Destruction of Organic Pollutants Through Zero Valent Metals

![Redox Processes in Environmental, Analytical, & Biological Chemistries -I] Destruction of Organic Pollutants by Redox Processes in Environmental, Analytical, & Biological Chemistries -I] Destruction of Organic Pollutants by](https://slidetodoc.com/presentation_image/2a9ed88085660b8196b36acb655e3e7e/image-2.jpg)
![I] Redox Pathways for Pollutant Destruction The Search for Alternatives for the Bulk Destruction I] Redox Pathways for Pollutant Destruction The Search for Alternatives for the Bulk Destruction](https://slidetodoc.com/presentation_image/2a9ed88085660b8196b36acb655e3e7e/image-3.jpg)






























- Slides: 53

Destruction of Organic Pollutants Through Zero. Valent Metals Via Reductive and Oxidative Pathways Department of Chemistry, University of Idaho Moscow, ID 83844 -2343, Tel (208) 885 -6387 ifcheng@uidaho. edu Frank Cheng, Assistant Professor of Chemistry Graduate Students Tina Noradoun Undergraduates Tim Cantrell Matt Mc. Laughlin Kristy Henscheid 9/15/2020 Mark Engelmann Jose’ Morales Ryan Hutcheson John Doyle Ryan Neale Teri. Ann Miles Edmund Wong Kevin Breen Layne Pitcher Erik Parker 1
![Redox Processes in Environmental Analytical Biological Chemistries I Destruction of Organic Pollutants by Redox Processes in Environmental, Analytical, & Biological Chemistries -I] Destruction of Organic Pollutants by](https://slidetodoc.com/presentation_image/2a9ed88085660b8196b36acb655e3e7e/image-2.jpg)
Redox Processes in Environmental, Analytical, & Biological Chemistries -I] Destruction of Organic Pollutants by Reductive and Oxidative Pathways -RTP Reductive Dechlorination of Halocarbons -RTP Hydrogenation of Aromatics -Hydrocarbon Skeleton GC/HPLC -RTP O 2 oxidation of Organic Pollutants -II] Understanding Redox Pathways in the Mechanisms of Dietary Antioxidants -Flavonoids -Salicylate 9/15/2020 2
![I Redox Pathways for Pollutant Destruction The Search for Alternatives for the Bulk Destruction I] Redox Pathways for Pollutant Destruction The Search for Alternatives for the Bulk Destruction](https://slidetodoc.com/presentation_image/2a9ed88085660b8196b36acb655e3e7e/image-3.jpg)
I] Redox Pathways for Pollutant Destruction The Search for Alternatives for the Bulk Destruction of Organic Pollutants « Field-based Destruction of Pollutant Through O 2 Activation; RTP Mineralization. « 9/15/2020 PCB, DDT, Pentachlorophenol, phenol Organophosphorous Nerve Agents 3

PCB History & Background • 209 possible congeners • Dielectric fluid, oil-based solvents • Valued for chemical stability and fire resistance • Used as insulating fluids and coolants in electrical equipment and machinery from 1929 -1977 • 150 million pounds of PCBs are dispersed throughout the environment, an additional 290 million pounds are located in landfills and storage in the USA • Toxic Substances Control Act 1978 9/15/2020 http: //www. epa. gov/history/topics/pcbs/01. htm, Analtyical Chemistry of PCBs, 2 nd Ed. M. E. Erikson, CRC Press 1997 4

Present Method of PCB Destruction Incineration Problems with Incineration « Dioxin Emission; T > 1300 0 C « Costs « NIMBY § DDT - Pentachloropehnol 9/15/2020 5

Ideal Characteristics for Pollutant Destruction Method Minimal Costs & Environmental Impact Abundant Reagents Spent Reagents & Products Must Have Minimal Environmental Consequences if Released Mild Reaction Conditions, i. e. aqueous, room temperature and pressure, (RTP) Field Portability 9/15/2020 6

Outline of Redox Pathways Reductive: M(0) + H+ + R-Cl R-H + M 2+ + Cl. M(0) + 2 H+ + -R=R- -RH-RH- + M 2+ Oxidative: Fe(0) + O 2 + 2 H+ Fe 2+ + H 2 O 2 9/15/2020 7

