Staff Dan Solomon MD MPH BWH Jacklyn Stratton

Staff: Dan Solomon, MD MPH (BWH) Jacklyn Stratton (BWH) Jack Ellrodt (BWH) Expert Faculty: Josef Smolen, MD (University of Vienna) Theodore Pincus, MD (Rush Medical) Jeff Katz, MD MSc (BWH) Nancy Shadick, MD, MPH (BWH) Treat-to-target in RA: Collaboration To Improve adoption and adherence, Lite (The TRACTION Lite Learning Collaborative) October 2020 Boston, MA Sponsored by Abbvie 1

Agenda – Day 1 � 1: 00 -1: 15 Introductions and TRACTION LC Overview � 1: 15 -2: 00 Treat-to-Target Principles (J. Smolen) � 2: 00 -2: 45 Measuring Disease Activity in the Office (T. Pincus) � 2: 45 -3: 00 BREAK � 3: 00 -3: 45 What is a PDSA? (D. Solomon) � 3: 45 -4: 00 Wrap-Up 2

Introductions � Teams: Everyone give name and role in practice + team information by team leader � Staff/Faculty: � Guests: 3 Your role What organization you represent

Key Elements of the Learning Collaborative (LC) Dan Solomon, MD, MPH 4

Why a Learning Collaborative? � Institute for Healthcare Improvement (IHI)/ Associates in Process Improvement (API) (1995): Bridge the gap between knowledge and practice 5
Why a Learning Collaborative? � Institute for Healthcare Improvement (IHI)/ Associates in Process Improvement (API) (1995): Bridge the gap between knowledge and practice We are here 6

Core Elements of a Learning Collaborative � Multi-level, � Expert faculty � Shared learning environment � Change � “The 7 inclusive teams Package (framework to guide priorities) Model for Improvement”

Review LC Language and Terms � Change Package: Framework that guides all work of the teams, laying out the needs, opportunities, and elements that must be in place to achieve the Collaborative mission � Principle: Overarching theme or value in the Framework � Change Concept: Element that describes what the principle looks like on the ground; describe how teams will operationalize the principles � Model for Improvement: Three questions that teams use to connect their tests of change to the principles, change concepts, and measurable improvements in practice 8

The Importance of a Change Package for Implementation 9

The Model for Improvement Change Package Principle • What are we trying to accomplish? Monthly Metrics • How will we know a change is an improvement? Change Concept • What changes can we make that will result in improvement? Adapted from 2001 Institute for Healthcare Improvement 10

Treat-to-Target Principles Josef Smolen, MD Editor, Annals of the Rheumatic Diseases Professor of Medicine 11

TRANSFORMING STANDARDS OF CARE: A CALL FOR “TREAT TO TARGET” – AND A SALUTE TO “TRACTION” Josef S. Smolen Medical University of Vienna and Hietzing Hospital, Vienna, Austria

Disclosures • Research Grants: Abbvie, Astra. Zeneca, Lilly, Novartis and Roche • Honoraria for consultancies and/or speaking engagements: Abb. Vie, Amgen, Astra. Zeneca, Astro, Bristol-Myers Squibb, Celgene, Celltrion, Chugai, Gilead, ILTOO, Janssen, Lilly, Merck Sharp & Dohme, Novartis- Sandoz, Pfizer, Roche, Samsung, Sanofi, and UCB • Other: Co-Editor, Rheumatology 7 E/8 E; Convenor of EULAR Task Forces, Convenor of T 2 T Task Forces, Editor Annals of the Rheumatic Diseases

Strategic Management of Chronic Diseases Condition Characteristics Treatment Target Diabetes • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. <7% Hb. A 1 C Hypertension/ Cardiovascular disease • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. (LDL)-cholesterol target of 70 mg/d. L to decrease incidence of cardiac events BP: 140/90 (135/80 for diabetes patients) Rheumatoid Arthritis • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. ACR-EULAR remission or low disease activity Spondyloarthritis (Ps. A, AS) • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. Remission or low disease activity

Conclusion: Intensive therapy was successfully carried out in the present trial by an expert team of diabetologists, nurses, dietitians, and behavioral specialists, and the time, effort, and cost required were considerable. Because the resources needed are not widely available, new strategies are needed to adapt methods of intensive treatment for use in the general community at less cost and effort. Meanwhile, the health care system should provide the support necessary to make intensive therapy available to those patients who will benefit.
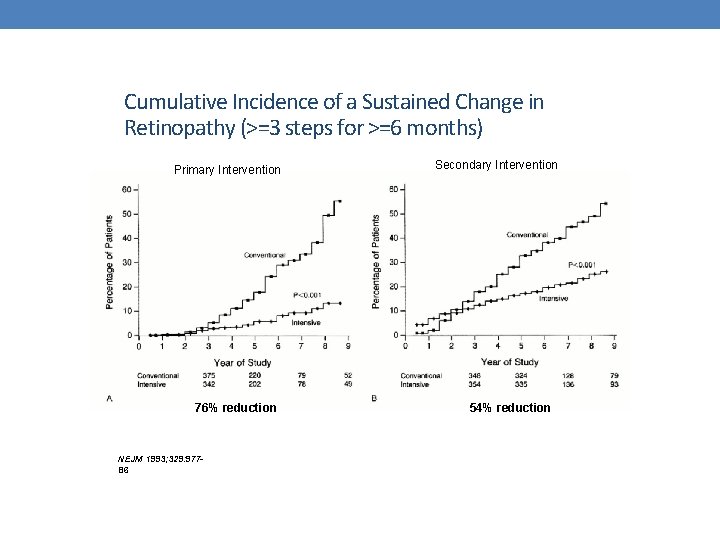
Cumulative Incidence of a Sustained Change in Retinopathy (>=3 steps for >=6 months) Primary Intervention 76% reduction NEJM 1993; 329: 97786 Secondary Intervention 54% reduction

