Destruction of Organophosphorus Nerve Agent Analogues by Activation

Destruction of Organophosphorus Nerve Agent Analogues by Activation of O 2 under Aqueous Room Temperature and Atmospheric Pressure Conditions Christina Noradoun, Ryan Hutcheson, Edmund Wong, I. Francis Cheng* University of Idaho, Department of Chemistry, Moscow, ID 83844 -2343 ifcheng@uidaho. edu; 208 -885 -6387 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 1

Overall Goal The destruction or neutralization of xenobiotics. Inexpensive & Safe Processes. • Room Temperature and Pressure Conditions (RTP) • Common Reagents – Long Term Storage • No Specialized Catalysts Yang, Y. -C. , Chemical Detoxification of Chemical Nerve Agent VX, Accounts of Chemical Research, 1999, 32, 109 -115. 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 2

Oxidative Pathways -Most attractive oxidant - O 2 from air -Cx. Hy. Xz + O 2 = CO 2 + H 2 O + HX (unbalanced) G<0 -Room temperature oxidations by air are kinetically slow -enzymatic or enzyme-mimics -Partially reduced O 2 (Reactive Oxygen Species) 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 3

Reactive Oxygen Forms • O=O • b. o. = 2 120 kcal/mol +e- • O-O • b. o. = 1. 5 80 kcal/mol G +4 e+4 H+ +2 e+2 H+ HO-OH b. o. = 1 50 kcal/mol 2 H 2 O 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 4
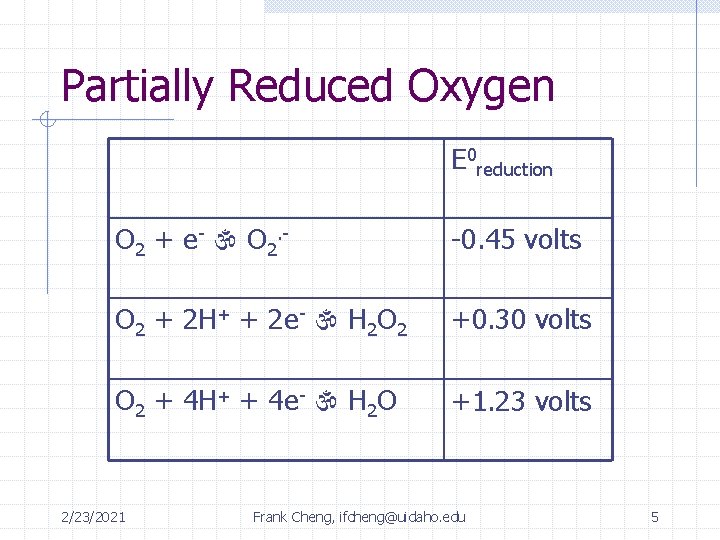
Partially Reduced Oxygen E 0 reduction O 2 + e- O 2. - -0. 45 volts O 2 + 2 H+ + 2 e- H 2 O 2 +0. 30 volts O 2 + 4 H+ + 4 e- H 2 O +1. 23 volts 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 5