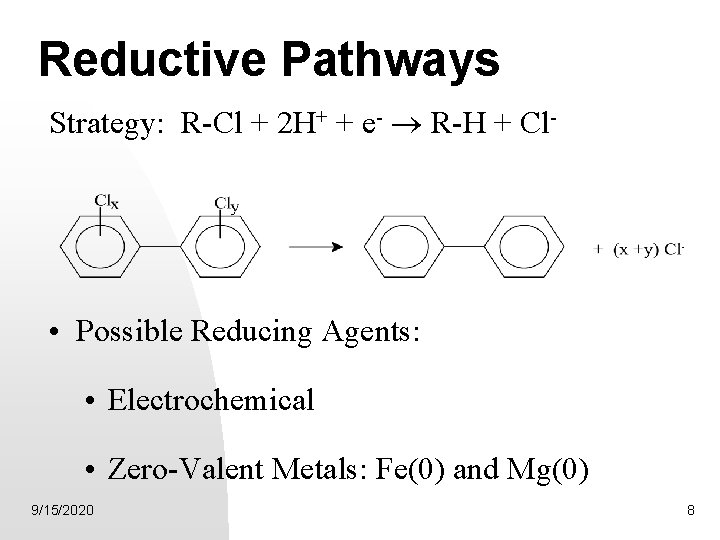
Reductive Pathways Strategy: R-Cl + 2 H+ + e- ® R-H + Cl- • Possible Reducing Agents: • Electrochemical • Zero-Valent Metals: Fe(0) and Mg(0) 9/15/2020 8

Reductive Dechlorination by Zero-Valent Metals M(0) Mn+ + ne- Eanode R-Cl + H+ + e- R-H + Cl- Ecathode Fe(0) + R-Cl + H+ Fe 2+ + R-H + Cl- Ecell • Both Fe(0) and Mg(0) are inexpensive • Both Fe 2+ and Mg 2+ are environmentally benign 9/15/2020 9

Thermodynamics of CCl 4 Dechlorination 9/15/2020 10

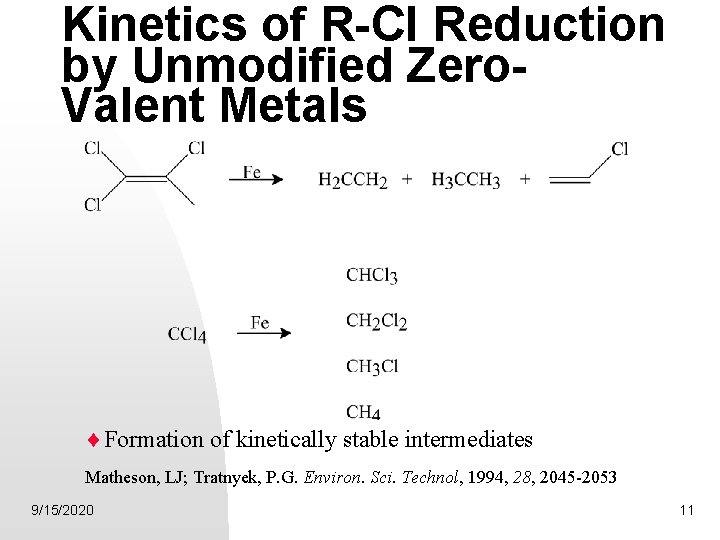
Kinetics of R-Cl Reduction by Unmodified Zero. Valent Metals Formation of kinetically stable intermediates Matheson, LJ; Tratnyek, P. G. Environ. Sci. Technol, 1994, 28, 2045 -2053 9/15/2020 11

PCBs are Kinetically Stable in the Presence of Fe(0) or Mg(0) 9/15/2020 12

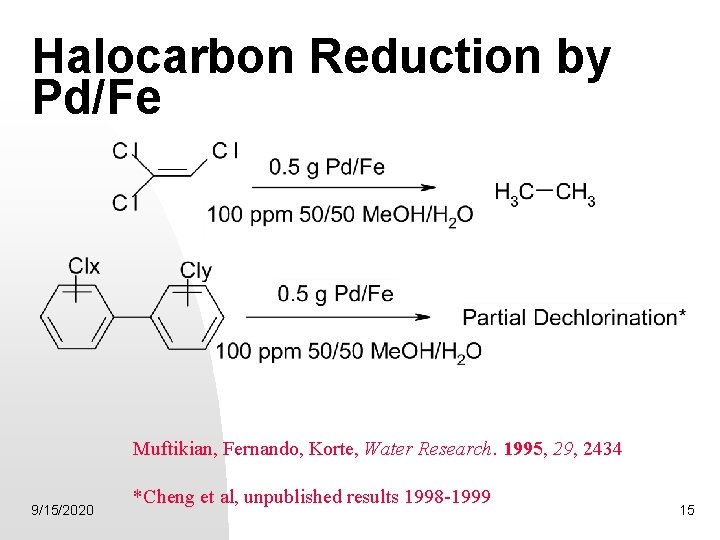
Catalyzed Reductions of Halocarbons by Pd modified Bimetallic Systems 2 M + Pd. Cl 62 - 2 M 2+ + 6 Cl- + Pd(s) 0. 05 -0. 25 % Pd (w/w) Results in complete dechlorination of TCE 9/15/2020 Muftikian, Fernando, Korte, Water Research. 1995, 29, 2434. 13