Strategic Management of Chronic Diseases Condition Characteristics Treatment Target Diabetes • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. <7% Hb. A 1 C Hypertension/ Cardiovascular disease • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. (LDL)-cholesterol target of 70 mg/d. L to decrease incidence of cardiac events BP: 140/90 (135/80 for diabetes patients) Rheumatoid Arthritis • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. ACR-EULAR remission or low disease activity Spondyloarthritis (Ps. A, AS) • Chronic illness • If poorly controlled, it can lead to serious complications and disability • Complications often cause serious illness and premature death. Remission or low disease activity
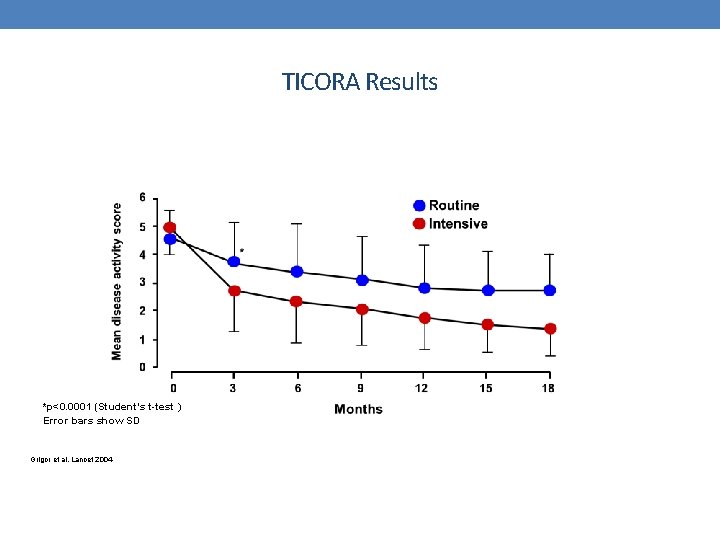
TICORA Results *p<0. 0001 (Student’s t-test ) Error bars show SD Grigor et al, Lancet 2004
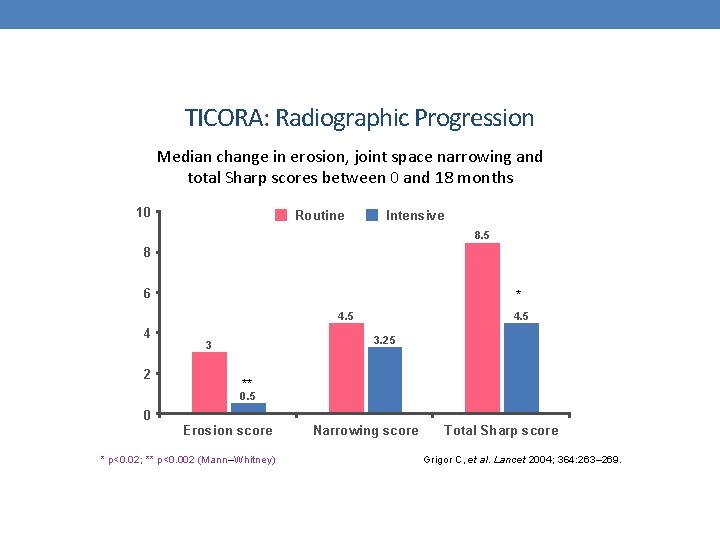
TICORA: Radiographic Progression Median change in erosion, joint space narrowing and total Sharp scores between 0 and 18 months 10 Routine Intensive 8. 5 8 6 * 4. 5 4 2 4. 5 3. 25 3 ** 0. 5 0 Erosion score * p<0. 02; ** p<0. 002 (Mann–Whitney) Narrowing score Total Sharp score Grigor C, et al. Lancet 2004; 364: 263– 269.
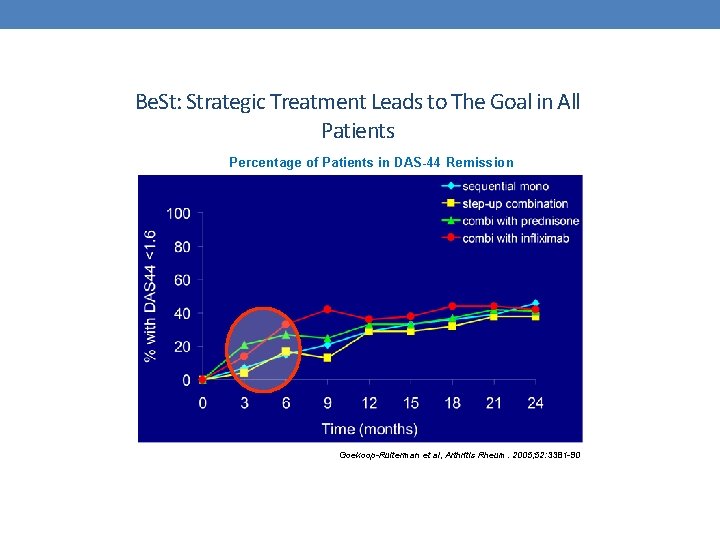
Be. St: Strategic Treatment Leads to The Goal in All Patients Percentage of Patients in DAS-44 Remission Goekoop-Ruiterman et al, Arthritis Rheum. 2005; 52: 3381 -90
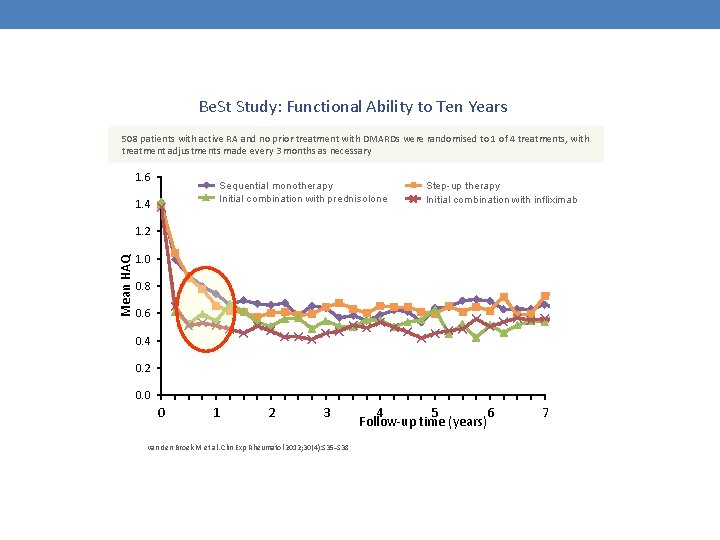
Be. St Study: Functional Ability to Ten Years 508 patients with active RA and no prior treatment with DMARDs were randomised to 1 of 4 treatments, with treatment adjustments made every 3 months as necessary 1. 6 Sequential monotherapy Initial combination with prednisolone 1. 4 Step-up therapy Initial combination with infliximab Mean HAQ 1. 2 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 0 1 2 3 van den Broek M et al. Clin Exp Rheumatol 2012; 30(4): S 35–S 38 4 5 6 Follow-up time (years) 7 8 9 10
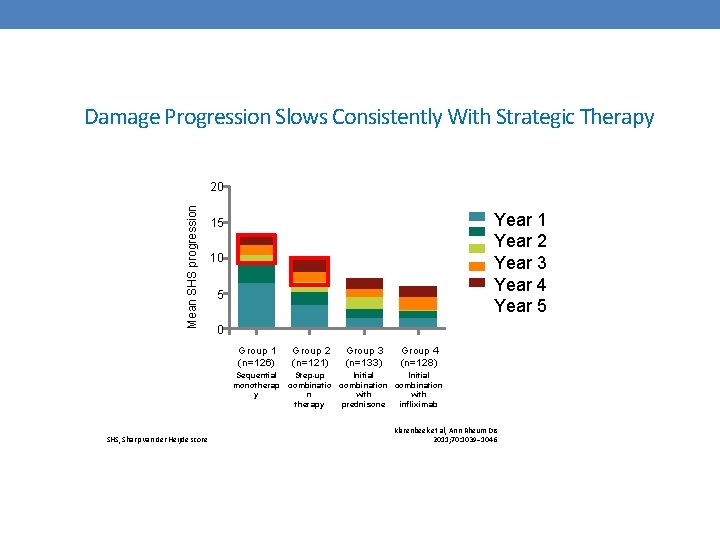
Damage Progression Slows Consistently With Strategic Therapy Mean SHS progression 20 Year 1 Year 2 Year 3 Year 4 Year 5 15 10 5 0 Group 1 (n=126) Group 2 (n=121) Group 3 (n=133) Group 4 (n=128) Initial Sequential Step-up monotherap combination with y n prednisone infliximab therapy SHS, Sharp van der Heijde score Klarenbeek et al, Ann Rheum Dis 2011; 70: 1039– 1046
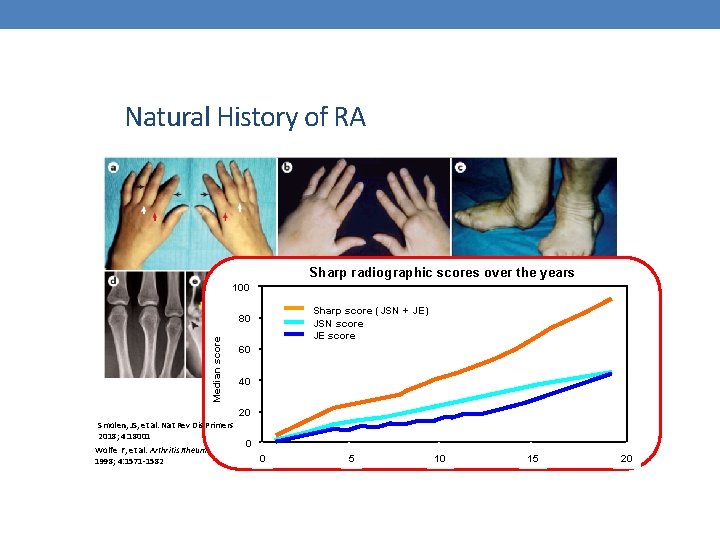
Natural History of RA Sharp radiographic scores over the years 100 Sharp score (JSN + JE) JSN score JE score Median score 80 60 40 20 Smolen, JS, et al. Nat Rev Dis Primers 2018; 4: 18001 Wolfe F, et al. Arthritis Rheum. 1998; 4: 1571 -1582 0 0 5 10 15 20
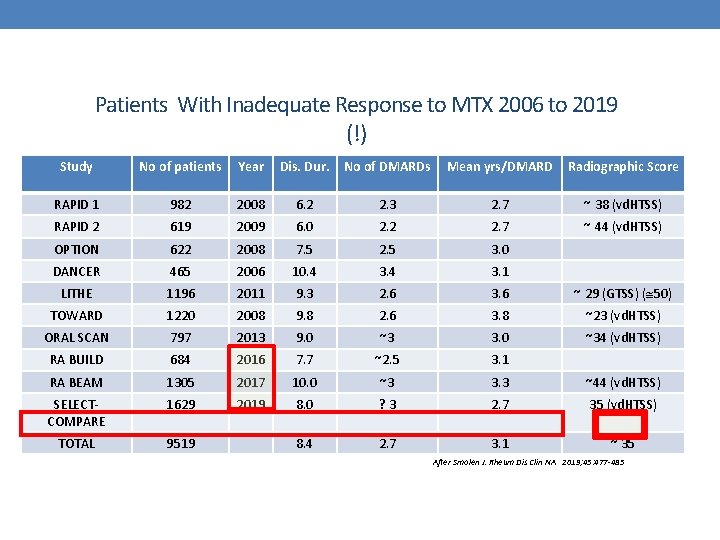
Patients With Inadequate Response to MTX 2006 to 2019 (!) Study No of patients Year Dis. Dur. No of DMARDs Mean yrs/DMARD Radiographic Score RAPID 1 982 2008 6. 2 2. 3 2. 7 ~ 38 (vd. HTSS) RAPID 2 619 2009 6. 0 2. 2 2. 7 ~ 44 (vd. HTSS) OPTION 622 2008 7. 5 2. 5 3. 0 DANCER 465 2006 10. 4 3. 1 LITHE 1196 2011 9. 3 2. 6 3. 6 ~ 29 (GTSS) ( 50) TOWARD 1220 2008 9. 8 2. 6 3. 8 ~23 (vd. HTSS) ORAL SCAN 797 2013 9. 0 ~3 3. 0 ~34 (vd. HTSS) RA BUILD 684 2016 7. 7 ~2. 5 3. 1 RA BEAM 1305 2017 10. 0 ~3 3. 3 ~44 (vd. HTSS) SELECTCOMPARE 1629 2019 8. 0 ? 3 2. 7 35 (vd. HTSS) TOTAL 9519 8. 4 2. 7 3. 1 ~ 35 After Smolen J. Rheum Dis Clin NA 2019; 45: 477 -485
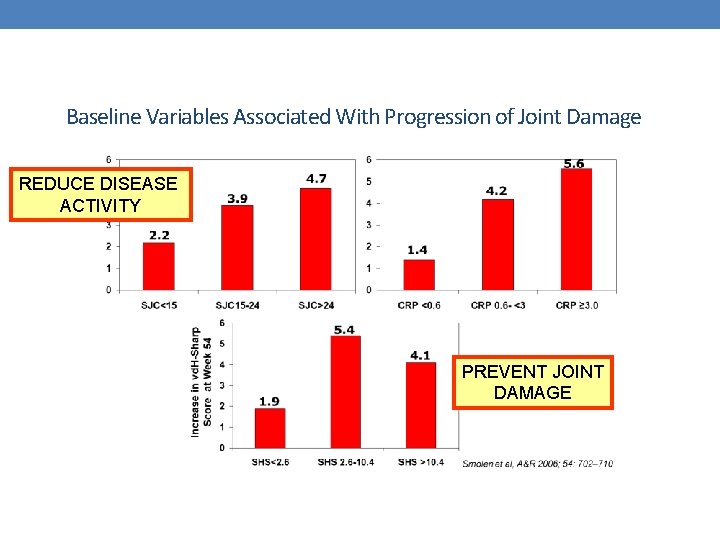
Baseline Variables Associated With Progression of Joint Damage REDUCE DISEASE ACTIVITY PREVENT JOINT DAMAGE
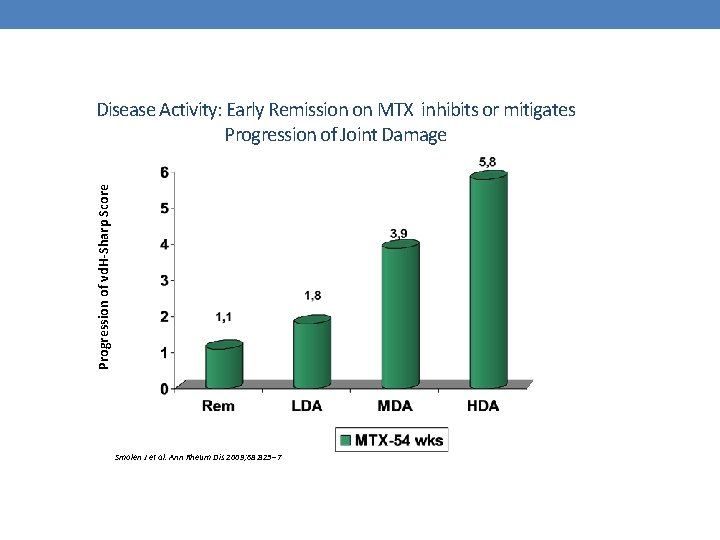
Progression of vd. H-Sharp Score Disease Activity: Early Remission on MTX inhibits or mitigates Progression of Joint Damage Smolen J et al. Ann Rheum Dis 2009; 68: 823– 7
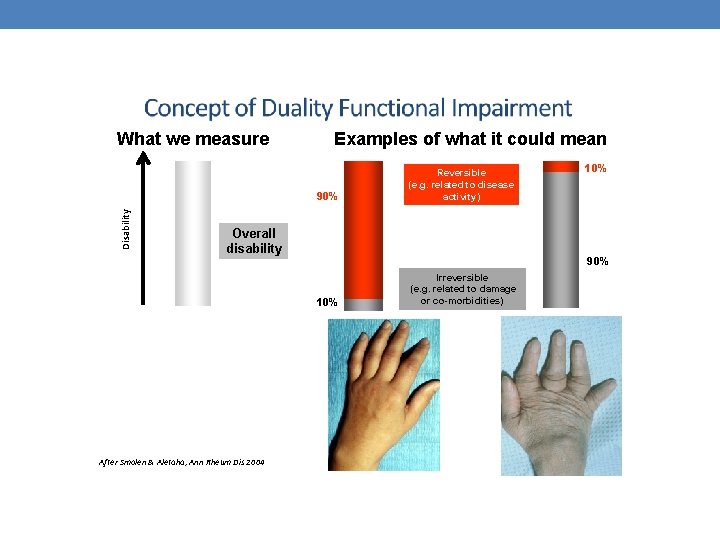
What we measure Examples of what it could mean Disability 90% Reversible (e. g. related to disease activity) Overall disability 90% 10% After Smolen & Aletaha, Ann Rheum Dis 2004 10% Irreversible (e. g. related to damage or co-morbidities)