That Last Step: The Fenton Reaction -Reduction of HO-OH H 2 O 2 + Fe. II = Fe. III + HO- + HO. + e- = HO- Eo = 1. 8 volts -HO. reacts with organics with diffusion limited kinetics -Possible Roles for O 2. 2 O 2. - + 2 H+ H 2 O 2 + O 2. - III II O 2 + Fe 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 6
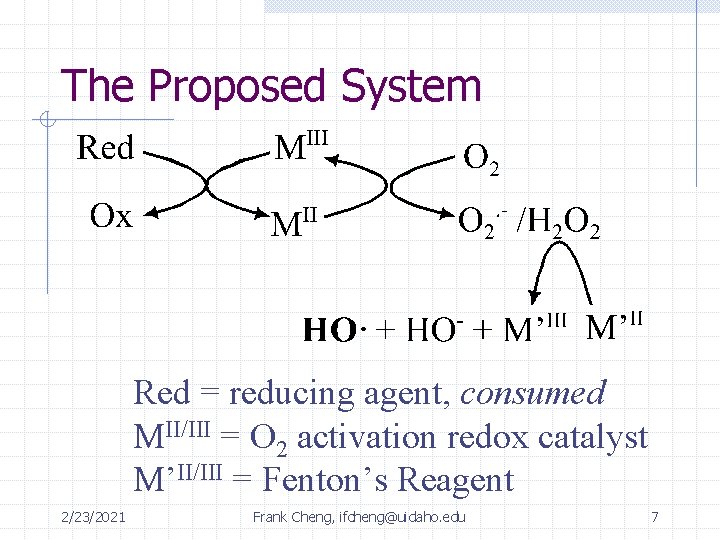
The Proposed System Red = reducing agent, consumed MII/III = O 2 activation redox catalyst M’II/III = Fenton’s Reagent 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 7

Putting the System Together – The Reducing Agent Considerations -Costs -Kinetics -Environmental Ered 0 Zero-valent metals Fe 2+ + 2 e-0. 44 volts Ascorbate dehydroxyascorbate + 2 e 0. 154 volts (p. H 4) 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 8

Putting the System Together – Oxygen Activation Agents RTP O 2 activation Biological Systems: -cytochrome p 450…. Chemical Systems: -non-heme iron complexes (cost? ) Que, L. JACS 2003, 125, 2113 -2128 -Fe. IIEDTA oxidation by O 2 Van Eldik, R. Inorg. Chem, 1997, 36, 4115 -4120 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 9

Putting the System Together – Fenton’s Reagent H 2 O 2 + e- HO- + HO Ered 0 = 0. 26(p. H 6) to 0. 08(p. H 3) V Fe. IIIEDTA + e- Fe. IIEDTAEred 0 = 0. 1 V • Fe. II/IIIEDTA is a classic Fenton’s Reagent Used in DNA footprinting studies • Can Fe. II/IIIEDTA do both O 2 activation and the Fenton reaction? 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 10