The role of Pd in hydrodehalogenation of chloro-organics by Pd/Fe Cheng, Fernando, Korte, Environ Sci &Technol, 1997, 31(4) 1074 -1078. 9/15/2020 14

Halocarbon Reduction by Pd/Fe Muftikian, Fernando, Korte, Water Research. 1995, 29, 2434 9/15/2020 *Cheng et al, unpublished results 1998 -1999 15

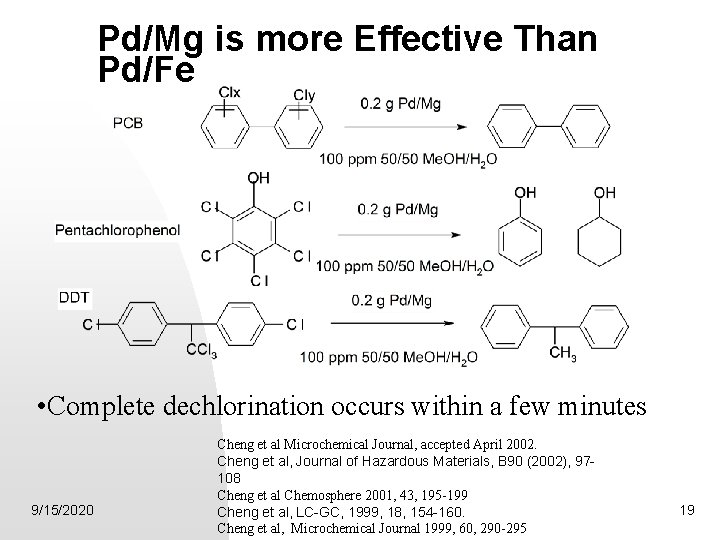
Reductive Dechlorination by Pd/Mg • Zero-valent Metals as Reducing Agents Fe(s) ® Fe 2+ + 2 e- E 0 red = -0. 44 volts Mg(s) ® Mg 2+ + 2 e- E 0 red = -2. 36 volts Na(s) ® Na+ + e- E 0 red = -2. 71 volts 9/15/2020 16

Reactivities of Metal Candidates Mn+ + n OH- M(OH)n(s) 9/15/2020 17

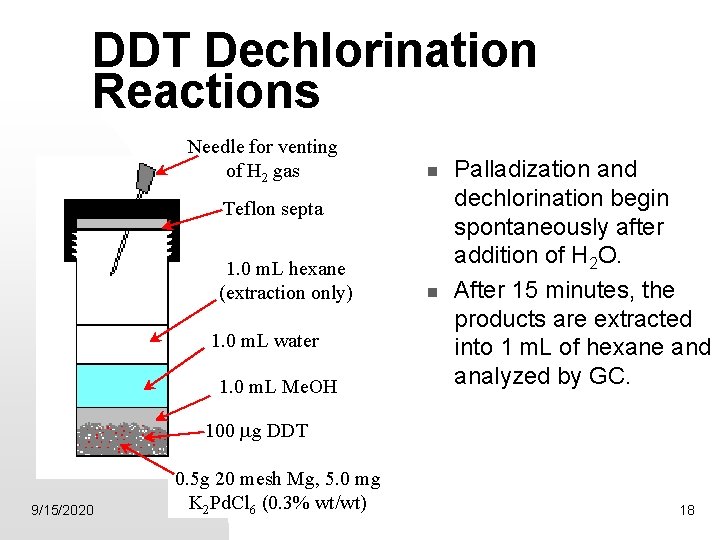
DDT Dechlorination Reactions Needle for venting of H 2 gas Teflon septa 1. 0 m. L hexane (extraction only) 1. 0 m. L water 1. 0 m. L Me. OH Palladization and dechlorination begin spontaneously after addition of H 2 O. After 15 minutes, the products are extracted into 1 m. L of hexane and analyzed by GC. 100 mg DDT 9/15/2020 0. 5 g 20 mesh Mg, 5. 0 mg K 2 Pd. Cl 6 (0. 3% wt/wt) 18

Pd/Mg is more Effective Than Pd/Fe • Complete dechlorination occurs within a few minutes 9/15/2020 Cheng et al Microchemical Journal, accepted April 2002. Cheng et al, Journal of Hazardous Materials, B 90 (2002), 97108 Cheng et al Chemosphere 2001, 43, 195 -199 Cheng et al, LC-GC, 1999, 18, 154 -160. Cheng et al, Microchemical Journal 1999, 60, 290 -295 19