Concept of Duality Functional Impairment What we measure Examples of what it could mean Disability 90% Reversible (e. g. related to disease activity) Overall disability 90% 10% Utilizing the concept of remission Removing the effect of active disease After Smolen & Aletaha, Ann Rheum Dis 2004 10% Irreversible (e. g. related to damage or co-morbidities)
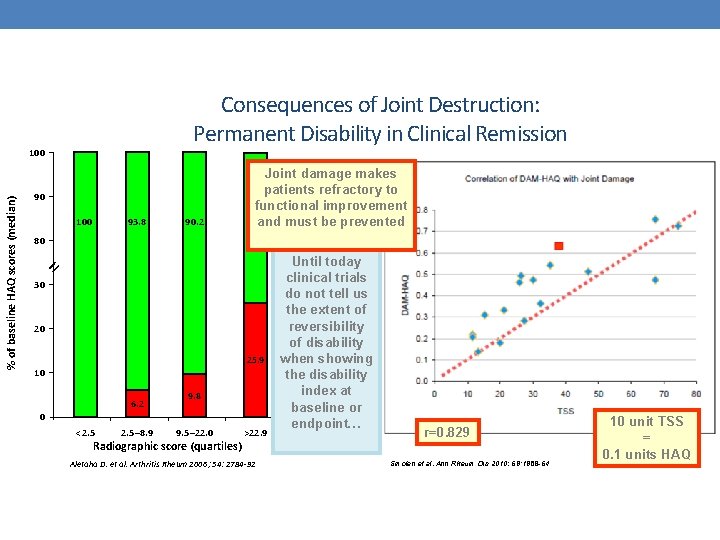
Consequences of Joint Destruction: Permanent Disability in Clinical Remission 90 100 93. 8 90. 2 Joint damage makes patients refractory to % Reversible functional improvement and must be prevented 74. 1 % Irreversible 80 = % of baseline HAQ scores (median) 100 30 20 25. 9 10 6. 2 9. 8 0 <2. 5– 8. 9 9. 5– 22. 0 Radiographic score (quartiles) >22. 9 Aletaha D. et al. Arthritis Rheum 2006; 54: 2784 -92 Until today clinical trials do not tell us the extent of reversibility of disability when showing the disability index at baseline or endpoint… r=0. 829 <2 2 -5 Smolen et al, Ann Rheum Dis 2010; 69: 1958 -64 5 -10 10 unit TSS >10 yrs = 0. 1 units HAQ
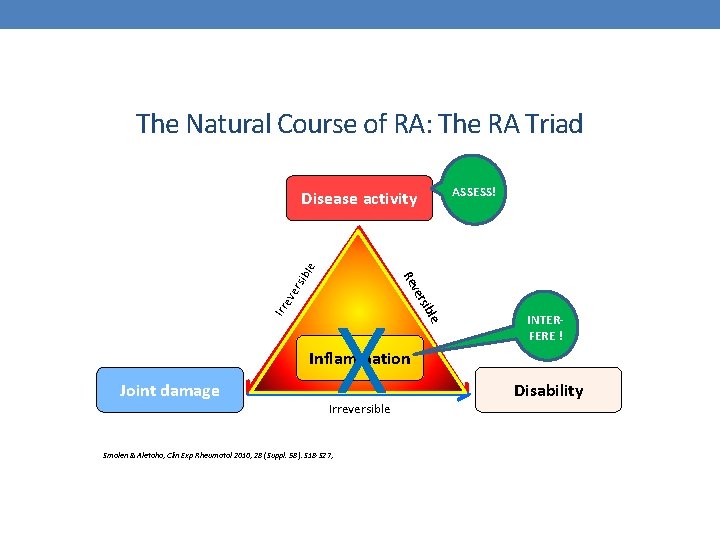
The Natural Course of RA: The RA Triad ASSESS! ers ev Irr le rsib X ve Re ibl e Disease activity INTERFERE ! Inflammation Joint damage Irreversible Smolen & Aletaha, Clin Exp Rheumatol 2010; 28 (Suppl. 58): S 18 -S 27; Disability
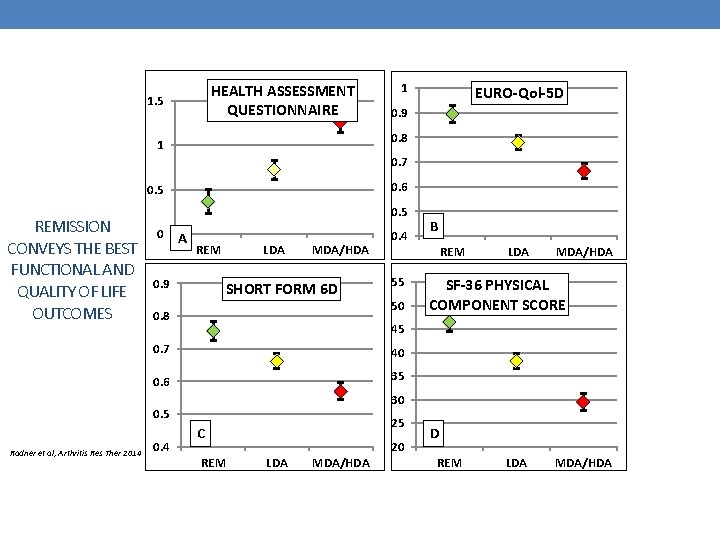
HEALTH ASSESSMENT QUESTIONNAIRE 1. 5 1 EURO-Qol-5 D 0. 9 0. 8 1 0. 7 0. 6 0. 5 REMISSION CONVEYS THE BEST FUNCTIONAL AND QUALITY OF LIFE OUTCOMES 0. 5 0 A REM 0. 9 LDA MDA/HDA SHORT FORM 6 D 0. 8 55 50 0. 7 40 0. 6 35 0. 4 B REM LDA MDA/HDA SF-36 PHYSICAL COMPONENT SCORE 45 30 0. 5 Radner et al, Arthritis Res Ther 2014 0. 4 25 C REM LDA MDA/HDA 20 D REM LDA MDA/HDA
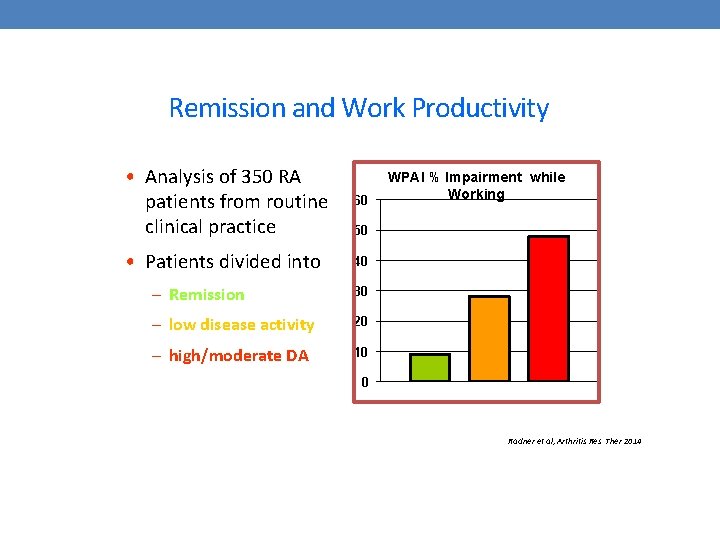
Remission and Work Productivity • Analysis of 350 RA patients from routine clinical practice 60 • Patients divided into 40 WPAI % Impairment while Working 50 – Remission 30 – low disease activity 20 – high/moderate DA 10 0 Radner et al, Arthritis Res Ther 2014
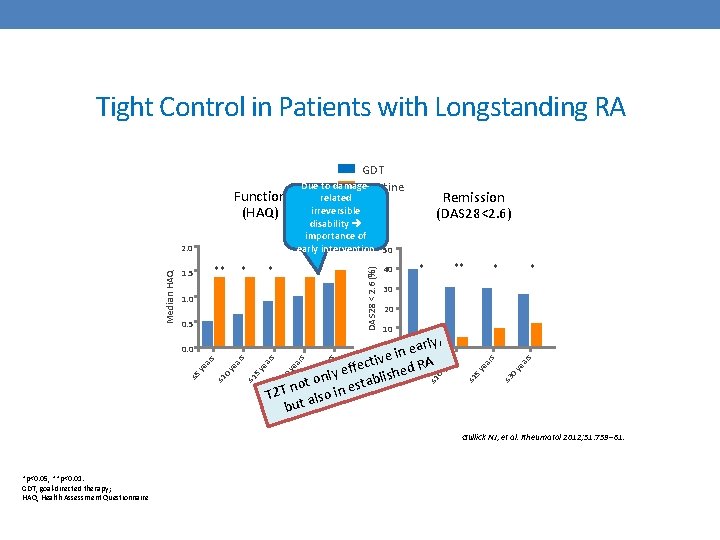
Tight Control in Patients with Longstanding RA Function (HAQ) * * Remission (DAS 28<2. 6) related irreversible disability importance of early intervention 50 DAS 28 <2. 6 (%) ** 1. 5 1. 0 0. 5 ** * 40 * * 30 20 10 , early n i ctive d RA e f f e ly lishe n b o a t t no es T 2 T also in but s ≤ 3 0 y e ar s 5 y e ar s ≤ 1 0 y e ar s ar ye ≤ 5 0 y e ≤ 3 ar ar s s 0 y e 5 y e ar s ≤ 1 ar s 0 y e ≤ 1 ar ye ≤ 5 s 0 0. 0 ≤ 2 Median HAQ 2. 0 GDT Due to damage. Routine Gullick NJ, et al. Rheumatol 2012; 51: 759– 61. *p<0. 05, **p<0. 01. GDT, goal-directed therapy; HAQ, Health Assessment Questionnaire

How it All Began rgets a t t n y atme t strateg e r t No men t a e r No t Saag KG, et al. Arthritis & Rheumatism 2008; 59: 762– 784
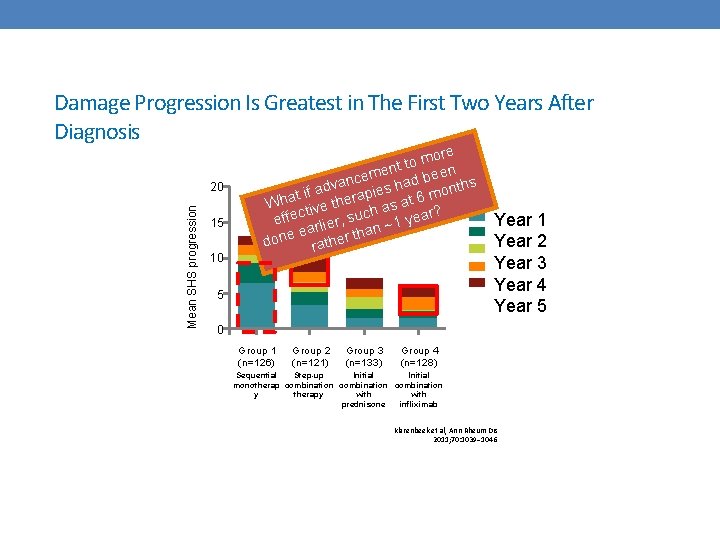
Damage Progression Is Greatest in The First Two Years After Diagnosis Mean SHS progression 20 15 10 more o t t men d been e c n a s a f adv rapies h 6 month i t a Wh tive the s at ? a h c c year effe rlier, su 1 ~ ea han done rather t 5 Year 1 Year 2 Year 3 Year 4 Year 5 0 Group 1 (n=126) Group 2 (n=121) Group 3 (n=133) Group 4 (n=128) Initial Sequential Step-up monotherap combination with y therapy prednisone infliximab Klarenbeek et al, Ann Rheum Dis 2011; 70: 1039– 1046
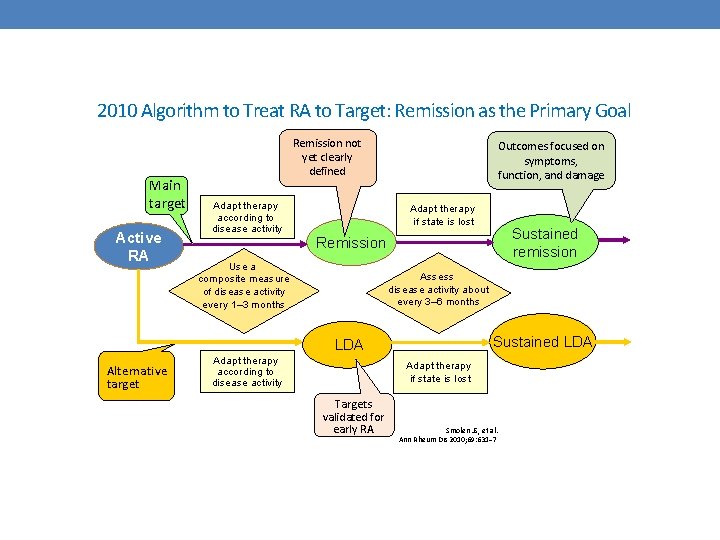
2010 Algorithm to Treat RA to Target: Remission as the Primary Goal Main target Active RA Remission not yet clearly defined Adapt therapy according to disease activity Outcomes focused on symptoms, function, and damage Adapt therapy if state is lost Use a composite measure of disease activity every 1– 3 months Assess disease activity about every 3– 6 months Sustained LDA Alternative target Sustained remission Remission Adapt therapy according to disease activity Adapt therapy if state is lost Targets validated for early RA Smolen JS, et al. Ann Rheum Dis 2010; 69: 631– 7

TREATMENT SHOULD AIM FOR “BEST CARE” WITH A SHARED DECISION Treatment should be goal-oriented and governed by a strategic treatment approach Primary target for treatment of RA: Remission or low disease activity Treatment for. Treatment RA in should be 2020 adapted if the Patients should be closely monitored using composite disease activity measures Smolen JS, et al. Ann Rheum Dis 2016; 75: 3– 15. treatment aim is not reached within 3 to 6 months (i. e. CDAI reduction >50% at 3 months, target at 6 months)