Summary of Van Eldik Study Fe. IIEDTAH(H 2 O) + O 2 Fe. IIEDTAH(O 2) + H 2 O Fe. IIEDTAH(O 2) Fe. IIIEDTAH(O 2 -) + Fe. IIEDTAH(H 2 O) Fe. IIIEDTAH(O 22 -)Fe. IIIEDTAH + H 2 O + 2 H+ 2 Fe. IIIEDTAH(H 2 O) + H 2 O 2 2 Fe. IIEDTAH(H 2 O) + H 2 O 2 2 Fe. IIIEDTAH(H 2 O) + H 2 O *Proposes H 2 O 2 as intermediate *Saw no evidence of H 2 O 2 Van Eldik, R. Inorg. Chem, 1997, 36, 4115 -4120 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 11
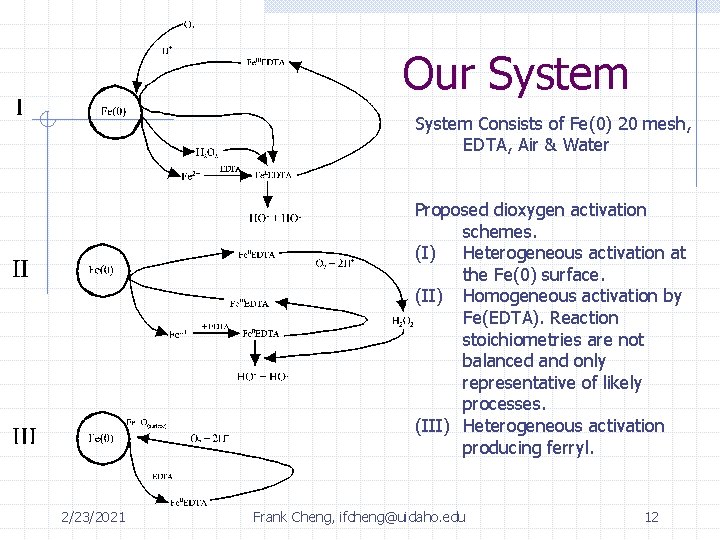
Our System Consists of Fe(0) 20 mesh, EDTA, Air & Water Proposed dioxygen activation schemes. (I) Heterogeneous activation at the Fe(0) surface. (II) Homogeneous activation by Fe(EDTA). Reaction stoichiometries are not balanced and only representative of likely processes. (III) Heterogeneous activation producing ferryl. 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 12
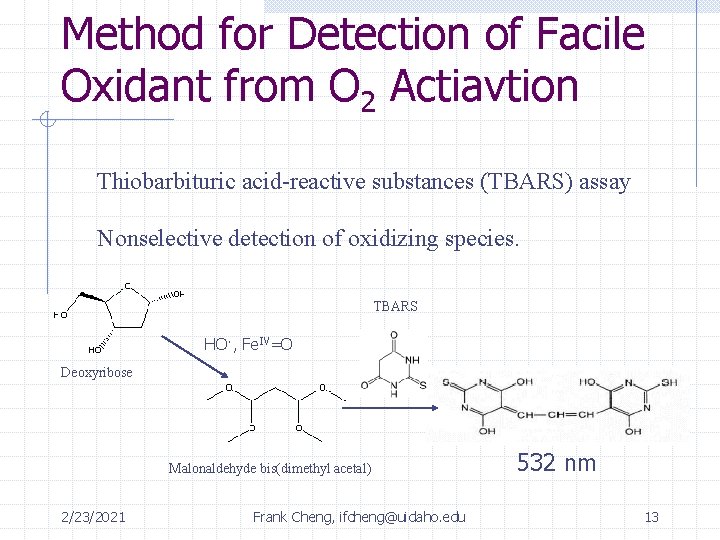
Method for Detection of Facile Oxidant from O 2 Actiavtion Thiobarbituric acid-reactive substances (TBARS) assay Nonselective detection of oxidizing species. TBARS HO·, Fe. IV=O Deoxyribose Malonaldehyde bis(dimethyl acetal) 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 532 nm 13

TBARS Results of HO· radical trapping by deoxyribose/thiobarbituric acid system forming a chromgen (532 nm). The conditions were 30 minutes of reaction time with 0. 1 g 40 -70 mesh Fe(0), under aerobic conditions. Other conditions stated below. Control 1 – 0 m. M deoxyribose, 2. 39 m. M EDTA Control 2 – 3. 18 m. M deoxyribose, 0 m. M EDTA, - also N 2 flow, -No Fe(0) 3. 18 m. M deoxyribose, 2. 39 m. M EDTA 2/23/2021 Absorbance Units at 532 nm 0. 0 Frank Cheng, ifcheng@uidaho. edu 0. 149 0. 846 14