Summary of Dechlorination by Pd/Mg First demonstration of the complete dechlorination of: PCP DDT PCB Reaction conditions: STP 9/15/2020 20

Hydrogenation of Phenol Common component of industrial waste streams. • As high as 1% m/m • Expensive treatment processes, i. e. high T and/or P processes, adsorption • Cyclohexanol/-one less problematic 9/15/2020 21

RTP Hydrogenation of Phenol 5 m. L aqueous 5 m. M Phenol, 10 minutes 1. Palladized 1. 00 g Mg particles 0. 079 m 2 2. Palladized 0. 66 g Fe particles 0. 079 m 2 3. 1. 00 g Mg 20 mesh - 0. 5 g 1/8” Pd/alumina Cheng et al; Industrial and Engineering Chemistry Research, 2002 41, 3071 -3074 9/15/2020 22

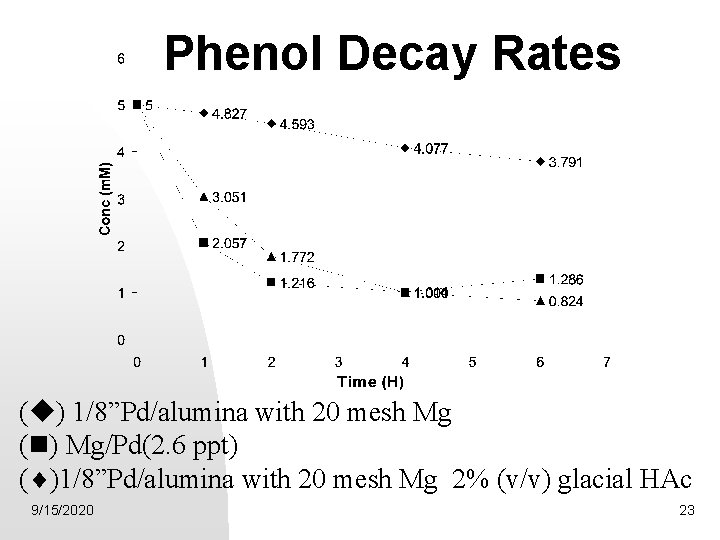
Phenol Decay Rates ( ) 1/8”Pd/alumina with 20 mesh Mg ( ) Mg/Pd(2. 6 ppt) ( )1/8”Pd/alumina with 20 mesh Mg 2% (v/v) glacial HAc 9/15/2020 23

RTP Hydrogenation Product Yields 9/15/2020 24

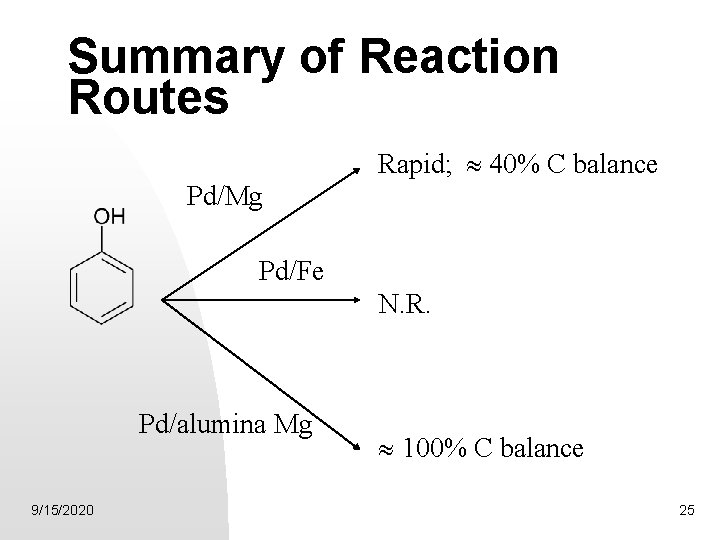
Summary of Reaction Routes Rapid; 40% C balance Pd/Mg Pd/Fe N. R. Pd/alumina Mg 9/15/2020 100% C balance 25

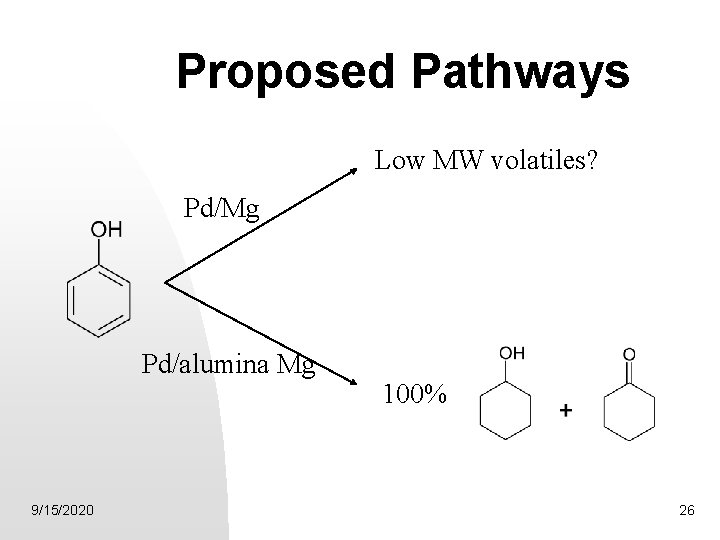
Proposed Pathways Low MW volatiles? Pd/Mg Pd/alumina Mg 9/15/2020 100% 26