ACR-EULAR 2011 Definition of Remission For Clinical Trials Boolean SJC, TJC, Pt. GA (0– 10 scale), CRP (in mg/d. L) all ≤ 1 For Clinical Practice Index-based SDAI ≤ 3. 3 SDAI = 28 SJC + 28 TJC + EGA + Pt. GA + CRP (mg/d. L) Index-based CDAI ≤ 2. 8 CDAI = 28 SJC + 28 TJC + EGA + Pt. GA Boolean SJC, TJC, Pt. GA (0– 10 scale) all ≤ 1 DAS 28 should not be used to define remission ! Felson D et al. Arthritis Rheum, 2011; 63: 573– 586, and Ann Rheum Dis. 2011; 70: 404– 413 EGA = evaluator global disease activity Pt. GA = patient global disease activity
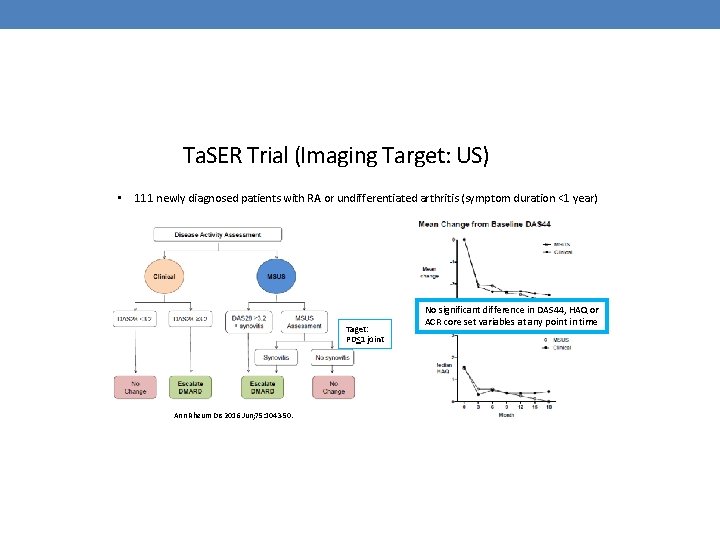
Ta. SER Trial (Imaging Target: US) • 111 newly diagnosed patients with RA or undifferentiated arthritis (symptom duration <1 year) Taget: PD<1 joint Ann Rheum Dis 2016 Jun; 75: 1043 -50. No significant difference in DAS 44, HAQ or ACR core set variables at any point in time
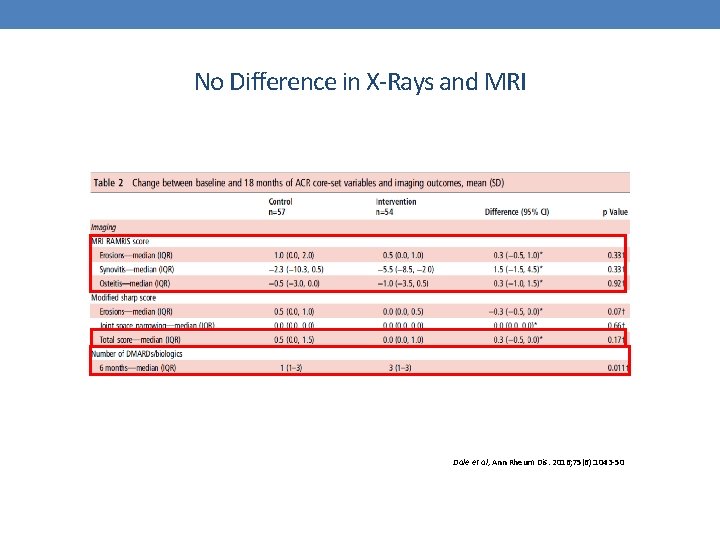
No Difference in X-Rays and MRI Dale et al, Ann Rheum Dis. 2016; 75(6): 1043 -50
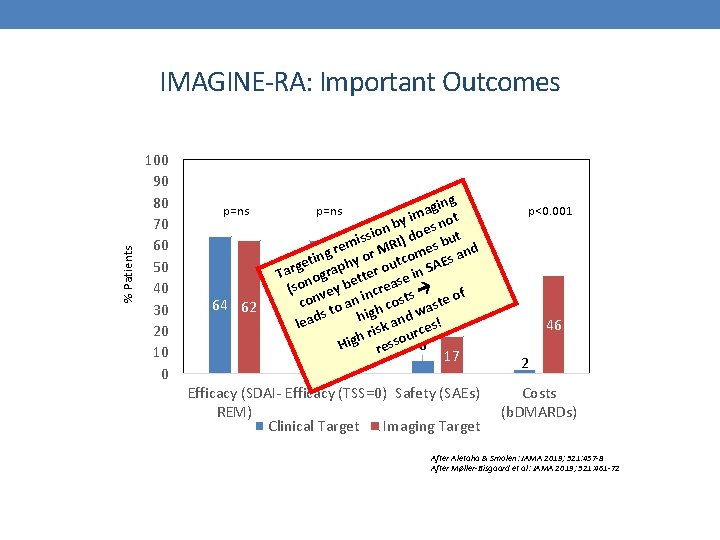
% Patients IMAGINE-RA: Important Outcomes 100 90 80 70 60 50 40 30 20 10 0 p=ns 64 62 ging a m by i es not n o issi RI) do s but m g re y or M come s and n i t h ut AE ge Tar ograp etter o se in S (son vey b increa s of n 66 co 62 an h cost aste o t s hig and w s! lead isk urce r h Hig resso 6 17 p=ns Efficacy (SDAI- Efficacy (TSS=0) Safety (SAEs) REM) Clinical Target Imaging Target p<0. 001 46 2 Costs (b. DMARDs) After Aletaha & Smolen: JAMA 2019; 321: 457 -8 After Møller-Bisgaard et al: JAMA 2019; 321: 461 -72
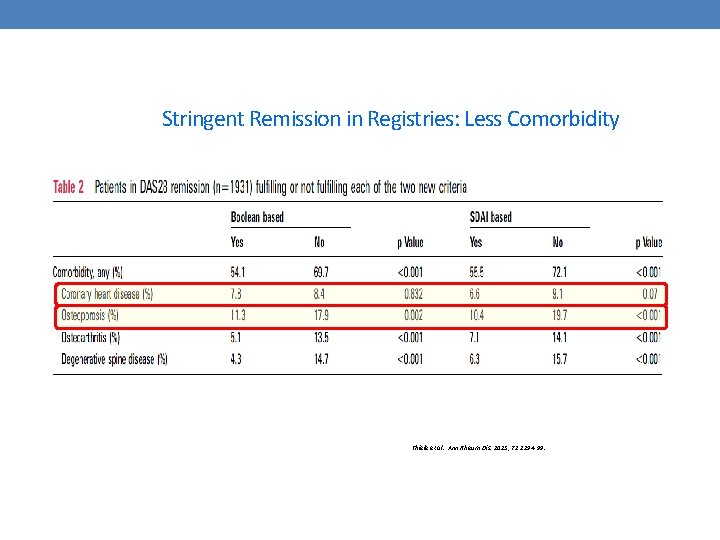
Stringent Remission in Registries: Less Comorbidity Thiele et al. Ann Rheum Dis. 2013; 72: 1194 -99.
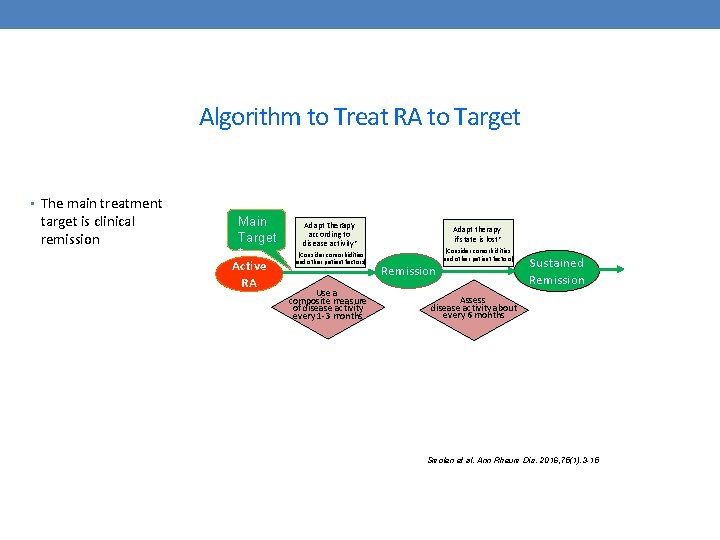
Algorithm to Treat RA to Target • The main treatment target is clinical remission Main Target * Active RA Adapt therapy according to disease activity* (Consider comorbidities and other patient factors) Use a composite measure of disease activity every 1 -3 months Adapt therapy if state is lost* (Consider comorbidities and other patient factors) Remission Sustained Remission Assess disease activity about every 6 months Smolen et al. Ann Rheum Dis. 2016; 75(1): 3 -15
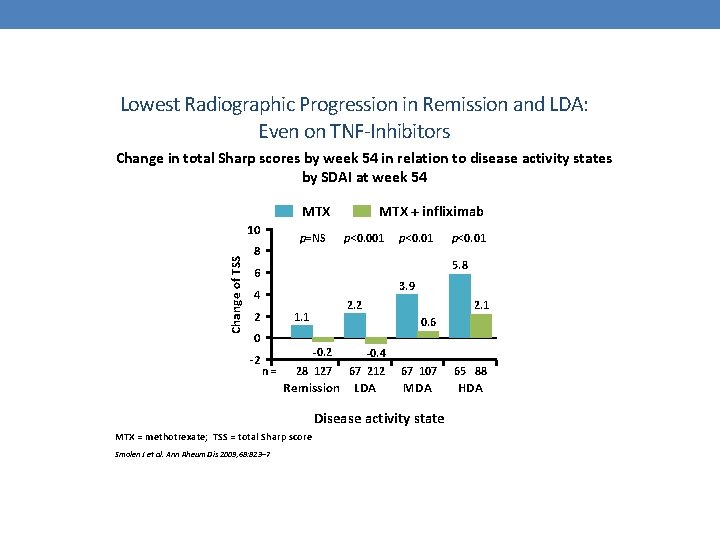
Lowest Radiographic Progression in Remission and LDA: Even on TNF-Inhibitors Change in total Sharp scores by week 54 in relation to disease activity states by SDAI at week 54 MTX Change of TSS 10 p=NS 8 MTX + infliximab p<0. 001 p<0. 01 5. 8 6 3. 9 4 2 2. 2 1. 1 0 -2 p<0. 01 0. 6 -0. 2 n= 2. 1 28 127 -0. 4 67 212 Remission LDA 67 107 65 88 MDA HDA Disease activity state MTX = methotrexate; TSS = total Sharp score Smolen J et al. Ann Rheum Dis 2009; 68: 823– 7

