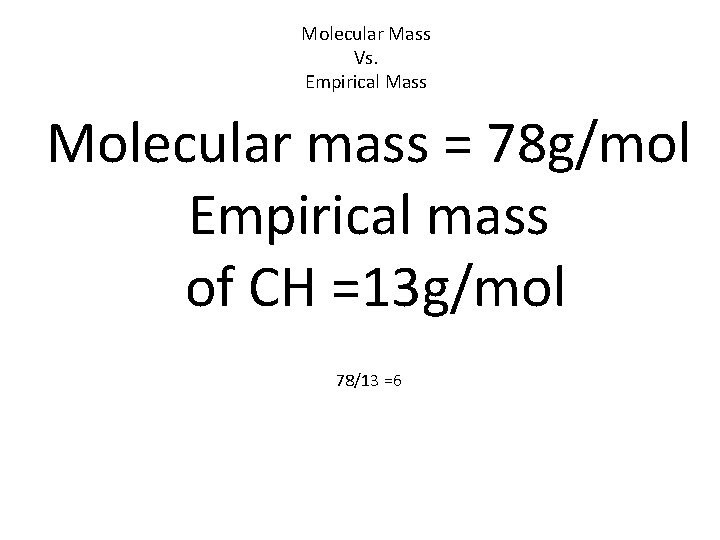
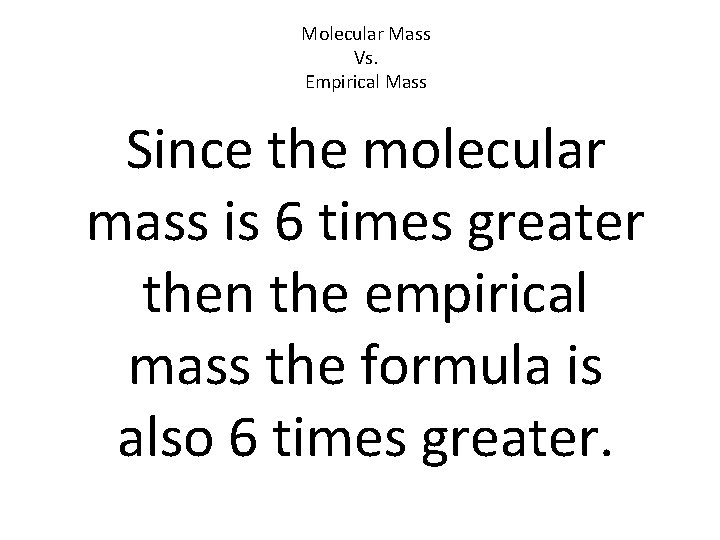
- Slides: 22

Empirical Formulas

Empirical Formula: Lowest whole # ratio H 2 O 2 (hydrogen peroxide) is it a empirical Formula? No, you can reduce it to HO

H 2 O 2 is the molecular formula Molecular formula shows the way the molecule is actually found in nature.

How do we write empirical formulas? ? ? 1. Take the % compositions and convert the % to grams.

11. 1 % H 88. 9% O changes to 11. 1 g H 88. 9 g O

1. Take the % compositions and convert the % to grams. 2. Convert grams to moles
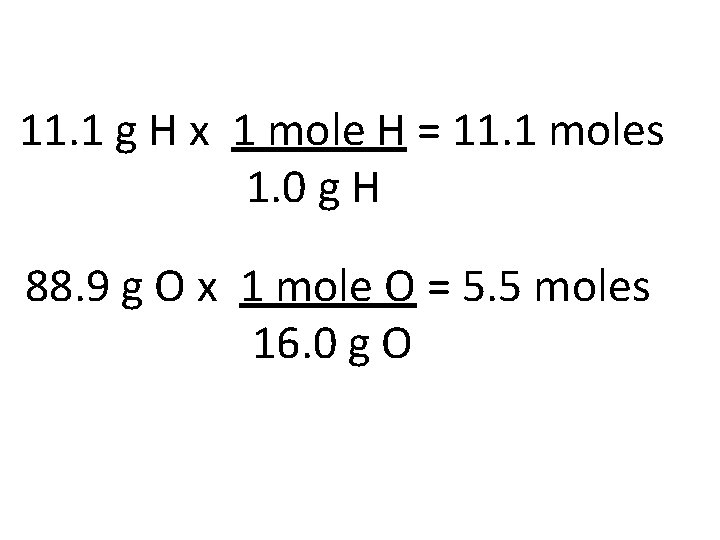
11. 1 g H x 1 mole H = 11. 1 moles 1. 0 g H 88. 9 g O x 1 mole O = 5. 5 moles 16. 0 g O