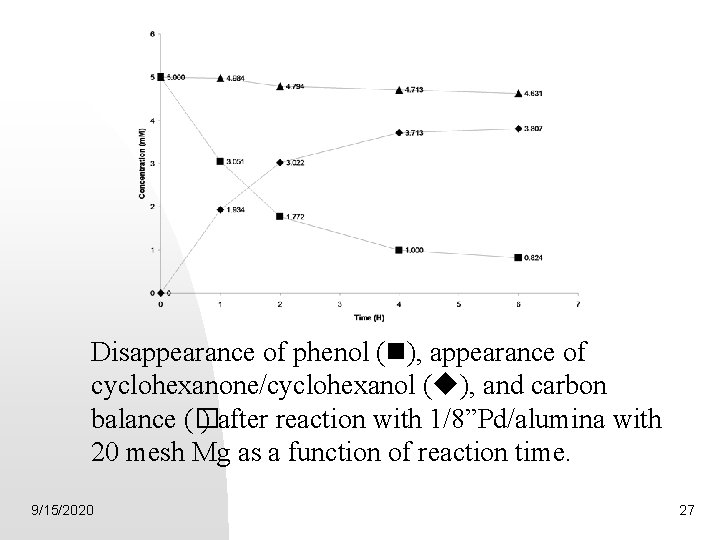
Disappearance of phenol ( ), appearance of cyclohexanone/cyclohexanol ( ), and carbon balance (� ) after reaction with 1/8”Pd/alumina with 20 mesh Mg as a function of reaction time. 9/15/2020 27

Analytical Chemistry of Halocarbon • Polychlorinated biphenyl, 209 possible conformation isomers (congeners) mixtures -problematic for GC-ECD -aging effects -environmental matrix more………. . • Complete dechlorination of PCB yields one product Biphenyl 9/15/2020 28

Typical GC-FID of a PCB Mixture 80 ppm of Arochlor 1260 Alltech AT-5 column; Splitless injection; initial temperature, 125 o. C; initial time, 5 min. ; rate, 10 o. C/min; final temperature, 275 o. C; final time, 5 min 9/15/2020 29

Treatment Procedure for Analytical Methods • Halocarbon mixture – 3 m. L of 100 ppb to 10 ppm in 50/50 isopropyl alcohol/water • Initial dechlorination agent – 0. 7 g 20 mesh Mg + 0. 4 mg K 2 Pd. Cl 6 • Reaction Time – 10 minutes • HPLC – C 18 reversed phase 65/35 (v/v) acetonitrile/water 200 nm absorbance detection 9/15/2020 30

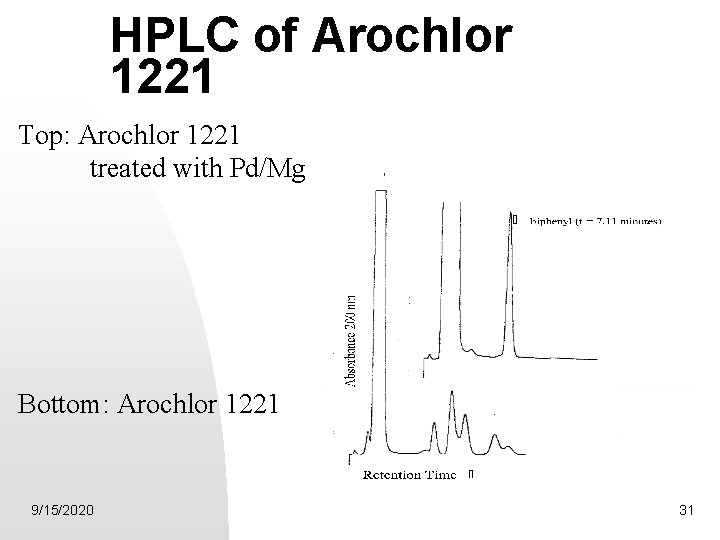
HPLC of Arochlor 1221 Top: Arochlor 1221 treated with Pd/Mg Bottom: Arochlor 1221 9/15/2020 31