LDA Is The 2 nd Best State on A b. DMARD: Progression of m. TSS on Anti-TNFs Smolen et al, RMD Open 2020; 6: e 001096
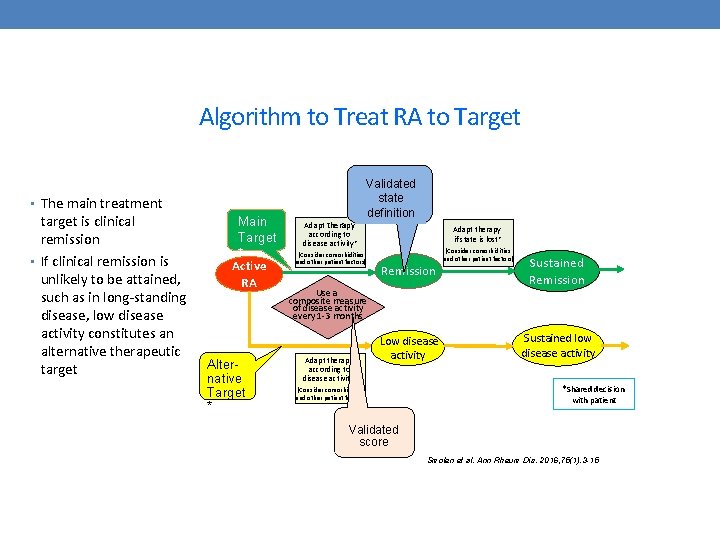
Algorithm to Treat RA to Target • The main treatment target is clinical remission • If clinical remission is unlikely to be attained, such as in long-standing disease, low disease activity constitutes an alternative therapeutic target Main Target * Active RA Alternative Target * Adapt therapy according to disease activity* Validated state definition (Consider comorbidities and other patient factors) Adapt therapy if state is lost* (Consider comorbidities and other patient factors) Remission Sustained Remission Low disease activity Sustained low disease activity Use a composite measure of disease activity every 1 -3 months Adapt therapy according to disease activity* (Consider comorbidities and other patient factors) *Shared decision with patient Validated score Smolen et al. Ann Rheum Dis. 2016; 75(1): 3 -15
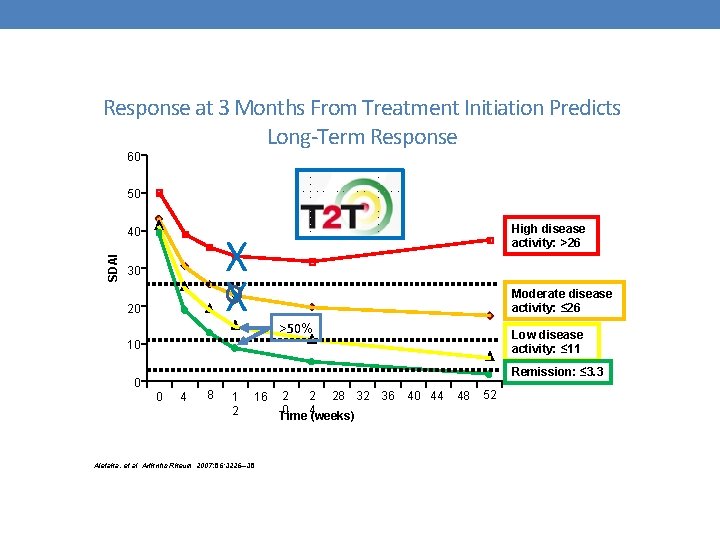
Response at 3 Months From Treatment Initiation Predicts Long-Term Response 60 50 SDAI 40 High disease activity: >26 X X 30 20 Moderate disease activity: ≤ 26 >50% Low disease activity: ≤ 11 10 Remission: ≤ 3. 3 0 0 4 8 1 2 Aletaha, et al. Arthritis Rheum 2007; 56: 3226– 35 16 2 2 28 32 36 0 4 Time (weeks) 40 44 48 52
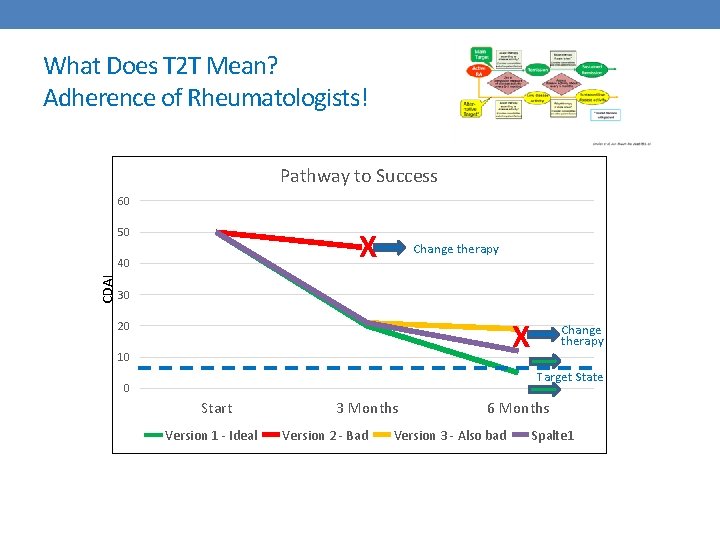
What Does T 2 T Mean? Adherence of Rheumatologists! Pathway to Success 60 50 X CDAI 40 Change therapy 30 X 20 10 Change therapy Target State 0 Start Version 1 - Ideal 3 Months Version 2 - Bad 6 Months Version 3 - Also bad Spalte 1
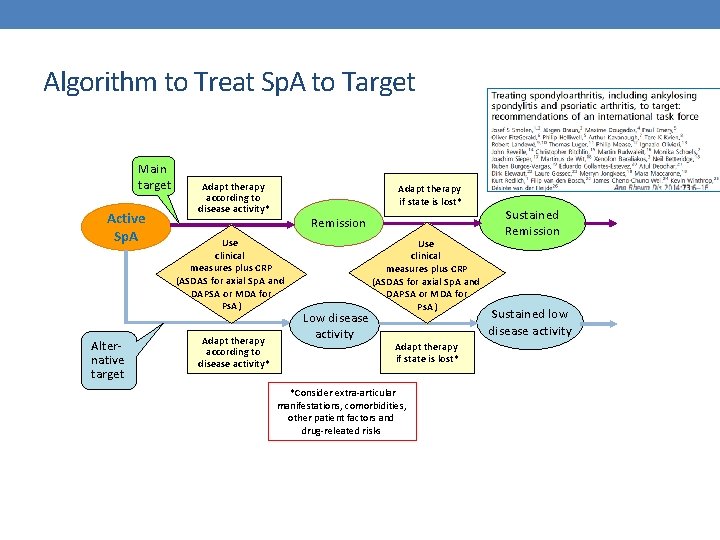
Algorithm to Treat Sp. A to Target Main target Active Sp. A Alternative target Adapt therapy according to disease activity* Adapt therapy if state is lost* Remission Use clinical measures plus CRP (ASDAS for axial Sp. A and DAPSA or MDA for Ps. A) Adapt therapy according to disease activity* Low disease activity Use clinical measures plus CRP (ASDAS for axial Sp. A and DAPSA or MDA for Ps. A) Adapt therapy if state is lost* *Consider extra-articular manifestations, comorbidities, other patient factors and drug-releated risks Sustained Remission Sustained low disease activity
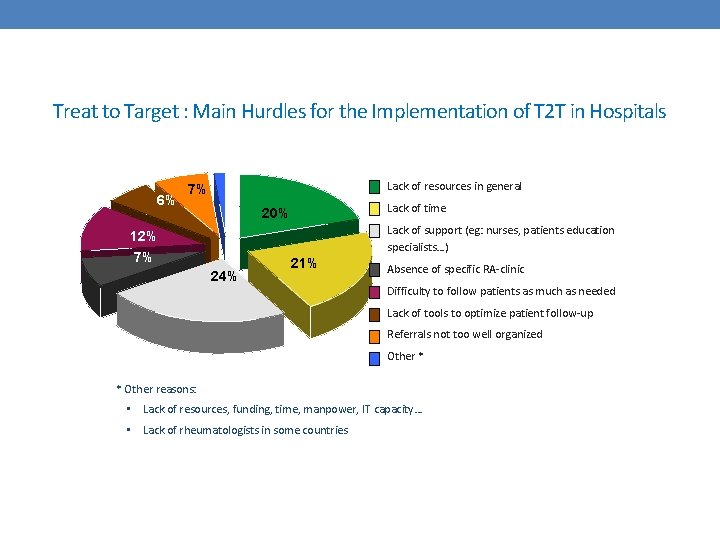
Treat to Target : Main Hurdles for the Implementation of T 2 T in Hospitals 6% Lack of resources in general 7% Lack of time 20% Lack of support (eg: nurses, patients education specialists…) 12% 7% 24% 21% Absence of specific RA-clinic Difficulty to follow patients as much as needed Lack of tools to optimize patient follow-up Referrals not too well organized Other * * Other reasons: • Lack of resources, funding, time, manpower, IT capacity… • Lack of rheumatologists in some countries

Company Confidential © 2019 Eli Lilly and Company Netherlands: Main Deviations Among 42% Who Did Not Use A T 2 T Strategy Despite Active Disease (DREAM-Study) • Not using composite measures of disease activity regularly to make treatment decisions • Not adjusting drug therapy every 3 months until target is reached • Not intensifying instead of intensifying • Taper/discontinuing instead of continuing • Determination of disease activity is often based on clinical judgment Vermeer M, et al. Arthritis Res Ther. 2012; 14: R 254
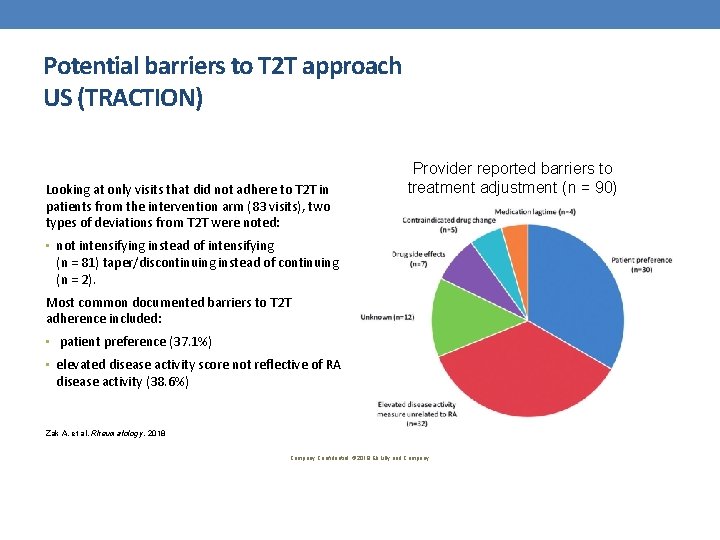
Potential barriers to T 2 T approach US (TRACTION) Looking at only visits that did not adhere to T 2 T in patients from the intervention arm (83 visits), two types of deviations from T 2 T were noted: Provider reported barriers to treatment adjustment (n = 90) • not intensifying instead of intensifying (n = 81) taper/discontinuing instead of continuing (n = 2). Most common documented barriers to T 2 T adherence included: • patient preference (37. 1%) • elevated disease activity score not reflective of RA disease activity (38. 6%) Zak A, et al. Rheumatology. 2018 Company Confidential © 2018 Eli Lilly and Company

Conclusion • Disease activity drives damage, disability and comorbidity in RA and Sp. A patients • Early interference with disease activity improves short- and long-term outcomes • Remission is the primary goal (in early disease), low disease activity a good alternative • The way to the goal is also a target: measure, measure adapt, adapt ! • To this end, it is important to assess disease activity from beginning to end • >50% improvement at 3 months and target achievement at 6 months is the strategic approach • Adapt therapy if this is not attained usually no difference in costs and we have many options today • Adherence to the T 2 T strategy will allow optimal clinical, structural and functional outcomes • Don‘t delay treatment start and do not leave patients with active disease on a therapy that is not working sufficiently!!

Acknowledgement • Daniel Aletaha • Farideh Alasti • Manuel Becede • Helga Radner • Paul Studenic

How to Measure Disease Activity in the Office Theodore Pincus, MD 55

Quantitative measurement for treat-to-target in rheumatoid arthritis: Indices of quantitative, protocol-driven, standard, scientific data to assess patient history with a self-report questionnaire and physical exam with a formal joint count Theodore Pincus, MD Professor of Medicine Rush University Medical Center tedpincus@gmail. com

Disclosure - Theodore Pincus, MD Dr. Pincus holds a copyright and trademark for MDHAQ, RAPID 3, and Clin. Dat, for which he receives royalties and license fees, all of which are used to support further development of quantitative measurement in clinical rheumatology care. No license fees are collected from clinicians and non-profit researchers, who may freely use paper copies of MDHAQ/RAPID 3 to monitor patient status in clinical care and research and/or participate in beta testing of electronic MDHAQ/RAPID 3 at this time.

Lord Kelvin When you can measure what you are speaking about, and express it in numbers, you know something about it; but when you cannot measure it… your knowledge is of a meager and unsatisfactory kind. - Lord Kelvin 1883, quoted - by Buchanan and Smythe J Rheumatol 9: 653, 1982

Advantages of measurement over narrative description • • My child has a fever – 101ºF or 106ºF? • • This wine is expensive – $60 or $6000? • The RA patient is better -- – DAS 28 4. 2 or 2. 4? – CDAI 16 or 5? – RAPID 3 10 or 4? 59

Meager and unsatisfactory knowledge concerning RA - 1984 concerning Arthritis Rheum 27: 1344 -1352, 1984 “patients with rheumatoid arthritis usually respond to a conservative program of nonsteroidal antiinflammatory drugs, rest, and physical therapy…” HE Paulus, HJ Williams, JR Ward, JC Reading MJ Egger, ML Coleman, CO Samuelson, Jr. RF Willkens, M Guttadauria, GS Alarcon SB Kaplan, EJ Mac. Laughlin, A Weinstein RL Wilder. MA Solsky, RF Meenan

RA without measurement - 1983 Clinicians may all too easily spend years writing “doing well” in the notes of a patient who has become progressively crippled before their eyes… – Verna Wright Smith T, et al. Br Med J 1983; 287: 569.

