Empirical Formulas Empirical formula lowest wholenumber ratio of

Empirical Formulas

• Empirical formula – • lowest whole-number ratio of the elements in a compound • (divide by – Greatest Common Factor) • What is the empirical formula of C 2 N 2 H 8 • CNH 4

• Empirical Formula Calculations • 1. ) Change the given information to moles for each element. • 2. ) Divide each mole answer by the smallest number of moles. • 3. ) If the answers from step 2 are not whole numbers then divide by the smallest decimal part. • 4. ) Write the formula with the mole ratio as subscripts.

• EX 1 Find the empirical formula. • A compound contains 9. 31 g of Ag and 0. 69 g of O.

Ag 2 O

• EX 2 A compound contains 25. 9% N and 74. 1% O. • Find the empirical formula.
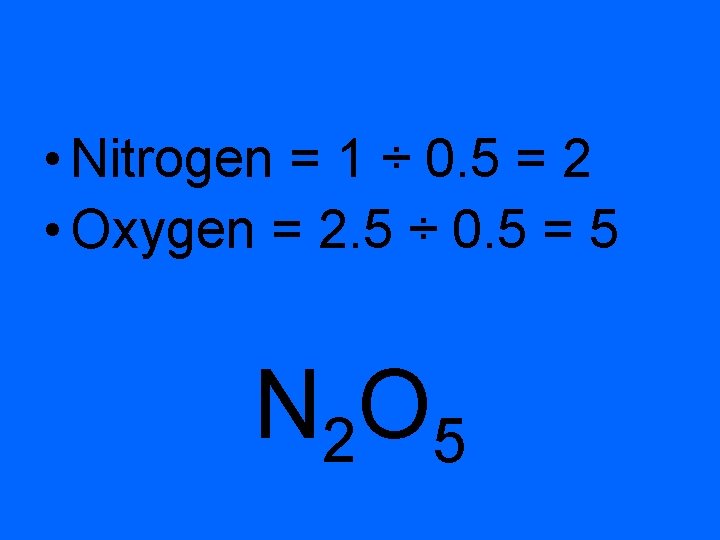
• Nitrogen = 1 ÷ 0. 5 = 2 • Oxygen = 2. 5 ÷ 0. 5 = 5 N 2 O 5

MOLECULAR FORMULAS

• Molecular formula – empirical formula or its multiple • Several compounds can have the same empirical formula but different molecular formulas. • • What is the empirical formula of C 2 N 2 H 8? CNH 4 What is the molecular formula of CNH 4? C 2 N 2 H 8

• Convert molecular empirical – Divide subscripts by greatest common factor • Convert empirical molecular – multiply subscripts by …

• EX 1 • The empirical formula is CH 4 N and the molar mass is 60. 0 g. Find the molecular formula. • • C 1 X 12. 0 = 12. 0 g 4 H 4 X 1. 0 = 4. 0 g N 1 X 14. 0 = 14. 0 g 30. 0 g C 2 H 8 N 2
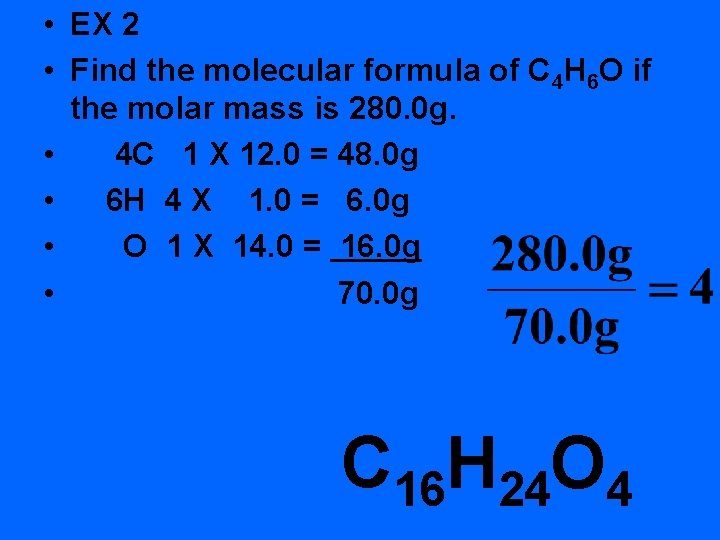
• EX 2 • Find the molecular formula of C 4 H 6 O if the molar mass is 280. 0 g. • 4 C 1 X 12. 0 = 48. 0 g • 6 H 4 X 1. 0 = 6. 0 g • O 1 X 14. 0 = 16. 0 g • 70. 0 g C 16 H 24 O 4
- Slides: 12