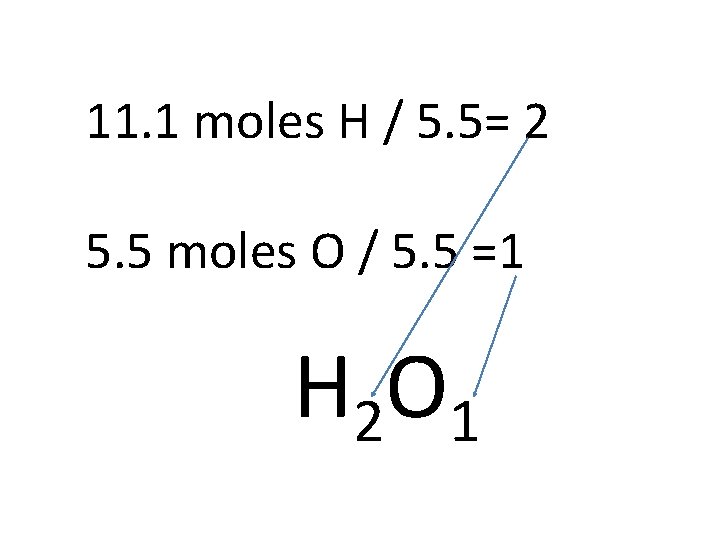
1. Take the % compositions and convert the % to grams. 2. Convert grams to moles 3. Divide by the smallest # of moles

11. 1 moles H / 5. 5= 2 5. 5 moles O / 5. 5 =1

1. Take the % compositions and convert the % to 4. Plug the whole #’s into grams. the empirical formula 2. Convert grams to moles 3. Divide by the smallest # of moles

11. 1 moles H / 5. 5= 2 5. 5 moles O / 5. 5 =1 H 2 O 1

1. 2. 3. 4. Take the % compositions and convert the % to grams. Convert grams to moles Divide by the smallest # of moles Plug the whole #’s into the empirical formula 5. If you do not have whole #’s after dividing you must multiply through to make them all whole #’s
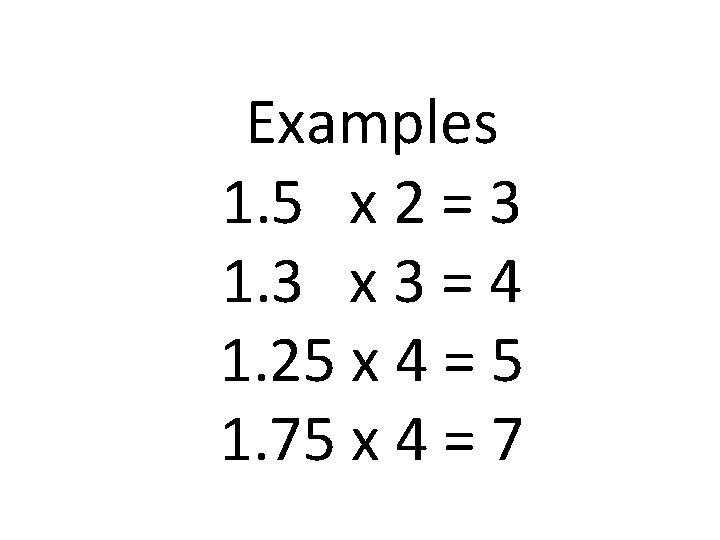
Examples 1. 5 x 2 = 3 1. 3 x 3 = 4 1. 25 x 4 = 5 1. 75 x 4 = 7
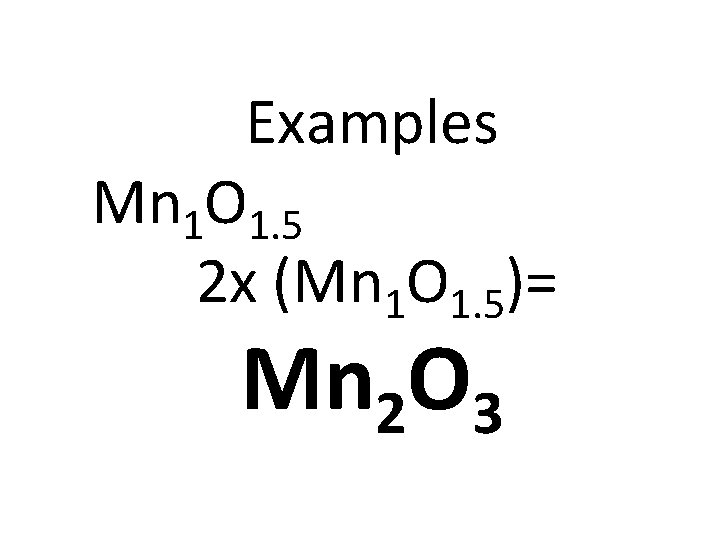
Examples Mn 1 O 1. 5 2 x (Mn 1 O 1. 5)= Mn 2 O 3
Molecular Mass Vs. Empirical Mass

Molecular Mass Is found experimentally

Empirical Mass Is found using the molar mass from the periodic table.