RA with Quantitative Measurement: Severe functional declines, work disability, and increased mortality in seventy-five rheumatoid arthritis patients studied over nine years T Pincus, LF Callahan, WG Sale, AL Brooks, LE Payne, WK Vaughn Arthritis Rheum 27: 864 -872, 1984
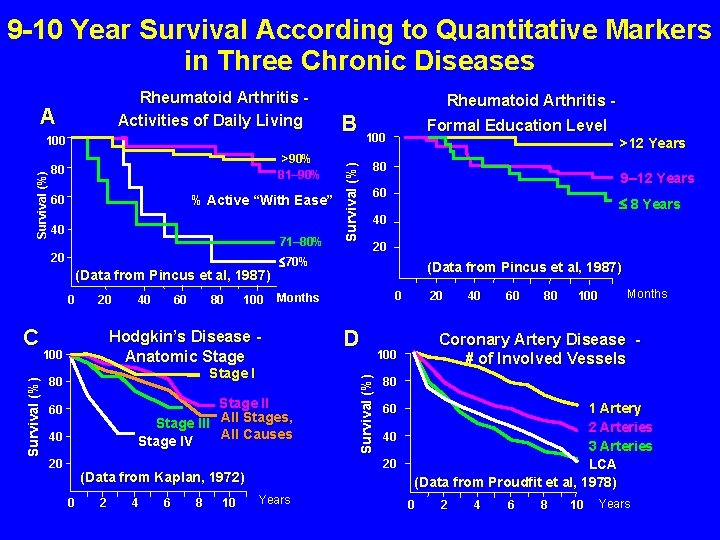
9 -10 Year Survival According to Quantitative Markers in Three Chronic Diseases >90% 81– 90% 80 % Active “With Ease” 60 40 71– 80% 20 (Data from Pincus et al, 1987) 0 Survival (%) C 20 40 60 80 100 Stage III All Stages, All Causes Stage IV 60 40 20 (Data from Kaplan, 1972) 0 2 4 6 8 10 80 Years >12 Years 9– 12 Years 60 8 Years 40 20 (Data from Pincus et al, 1987) 0 Months Stage I 80 Formal Education Level 100 70% Hodgkin’s Disease Anatomic Stage 100 B 20 40 60 80 Months 100 D Coronary Artery Disease 100 # of Involved Vessels Survival (%) 100 Rheumatoid Arthritis - Survival (%) Rheumatoid Arthritis A Activities of Daily Living 80 1 Artery 2 Arteries 40 3 Arteries 20 LCA (Data from Proudfit et al, 1978) 60 0 2 4 6 8 10 Years
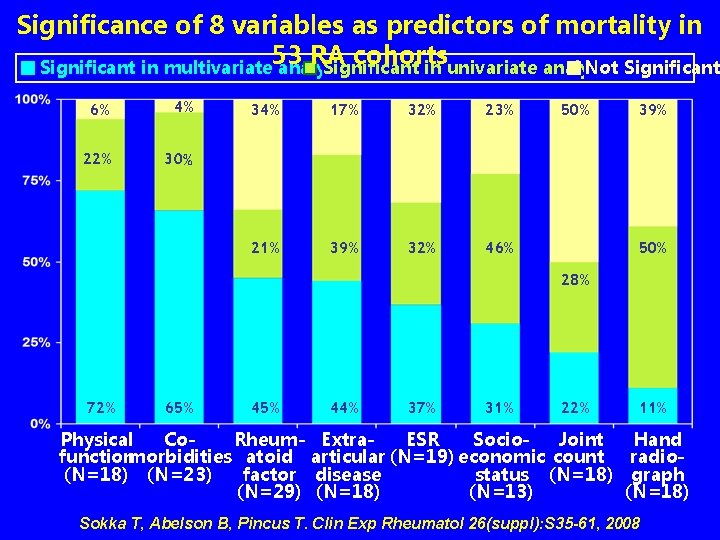
Significance of 8 variables as predictors of mortality in 53 RA cohorts Significant in multivariate analyses Significant in univariate analyses Not Significant 6% 4% 22% 30% 34% 17% 32% 23% 21% 39% 32% 46% 50% 39% 50% 28% 72% 65% 44% 37% 31% 22% 11% Physical Co. Rheum- Extra. ESR Socio. Joint Hand functionmorbidities atoid articular (N=19) economic count radio(N=18) (N=23) factor disease status (N=18) graph (N=29) (N=18) (N=13) (N=18) Sokka T, Abelson B, Pincus T. Clin Exp Rheumatol 26(suppl): S 35 -61, 2008

Treat-to-target in RA differs from hypertension or diabetes § No single ‘gold standard’ abnormal biomarker in all individual RA patients, such as BP or Hg. A 1 C § Lab tests are positive in only 50% (ESR, CRP) - 70% (rheumatoid factor, ACPA) § Therefore, quantitative RA measurement requires an index of 3 or 4 measures § Patient history and physical exam are more prominent in RA indices than biomarkers
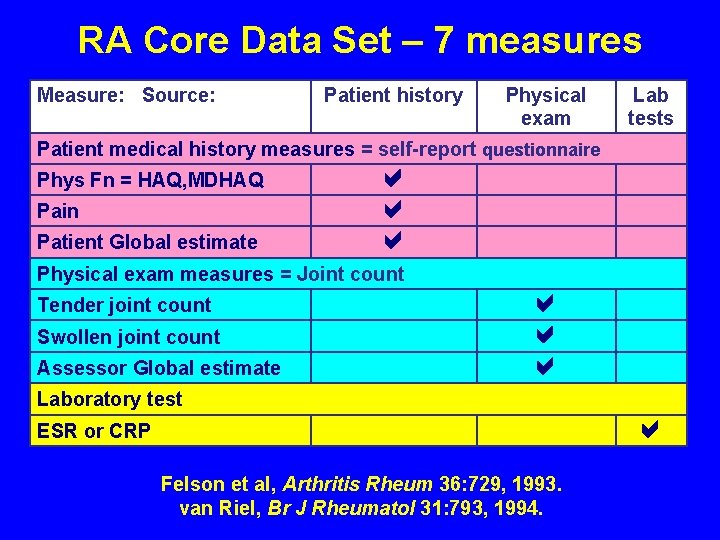
RA Core Data Set – 7 measures Measure: Source: Patient history Physical exam Lab tests Patient medical history measures = self-report questionnaire Phys Fn = HAQ, MDHAQ Pain Patient Global estimate Physical exam measures = Joint count Tender joint count Swollen joint count Assessor Global estimate Laboratory test ESR or CRP Felson et al, Arthritis Rheum 36: 729, 1993. van Riel, Br J Rheumatol 31: 793, 1994.

Clinical Decisions Survey Please indicate your opinion of the importance of each of 5 sources: 1) vital signs, 2) patient history, 3) physical examination, 4) laboratory tests, 5) ancillary studies, to provide 0 -20%, 21 -40%, 41 -60%, 61 -80%, or 81 -100% of information for diagnosis and management of: 1. hypertension 2. diabetes mellitus 3. rheumatoid arthritis 4. hypercholesterolemia 5. pulmonary fibrosis 6. ulcerative colitis 7. lymphoma 8. congestive heart failure Completed by 313 doctors, 154 rheumatologists and 159 non-rheumatologists Castrejón I, Mc. Collum L, Tanriover MD, Pincus T. Arthritis Care Res 2012; 64: 1250
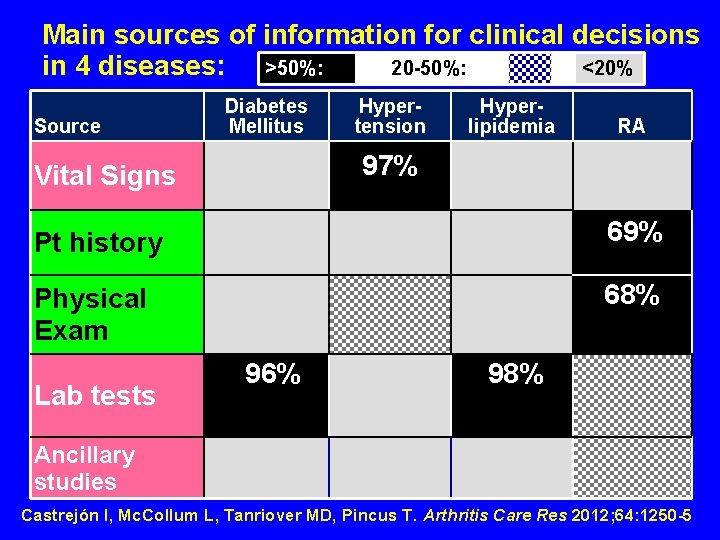
Main sources of information for clinical decisions in 4 diseases: >50%: 20 -50%: <20% Source Diabetes Mellitus Hypertension Hyperlipidemia 97% Vital Signs 69% Pt history 68% Physical Exam Lab tests RA 96% 98% Ancillary studies Castrejón I, Mc. Collum L, Tanriover MD, Pincus T. Arthritis Care Res 2012; 64: 1250 -5
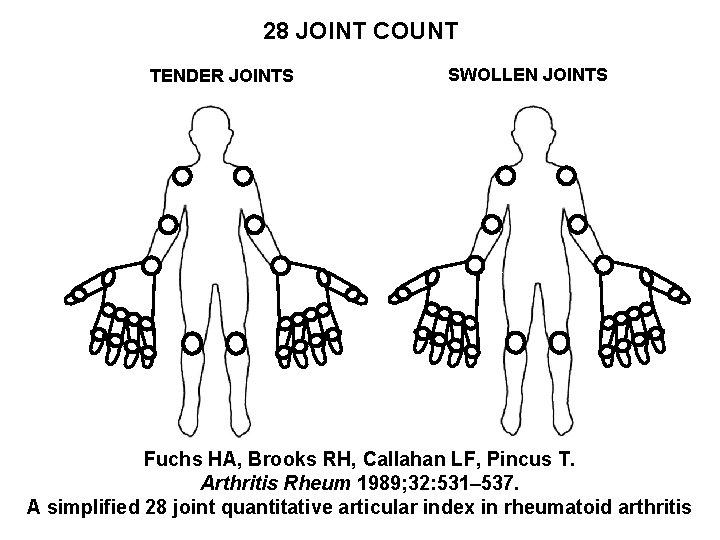
28 JOINT COUNT TENDER JOINTS SWOLLEN JOINTS Fuchs HA, Brooks RH, Callahan LF, Pincus T. Arthritis Rheum 1989; 32: 531– 537. A simplified 28 joint quantitative articular index in rheumatoid arthritis

MDHAQ (multi-dimensional health assessment questionnaire) 70 Pincus T, Swearingen C, Wolfe F. Arth Rheum. 42: 2220 -30, 1999. Pincus T, Sokka T, Kautiainen H. J Rheumatol. 32: 1432 -39, 2005.

Relative efficiencies of 7 RA Core Data Set to distinguish active from control treatments in 9 clinical trials Measure: Patient history = self-report questionnaire PATGL Phys Function Swollen JC Relative efficiency Active vs control in 9 clinical trials (rank) ESR or CRP Tender JC Lab tests DOCGL Pain Physical exam = Joint count Pincus, et al Clin Exp Rheumatol 32: S 47 -S 54 2014. 1 2 3 4 5 6 7
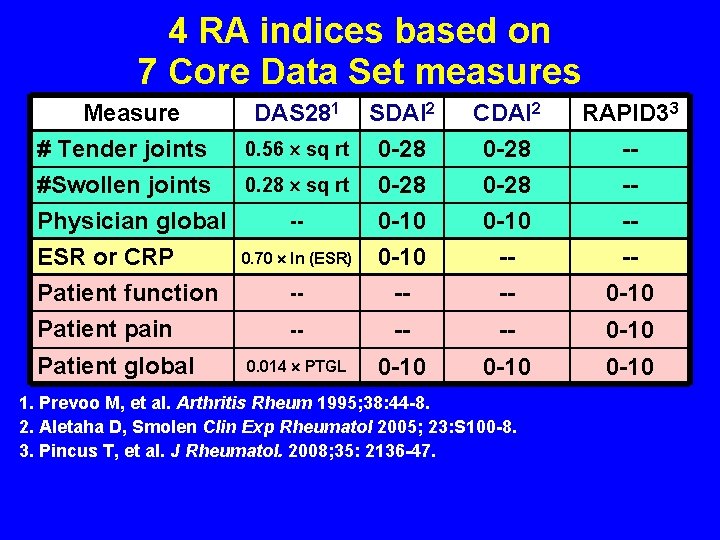
4 RA indices based on 7 Core Data Set measures Measure DAS 281 SDAI 2 0. 56 sq rt 0 -28 # Tender joints #Swollen joints 0. 28 sq rt 0 -28 -Physician global 0 -10 0. 70 ln (ESR) 0 -10 ESR or CRP -Patient function -Patient pain Patient global CDAI 2 0 -28 0 -10 RAPID 33 ---- -0 -10 -- -- -- 0 -10 0. 014 PTGL 0 -10 1. Prevoo M, et al. Arthritis Rheum 1995; 38: 44 -8. 2. Aletaha D, Smolen Clin Exp Rheumatol 2005; 23: S 100 -8. 3. Pincus T, et al. J Rheumatol. 2008; 35: 2136 -47.
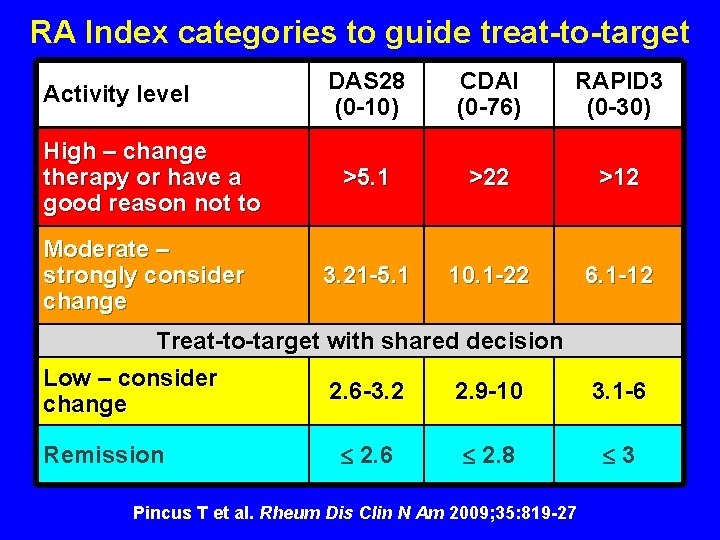
RA Index categories to guide treat-to-target Activity level High – change therapy or have a good reason not to Moderate – strongly consider change DAS 28 (0 -10) CDAI (0 -76) RAPID 3 (0 -30) >5. 1 >22 >12 3. 21 -5. 1 10. 1 -22 6. 1 -12 Treat-to-target with shared decision Low – consider change Remission 2. 6 -3. 2 2. 9 -10 3. 1 -6 2. 8 3 Pincus T et al. Rheum Dis Clin N Am 2009; 35: 819 -27