Summary of the New System Consists of Fe(0), EDTA, Air, and Water Fulfills Several Requirements *Inexpensive Reagents – Long Shelf Life *Nontoxic *No Precious Metal Catalysts *No Special Reaction Vessels *RTP *Possibility for Portability – Field Destruction 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 15
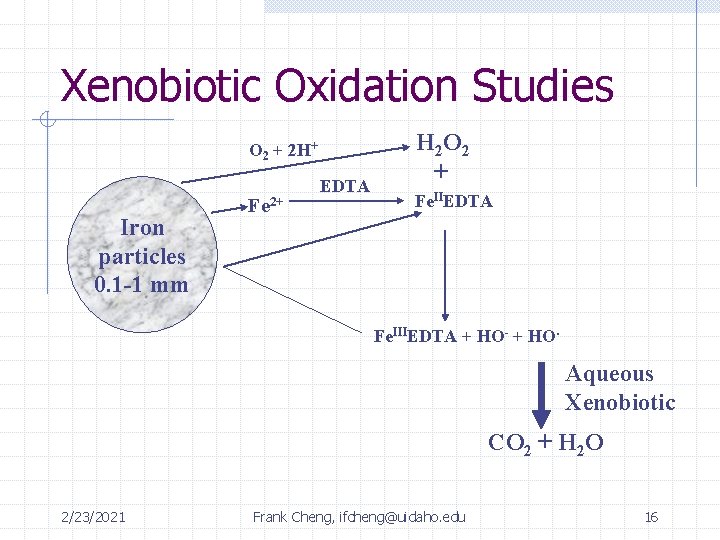
Xenobiotic Oxidation Studies O 2 + 2 H+ Iron particles 0. 1 -1 mm Fe 2+ EDTA H 2 O 2 + Fe. IIEDTA Fe. IIIEDTA + HO- + HO. Aqueous Xenobiotic CO 2 + H 2 O 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 16

Xenobiotic Oxidation Studies *Chlorophenols – recalcitrant and oxidatively stable *Malathion – Surrogate for organophosphorous compounds Kinetics Intermediates Final Products -50/50 Ethyl Acetate/Hexane GC-FID -Direct Aqueous LC-GC -Ion Chromatography 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 17

Oxidation of Chlorophenols • 0. 5 g of 40 -70 mesh Fe(0) • 10 m. L of solution • 0. 32 m. M EDTA • 140 ppm 4 -chlorophenol • 4 hour reaction time • GC and LC-MS • RTP 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 18
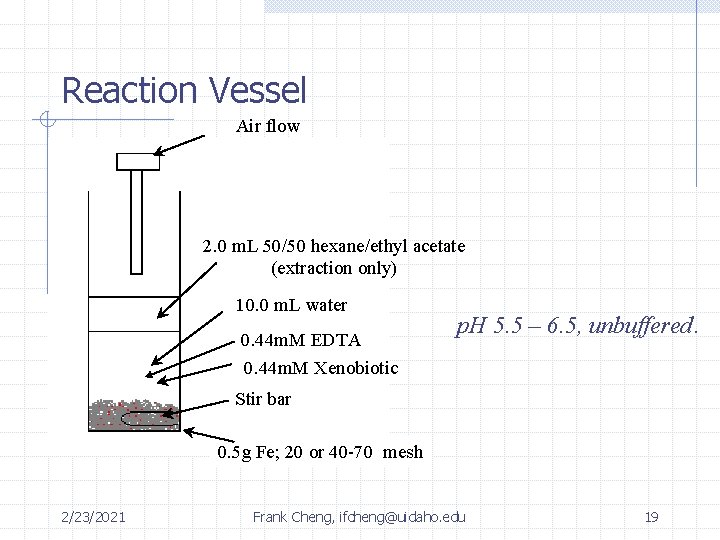
Reaction Vessel Air flow 2. 0 m. L 50/50 hexane/ethyl acetate (extraction only) 10. 0 m. L water 0. 44 m. M EDTA 0. 44 m. M Xenobiotic p. H 5. 5 – 6. 5, unbuffered. Stir bar 0. 5 g Fe; 20 or 40 -70 mesh 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 19
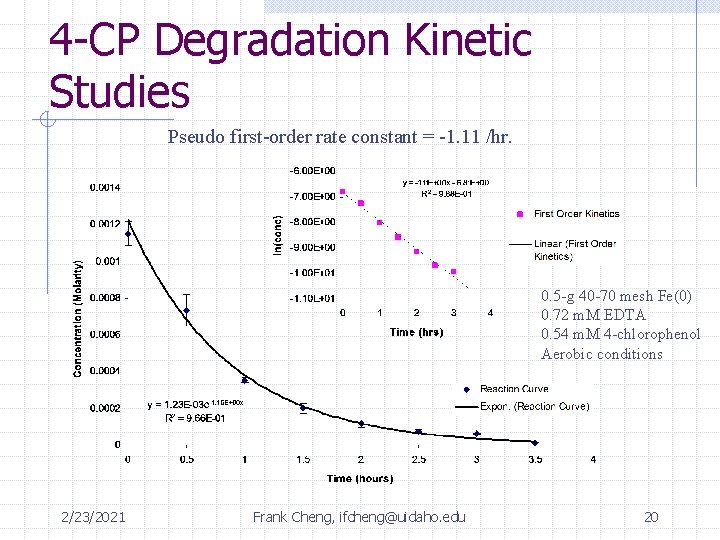
4 -CP Degradation Kinetic Studies Pseudo first-order rate constant = -1. 11 /hr. 0. 5 -g 40 -70 mesh Fe(0) 0. 72 m. M EDTA 0. 54 m. M 4 -chlorophenol Aerobic conditions 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 20