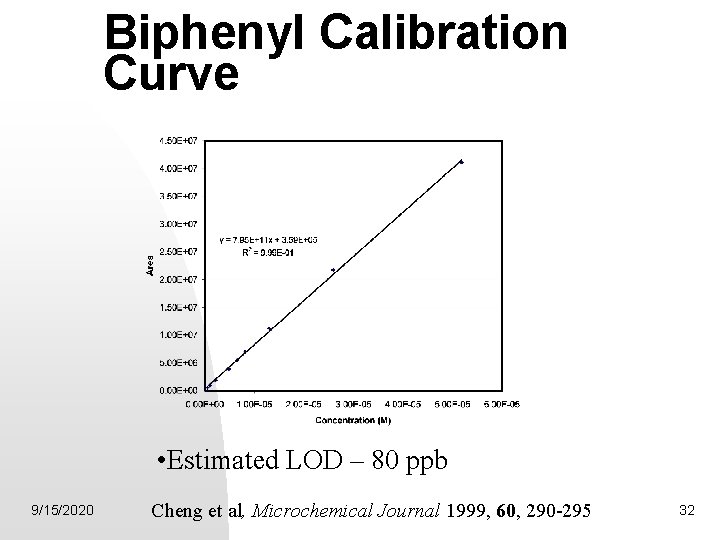
Biphenyl Calibration Curve • Estimated LOD – 80 ppb 9/15/2020 Cheng et al, Microchemical Journal 1999, 60, 290 -295 32

Biphenyl Yields From 1221 & 1260 9/15/2020 33

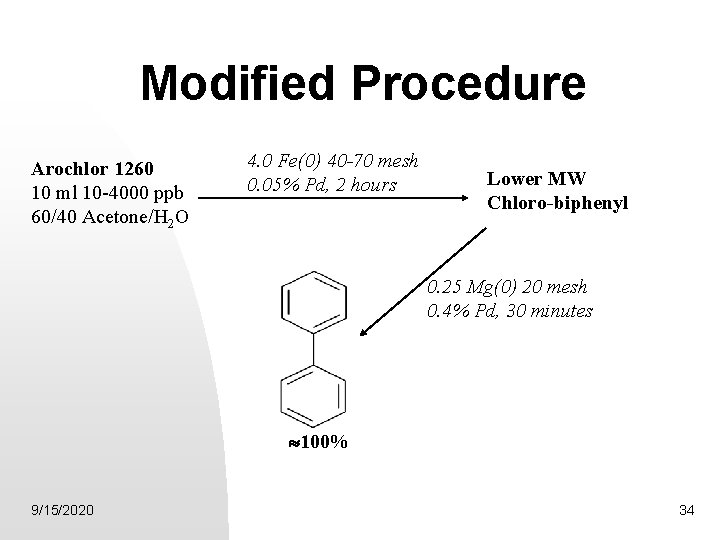
Modified Procedure Arochlor 1260 10 ml 10 -4000 ppb 60/40 Acetone/H 2 O 4. 0 Fe(0) 40 -70 mesh 0. 05% Pd, 2 hours Lower MW Chloro-biphenyl 0. 25 Mg(0) 20 mesh 0. 4% Pd, 30 minutes 100% 9/15/2020 34

Factors Controlling BP Yield • Reaction time with 0. 25 g Pd (0. 4%)/Mg • Acetone/Water (v/v) > 50% Acetone 100% yield < 50% yield decreases • 60/40 optimal for extraction into hexane 9/15/2020 35

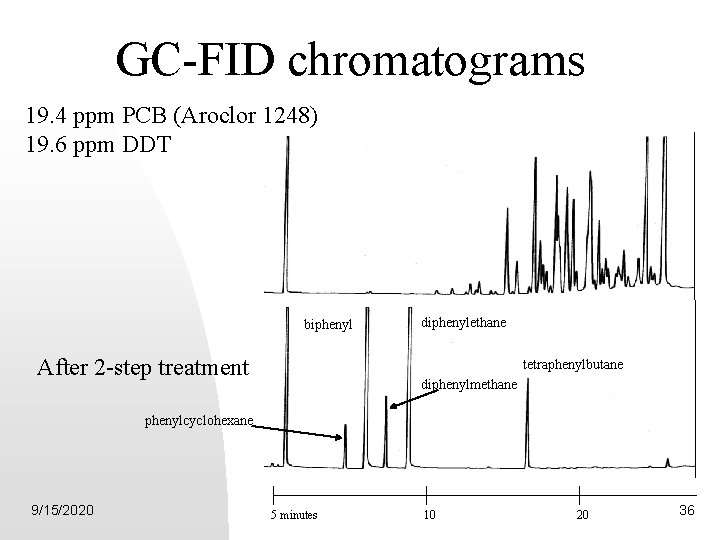
GC-FID chromatograms 19. 4 ppm PCB (Aroclor 1248) 19. 6 ppm DDT Internal Standard biphenyl After 2 -step treatment diphenylethane tetraphenylbutane diphenylmethane phenylcyclohexane 9/15/2020 5 minutes 10 20 36