Molecular Mass Vs. Empirical Mass If they are the same GREAT the Molecular Formula and Empirical Formula are the same

Molecular Mass Vs. Empirical Mass If they are not the same you must divide the Molecular mass by the Empirical mass to see how many time greater it is.
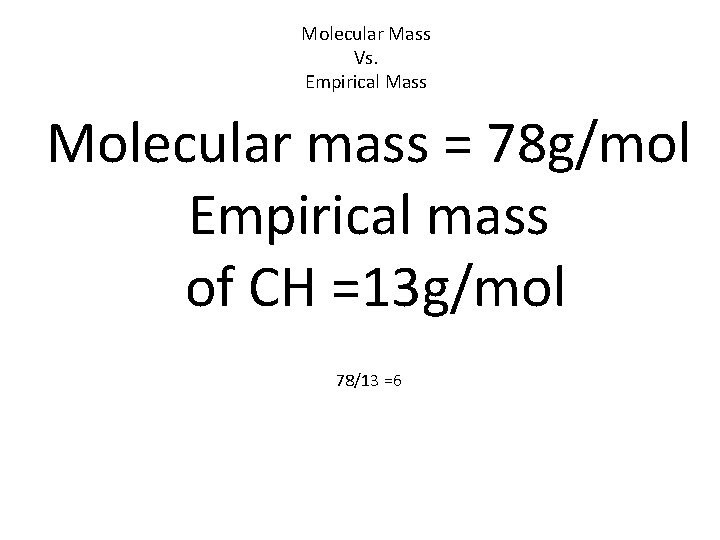
Molecular Mass Vs. Empirical Mass Molecular mass = 78 g/mol Empirical mass of CH =13 g/mol 78/13 =6
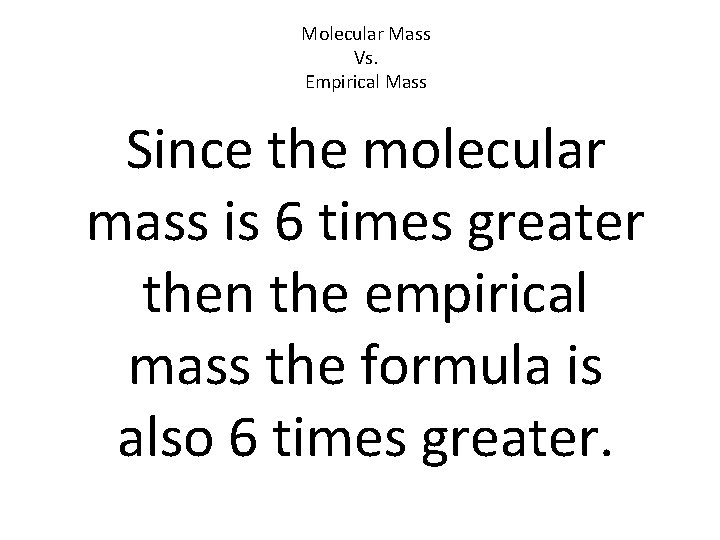
Molecular Mass Vs. Empirical Mass Since the molecular mass is 6 times greater then the empirical mass the formula is also 6 times greater.

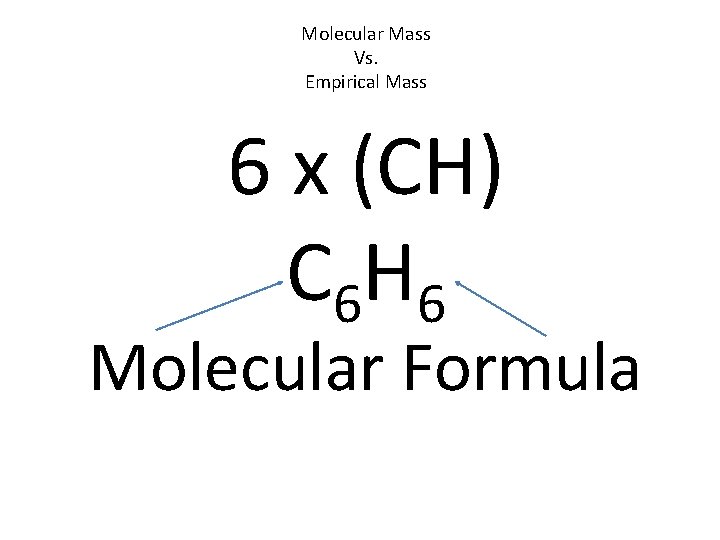
Molecular Mass Vs. Empirical Mass 6 x (CH) C 6 H 6 Molecular Formula