Summary of the degradation of the chlorophenols in this study All runs included 0. 5 g of 40 -70 mesh Fe(0) in 10 m. L of solution. . 140 ppm 4 chlorophenol 162 ppm Pentachlorophenol suspension 2/23/2021 I) 0. 32 m. M EDTA Aerobic II) 0. 0 m. M EDTA Aerobic III) 0. 32 m. M EDTA Anaerobic (N 2 purge) IV) dark conditions 0. 32 m. M EDTA aerobic Complete degradation after 4 h. (GCFID) Complete degradation after 70 h. (GCFID) No reaction after 4 h. Complete degradation after 4 h. No reaction after 70 h. N/A Frank Cheng, ifcheng@uidaho. edu 21

Organophosphorous Agents Malathion VX Sarin, GB 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 22

Control Studies • Control experiments conducted using: No EDTA No O 2 No Fe 0 showed no malathion degradation. Post reaction extractions of the iron solids using • Ethyl acetate • Toluene • Butanol • Hexane showed the absence of any organics absorbed to the iron surface. 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 23
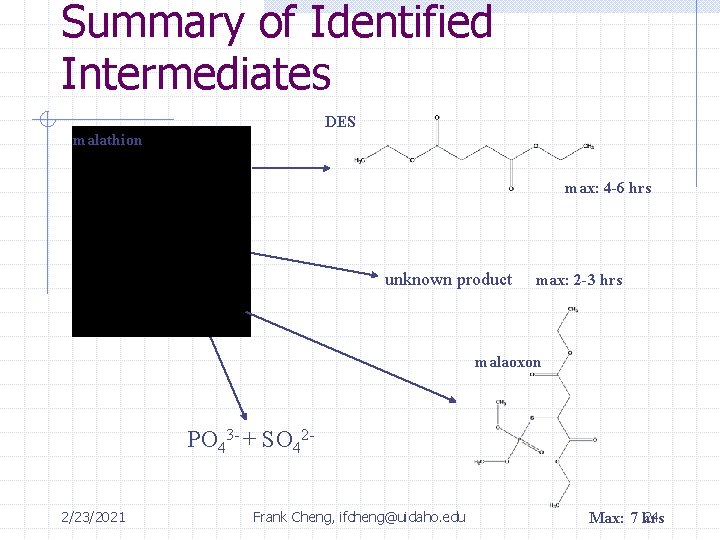
Summary of Identified Intermediates DES malathion max: 4 -6 hrs unknown product max: 2 -3 hrs malaoxon PO 43 - + SO 422/23/2021 Frank Cheng, ifcheng@uidaho. edu 24 Max: 7 hrs
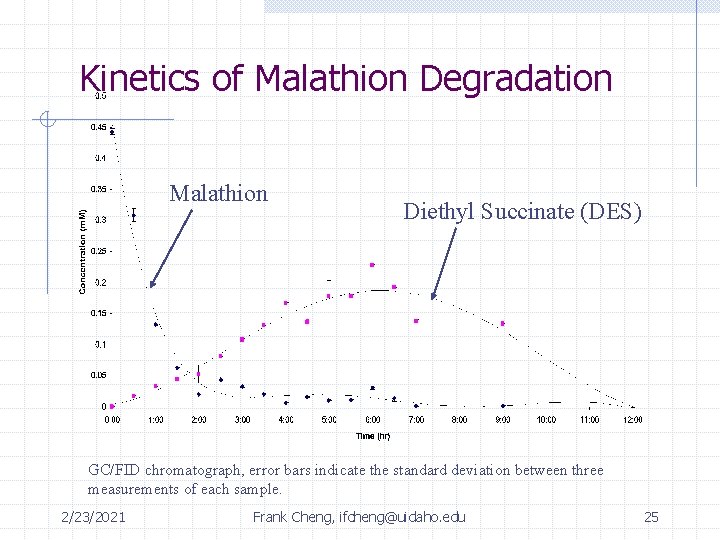
Kinetics of Malathion Degradation Malathion Diethyl Succinate (DES) GC/FID chromatograph, error bars indicate the standard deviation between three measurements of each sample. 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 25
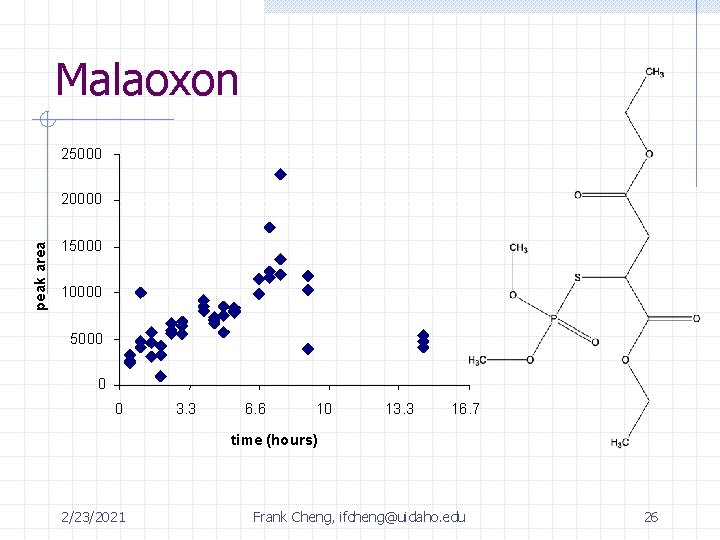
Malaoxon 25000 peak area 20000 15000 10000 5000 0 0 3. 3 6. 6 10 13. 3 16. 7 time (hours) 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 26
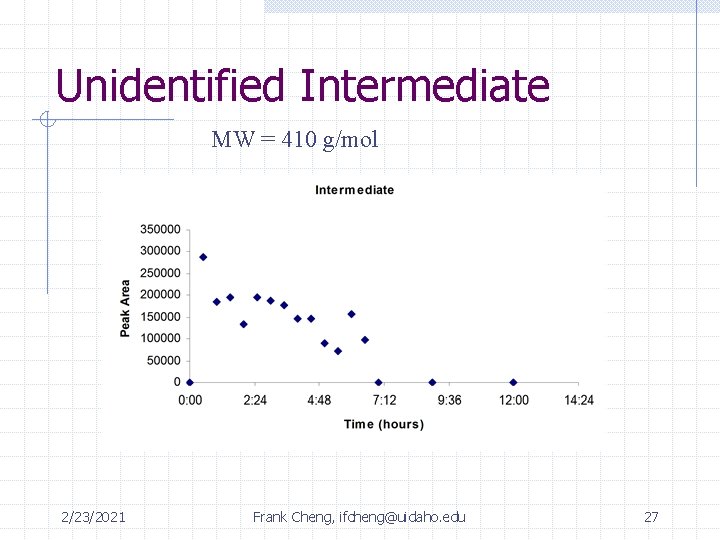