Detection Limits • 100 ppb for DDT & 40 ppb for PCB • EPA Methods 508 A, 8081 A, & 8082 - 0. 6 ppb for DDT & 0. 14 ppb for PCB • Advantages over EPA methods - Simplified procedure - Better Precision - Faster work up time - Ability to resolve halocarbon interferences 9/15/2020 37

Oxidative Pathways -Active Oxygen -Oxidations by O 2 from air -Cx. Hy. Clz + O 2 = CO 2 + H 2 O + HCl (unbalanced) G<0 -Room temperature oxidations by air are kinetically slow -enzymatic or enzyme-mimics -Partially reduced O 2 (active oxygen) 9/15/2020 38

Partially Reduced Oxygen E 0 reduction 9/15/2020 O 2 + e- = O 2. - -0. 45 volts O 2 + 2 H+ + 2 e- = H 2 O 2 +0. 30 volts O 2 + 4 H+ + 4 e- = H 2 O +1. 23 volts 39

Fenton Reaction -Reduction of HO-OH H 2 O 2 + Fe. II = Fe. III + HO- + HO. + e- = HO- Eo = 1. 8 volts -HO. reacts with organics with diffusion limited kinetics 9/15/2020 40

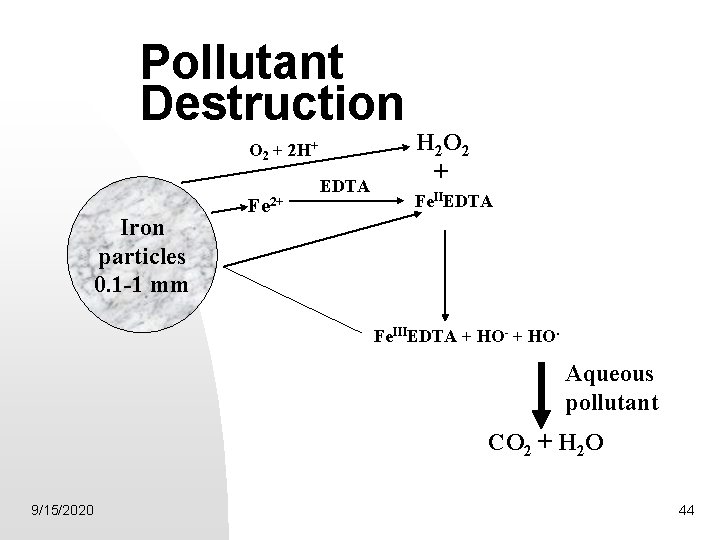
RTP Production of Active Oxygen O 2 reducing agents -Zero-valent iron, Fe(0) Inexpensive Spent agent, Fe 2+/Fe 3+ environmentally benign. Chelated forms, capable Fenton reaction agents Fe(0) + O 2 + 2 H+ Fe 2+ + H 2 O 2 9/15/2020 41

“Aero”-Fenton Reaction -Fe 2+ chelation agent - EDTA Fe. IIETDA + HOOH Fe. IIIEDTA + HO- + HO. • Inexpensive • Efficient Binding Agent for Fe 2+/3+ • Fe. IIEDTA highly effective Fenton Reaction reagent 9/15/2020 42

Proposed Overall Scheme Fe(0) + O 2(aq) + 2 H+(aq) Fe 2+(aq) + HOOH(aq) Fe 2+(aq) + EDTA Fe. IIEDTA + HOOH Fe. IIIEDTA + HO- + HO· Regeneration: 2 Fe. IIIEDTA + Fe(0) Fe 2+(aq) + 2 Fe. IIEDTA 9/15/2020 43

Pollutant Destruction O 2 + 2 H+ Iron particles 0. 1 -1 mm Fe 2+ EDTA H 2 O 2 + Fe. IIEDTA Fe. IIIEDTA + HO- + HO. Aqueous pollutant CO 2 + H 2 O 9/15/2020 44

Aero-Fenton Reaction • 0. 5 g of 40 -70 mesh Fe(0) • 10 m. L of solution • 0. 32 m. M EDTA • 140 ppm 4 -chlorophenol • 4 hour reaction time • GC and LC-MS • RTP 9/15/2020 45