DAS 28 (Disease Activity Score 28) (0. 56 sq rt TJC) + (0. 23 sq rt SJC) +[0. 70 ln (ESR)] + (0. 014 PATGL) = 0 -10 Based on discriminant analysis of clinician’s change of DMARD Advantages § Traditional - most widely used index in clinical trials § Most specific for RA § Excellent website for scoring Limitations § Need computer to score - albeit excellent website § Need laboratory test – not available or not informative § TJC weighted 2 X vs SJC – may not be ideal in index of inflammation § May be elevated in 2° OA, fibromyalgia, comorbidities

CDAI (Clinical Disease Activity Index) TJC (0 -28) + SJC (0 -28) + DOCGL (0 -10) +PATGL (0 -10) = 0 -76 Advantages § Easily calculated § No lab test needed § TJC and SJC specific to RA § TJC and SJC weighted equally Limitations: § TJC and SJC weighted 2. 8 X DOCGL & PATGL § May be elevated in 2° OA, fibromyalgia, comorbidities

RAPID 3 (Routine Assessment of Patient Index Data) Function (0 -10) + Pain (0 -10)+ Patient global (0 -10)=0 -30 Advantages § Patient does 98% of the work § Scoring in 5 secs vs 90 secs formal joint count § MDHAQ saves 2 -3 minutes for doctor at most visits § Data available to doctor before encounter § Does not require same examiner at each assessment § Informative in all rheumatic diseases studied Limitations: § Mild practice redesign - educate staff § Some patients complain – educate patients § Need for translation § Cultural differences in report of pain, fatigue, etc. § Elevated in secondary OA, fibromyalgia, comorbidities

MDHAQ multi-dimensional health assessment questionnaire Physical function (0 -10) RAPID 3 (0 -30) Pain VAS (0 -10) Patient global VAS (0 -10) 77

Let the patient do most of the work! Most patients are more accurate about many details of their own medical history than health professionals

MDHAQ helps the patient prepare for a better visit

Doctor reviews information with the patient, and interprets data for clinical decisions
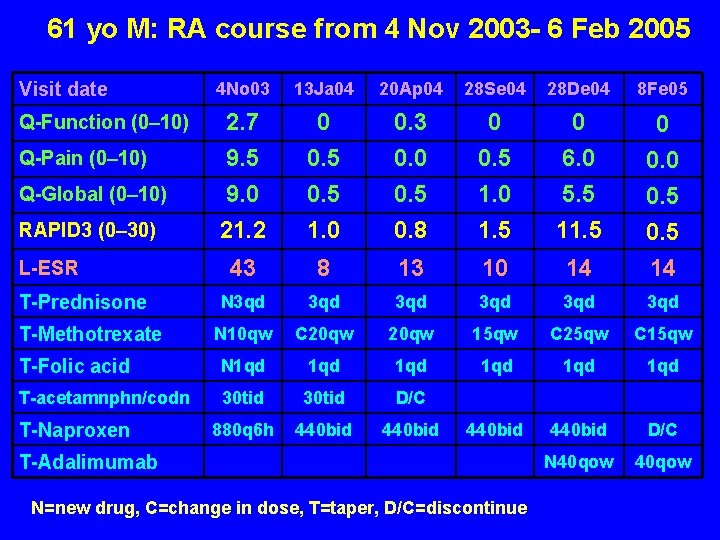
61 yo M: RA course from 4 Nov 2003 - 6 Feb 2005 Visit date 4 No 03 13 Ja 04 20 Ap 04 28 Se 04 28 De 04 8 Fe 05 Q-Global (0– 10) 2. 7 9. 5 9. 0 0 0. 5 0. 3 0. 0 0. 5 1. 0 0 6. 0 5. 5 0 0. 5 RAPID 3 (0– 30) 21. 2 1. 0 0. 8 1. 5 11. 5 43 8 13 10 14 0. 5 14 N 3 qd 3 qd 3 qd N 10 qw C 20 qw 15 qw C 25 qw C 15 qw T-Folic acid N 1 qd 1 qd 1 qd T-acetamnphn/codn 30 tid D/C 880 q 6 h 440 bid D/C N 40 qow Q-Function (0– 10) Q-Pain (0– 10) L-ESR T-Prednisone T-Methotrexate T-Naproxen T-Adalimumab N=new drug, C=change in dose, T=taper, D/C=discontinue
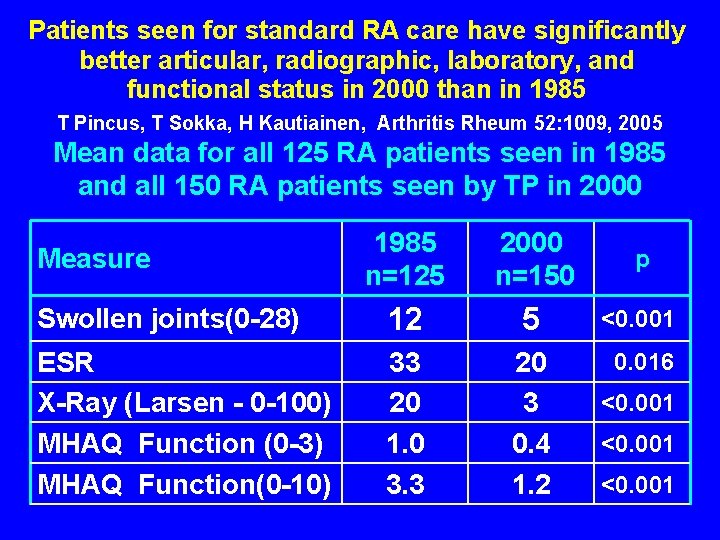
Patients seen for standard RA care have significantly better articular, radiographic, laboratory, and functional status in 2000 than in 1985 T Pincus, T Sokka, H Kautiainen, Arthritis Rheum 52: 1009, 2005 Mean data for all 125 RA patients seen in 1985 and all 150 RA patients seen by TP in 2000 1985 n=125 2000 n=150 p Swollen joints(0 -28) 12 5 <0. 001 ESR X-Ray (Larsen - 0 -100) MHAQ Function (0 -3) MHAQ Function(0 -10) 33 20 1. 0 3. 3 20 3 0. 4 1. 2 0. 016 Measure <0. 001

Electronic MDHAQ Report to the Rheumatologist and other Health Professionals Generated after Patient Completes Electronic MDHAQ

A caveat about RA Indices § RA indices indicating moderate/high “activity” may indicate OA, fibromyalgia, or other comorbidity, with or without inflammation. § Most information in the literature concerning DAS 28, CDAI, RAPID 3 is derived from clinical trials, in patients are selected for high disease activity, with SJC≥ 6, TJC≥ 6 and elevated ESR or CRP. § High index scores in clinical trials reflect primarily high inflammatory activity. § Only 5– 20% of patients meet inclusion criteria for RA clinical trials. § In routine rheumatology care, many more RA patients have comorbid OA, and/or fibromyalgia and/or other comorbidities, which may elevate RA index scores even if SJC=0 and ESR=20. § This matter is no different from a newly elevated ESR in an RA patient, which may indicate an RA flare, but infection or neoplasm must be excluded initially. § The treating physician must always interpret any quantitative measure, whether a laboratory test or an RA index.
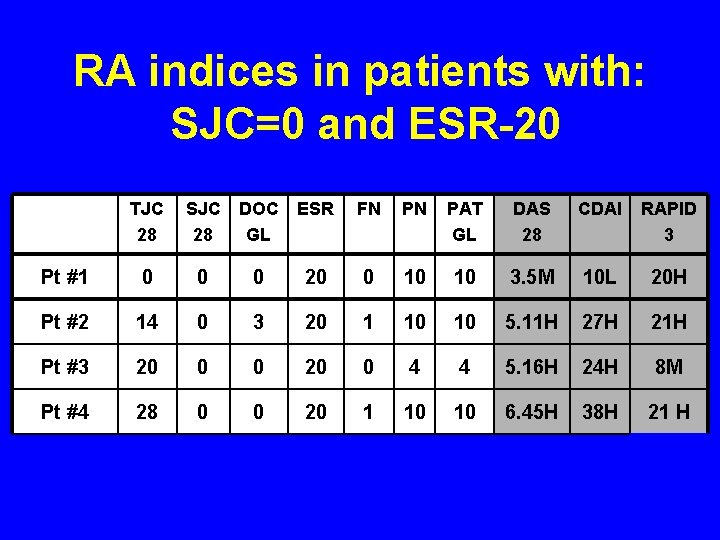
RA indices in patients with: SJC=0 and ESR-20 TJC 28 SJC 28 DOC GL ESR FN PN PAT GL DAS 28 CDAI RAPID 3 Pt #1 0 0 0 20 0 10 10 3. 5 M 10 L 20 H Pt #2 14 0 3 20 1 10 10 5. 11 H 27 H 21 H Pt #3 20 0 0 20 0 4 4 5. 16 H 24 H 8 M Pt #4 28 0 0 20 1 10 10 6. 45 H 38 H 21 H
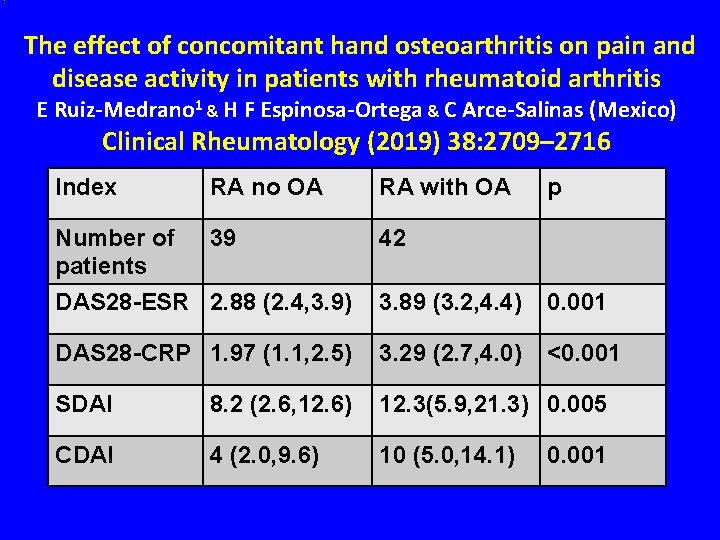
The effect of concomitant hand osteoarthritis on pain and disease activity in patients with rheumatoid arthritis E Ruiz-Medrano 1 & H F Espinosa-Ortega & C Arce-Salinas (Mexico) Clinical Rheumatology (2019) 38: 2709– 2716 Index RA no OA RA with OA p Number of 39 patients DAS 28 -ESR 2. 88 (2. 4, 3. 9) 42 3. 89 (3. 2, 4. 4) 0. 001 DAS 28 -CRP 1. 97 (1. 1, 2. 5) 3. 29 (2. 7, 4. 0) <0. 001 SDAI 8. 2 (2. 6, 12. 6) 12. 3(5. 9, 21. 3) 0. 005 CDAI 4 (2. 0, 9. 6) 10 (5. 0, 14. 1) 0. 001
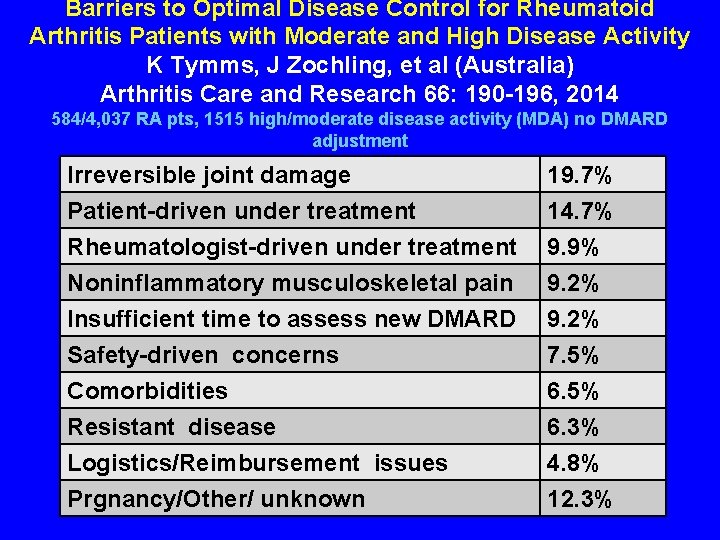
Barriers to Optimal Disease Control for Rheumatoid Arthritis Patients with Moderate and High Disease Activity K Tymms, J Zochling, et al (Australia) Arthritis Care and Research 66: 190 -196, 2014 584/4, 037 RA pts, 1515 high/moderate disease activity (MDA) no DMARD adjustment Irreversible joint damage Patient-driven under treatment Rheumatologist-driven under treatment Noninflammatory musculoskeletal pain 19. 7% 14. 7% 9. 9% 9. 2% Insufficient time to assess new DMARD Safety-driven concerns Comorbidities Resistant disease Logistics/Reimbursement issues Prgnancy/Other/ unknown 9. 2% 7. 5% 6. 3% 4. 8% 12. 3%