Summary of degradation of the chlorophenols in this study All runs included 0. 5 g of 40 -70 mesh Fe(0) in 10 m. L of solution. . I) 0. 32 m. M EDTA Aerobic II) 0. 0 m. M EDTA Aerobic III) 0. 32 m. M EDTA Anaerobic (N 2 purge) IV) dark conditions 0. 32 m. M EDTA aerobic 140 ppm 4 chlorophenol Complete degradation after 4 h. (GCFID) Complete degradation after 70 h. (GCFID) No reaction after 4 h. Complete degradation after 4 h. No reaction after 70 h. N/A 162 ppm pentachlorophe nol suspension 9/15/2020 46

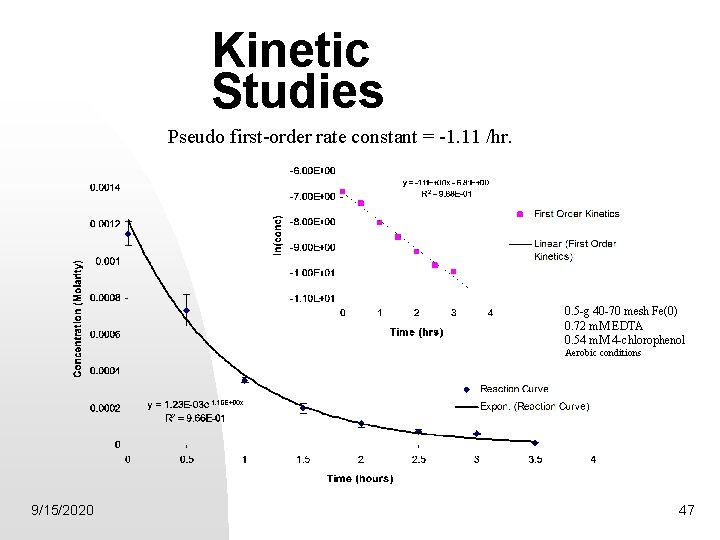
Kinetic Studies Pseudo first-order rate constant = -1. 11 /hr. 0. 5 -g 40 -70 mesh Fe(0) 0. 72 m. M EDTA 0. 54 m. M 4 -chlorophenol Aerobic conditions 9/15/2020 47

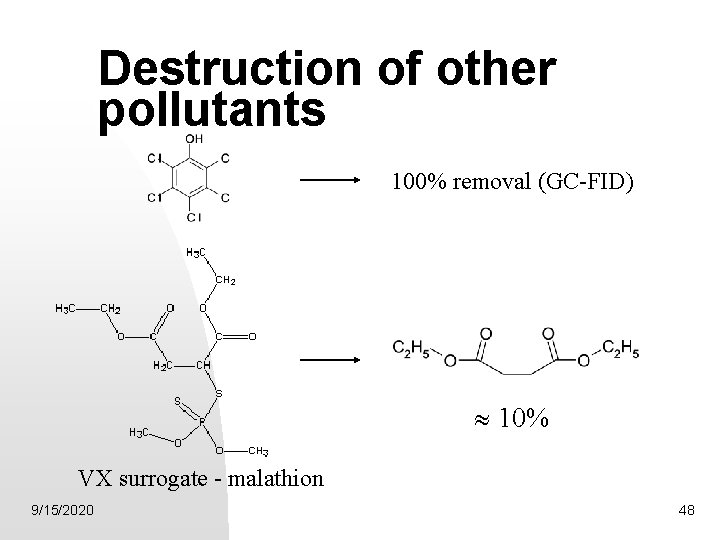
Destruction of other pollutants 100% removal (GC-FID) 10% VX surrogate - malathion 9/15/2020 48

Technical Summary Ability to degrade organic pollutants • Under room temperature, atmospheric pressure conditions • Inexpensive Reagents – Iron particles, water, air & EDTA • Unspecialized reactors • Process is easily transportable, iron particles & EDTA • Strong possibility of scale-up 9/15/2020 49

Scientific Summary • First example of abiotic RTP activation of O 2 able to oxidize destructively organics • Control experiments indicate process is dependent on Fe(0), EDTA, air, and water 9/15/2020 50

Future Investigations • Mechanisms – Understanding the process • Kinetics – Speeding up the process • Search for an oxidatively stable iron chelate • Survey of Pollutants – oxidizable and nonoxidizable functional groups • Application & Scale-up 9/15/2020 51

Acknowledgements 9/15/2020 52

Acknowledgements • Thank you for Attention • University of Idaho Research Foundation • Mark Engelmann – Ph. D. candidate – Dechlorination • Jose’ Morales – M. S. 2001 – Hydrogenation • Tina Noradoun – Ph. D. candidate – Aero-Fenton 9/15/2020 53