FAST 3 – Fibromyalgia assessment screening tool 60 -symptom checklist (0 -60) Physical function (0 -10) RAPID 3 (0 -30) Pain VAS (0 -10) Fatigue VAS (0 -10) FAST 3 -P or F (0 -3) or 4=(0 -4) Self-report painful joint count (0 -54) Pain or Fatigue ≥ 6 (0 -1) Joint count ≥ 16 (0 -1) #Symptoms ≥ 16 (0 -1) Patient global VAS (0 -10) FM positive if FAST 3 -P ≥ 2/3 8 or FAST 4 ≥ 3/4 8 Schmukler et al, Fibromyalgia Assessment Screening Tools (FAST) Based on Only Multidimensional Health Assessment Questionnaire (MDHAQ) Scores as Clues to Fibromyalgia. ACR Open Rheumatol. 2019; 1(8): 516 -25.
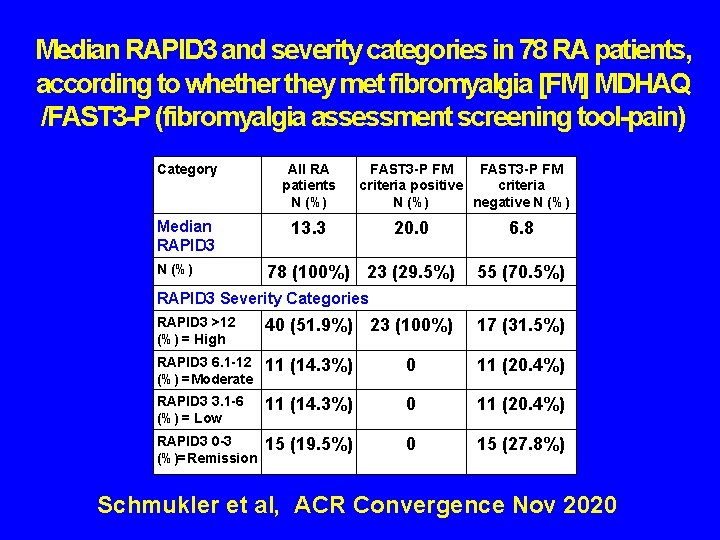
Median RAPID 3 and severity categories in 78 RA patients, according to whether they met fibromyalgia [FM] MDHAQ /FAST 3 -P (fibromyalgia assessment screening tool-pain) Category All RA patients N (%) Median RAPID 3 13. 3 N (%) FAST 3 -P FM criteria positive criteria N (%) negative N (%) 20. 0 78 (100%) 23 (29. 5%) 6. 8 55 (70. 5%) RAPID 3 Severity Categories RAPID 3 >12 (%) = High 40 (51. 9%) 23 (100%) 17 (31. 5%) RAPID 3 6. 1 -12 (%) =Moderate 11 (14. 3%) 0 11 (20. 4%) RAPID 3 3. 1 -6 (%) = Low 11 (14. 3%) 0 11 (20. 4%) RAPID 3 0 -3 (%)=Remission 15 (19. 5%) 0 15 (27. 8%) Schmukler et al, ACR Convergence Nov 2020

3 indices on 2 -page MDHAQ for routine care RAPID 3 (0 -30)= FN (0 -10) + PN (010) + PATGL (0 -10) FAST 3 -P ≥ 2/3=FM PN ≥ 6=1, RADAI ≥ 16=1, Sx. List ≥ 16=1 MDHAQ-Dep: Yes on either item = positive depression screen 9 0

Take home messages · Quantitative measures are keys to advance medical care · History and physical exam are prominent in diagnosis and management of RA – no “gold standard” biomarker · An index of multiple measures is needed to assess RA · Any valid index chosen by treating physician may be used · Treat-to-target is designed to control inflammation by identifying patients whose index scores indicate high/ moderate activity and escalating therapy in RA toward remission/low activity, and shared clinical decisions. · High index scores may indicate comorbidities, OA, FM, rather than, or in addition to high inflammatory activity. · As in all clinical medicine, quantitative data, whether lab tests or RA indices, require interpretation by a caring and knowledgeable physician.

Special Thanks To… Rheumatologists Frederick Wolfe Tuulikki Sokka Yusuf Yazici Martin Bergman Isabel Castrejon Kathryn Gibson Michael Stein Howard Fuchs Cecilia Chung Joe Huston Joel Block Juan Schmukler Research Associates Leigh Callahan Raye Brooks Christopher Swearingen Melissa Gibson Ben Abelson Annie Huang Benjamin Richardson Biostatisticians Hannu Kautiainen Gary Koch William Vaughn Ingrid Amara George Reed William Brown Dan Bloch Hal Morgenstern Sponsors Arthritis Foundation Abbvie Amgen Bristol-Meyers-Squibb Centocor Genentech Jack C Massey Foundation Novartis Pfizer UCB Celgene

Thank you very much for your attention Always available for critique, questions, comments tedpincus@gmail. com 615 -479 -5303 Questions?

94 2: 453: 00

Moving from Strategy to Implementation: What is a PDSA? 95

Why Bother Using PDSAs? � Test hunches quickly (by getting & studying results) � Multiple hunches can be tested simultaneously � Identify problems while they have minimal impact � Get buy-in as you go based on proof of success � Always have the end in mind: What Am I 96 Trying to Accomplish AND How Will I Ensure It Will Be Sustained? Adapted from © 2001 Institute for Healthcare Improvement
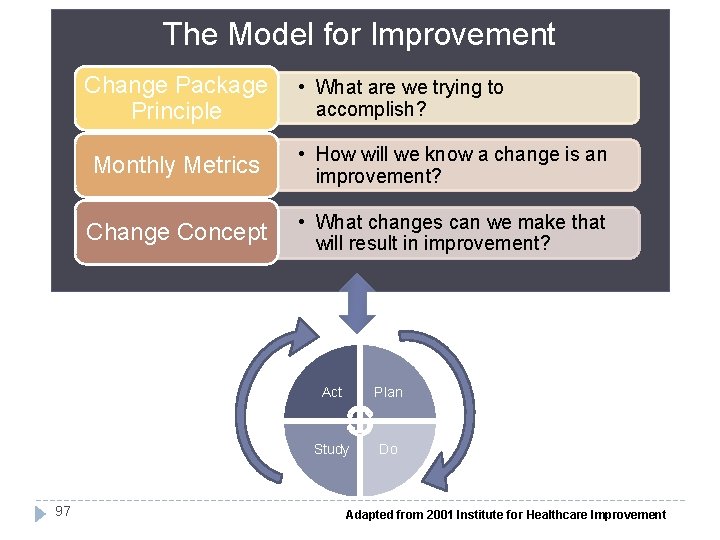
The Model for Improvement Change Package Principle 97 • What are we trying to accomplish? Monthly Metrics • How will we know a change is an improvement? Change Concept • What changes can we make that will result in improvement? Act Plan Study Do Adapted from 2001 Institute for Healthcare Improvement
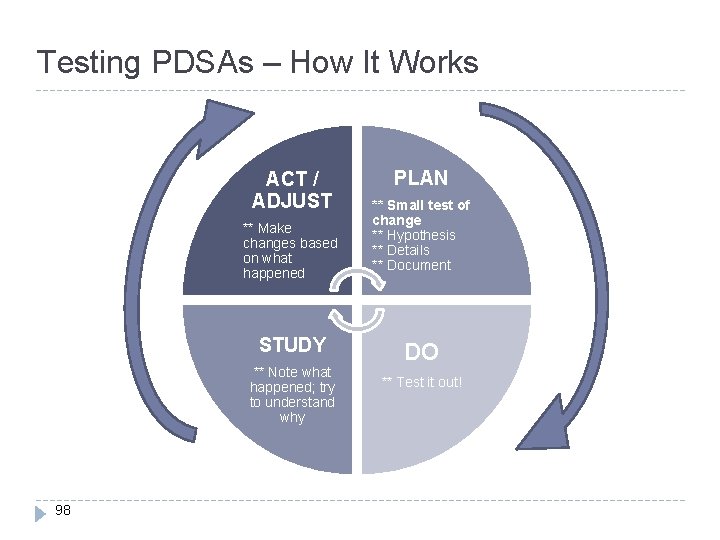
Testing PDSAs – How It Works ACT / ADJUST ** Make changes based on what happened STUDY ** Note what happened; try to understand why 98 PLAN ** Small test of change ** Hypothesis ** Details ** Document DO ** Test it out!
Moving to an Example: Start with a Principle 99

Apply the Model for Improvement The Model for Improvement Change Packag e Principl e Monthly Metrics Change Concept 10 0 • What are we trying to accomplish? • The treatment of RA is based on a process of shared decision making (SDM) between patient and rheumatologist. • How will we know a change is an improvement? • SDM is indicated in the patient chart • What changes can we make that will result in improvement? • Patient and provider take active steps to participate in the process of treatment decision making (active and inclusive role for patient)

Developing a PDSA: First Cycle PDSA Title: Goal Sheet PLAN OBJECTIVE: What is the objective for this cycle? Using a goal sheet before the appointment starts will help the patient reflect on her goals for treatment and will open a clearer and directed discussion with the doctor. Additionally, it will help the patient feel more directly engaged in her care and planning. PREDICTION: What do you predict will happen? The patient will have some questions about the goals on the form and the doctor will have a chance to better explain Treatto-Target and her role in her own treatment. What is the plan for the cycle? What are the steps to execute the cycle, including data collection (who, what, where, when)? Our Core Team will adapt the Goal Sheet provided as a sample by the LC Project Team. Patient will be asked to fill out goal sheet in waiting room next week. We will see if the information gathered was helpful, if the sheet was clear and easy to use, and we will track how long it took to fill out and then discuss (how much time it added to the appointment). 10 1
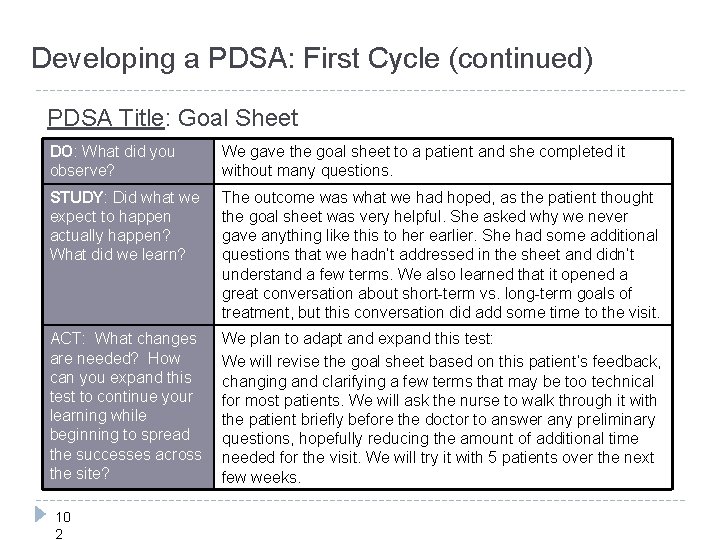
Developing a PDSA: First Cycle (continued) PDSA Title: Goal Sheet DO: What did you observe? We gave the goal sheet to a patient and she completed it without many questions. STUDY: Did what we expect to happen actually happen? What did we learn? The outcome was what we had hoped, as the patient thought the goal sheet was very helpful. She asked why we never gave anything like this to her earlier. She had some additional questions that we hadn’t addressed in the sheet and didn’t understand a few terms. We also learned that it opened a great conversation about short-term vs. long-term goals of treatment, but this conversation did add some time to the visit. ACT: What changes are needed? How can you expand this test to continue your learning while beginning to spread the successes across the site? We plan to adapt and expand this test: We will revise the goal sheet based on this patient’s feedback, changing and clarifying a few terms that may be too technical for most patients. We will ask the nurse to walk through it with the patient briefly before the doctor to answer any preliminary questions, hopefully reducing the amount of additional time needed for the visit. We will try it with 5 patients over the next few weeks. 10 2

One Small Test of Change… So What? � First PDSA is just the first step � Keep it small so that it can be tested quickly, but… � Small test does not equal small change � Goal is to apply learnings and continue to make tests bigger and broader � Pull in more people as you go – moving toward implementation as you continue to learn and refine tools/develop processes 10 3

Small Test of Change PDSA CHECKLIST � Is actual change -- making something different � Can be accomplished quickly � No over-planning (Plan only as much as you can do) � Consensus not required � Adapt known results and tools � Base on learning from prior cycle 10

Wrap-Up and Homework � Key points from Session 1 � TTT � DAMs � Learning Collaborative ethos � PDSA planning � What we expect from you before next week � Team meeting and huddle schedule � PDSA ideas and plans � Log into the Sharepoint 105
- Slides: 105