Unidentified Intermediate MW = 410 g/mol 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 27
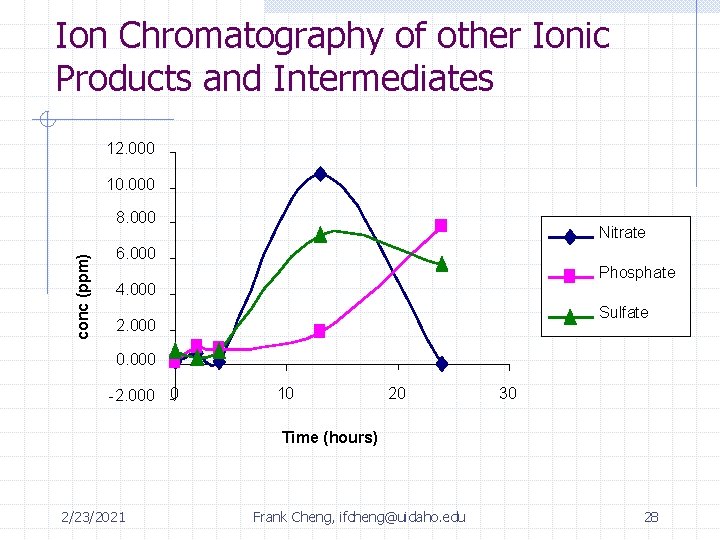
Ion Chromatography of other Ionic Products and Intermediates 12. 000 10. 000 conc (ppm) 8. 000 Nitrate 6. 000 Phosphate 4. 000 Sulfate 2. 000 0. 000 -2. 000 0 10 20 30 Time (hours) 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 28
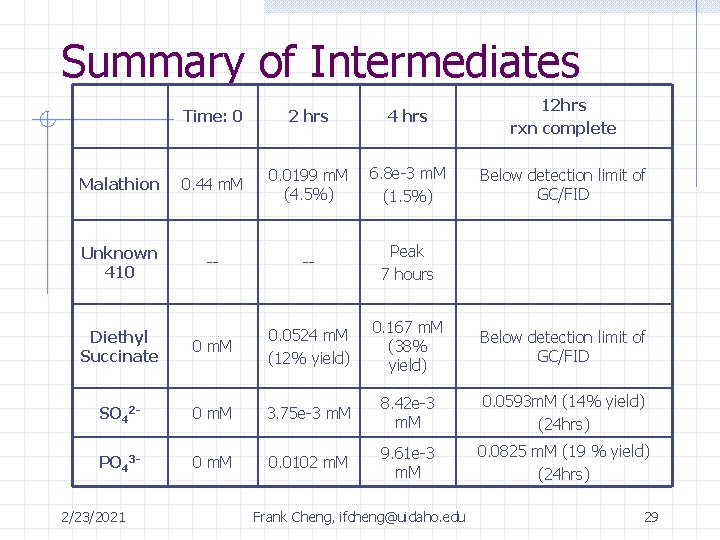
Summary of Intermediates Time: 0 2 hrs 4 hrs 12 hrs rxn complete Malathion 0. 44 m. M 0. 0199 m. M (4. 5%) 6. 8 e-3 m. M (1. 5%) Below detection limit of GC/FID Unknown 410 -- -- Peak 7 hours Diethyl Succinate 0 m. M 0. 0524 m. M (12% yield) 0. 167 m. M (38% yield) Below detection limit of GC/FID SO 42 - 0 m. M 3. 75 e-3 m. M 8. 42 e-3 m. M 0. 0593 m. M (14% yield) (24 hrs) PO 43 - 0 m. M 0. 0102 m. M 9. 61 e-3 m. M 0. 0825 m. M (19 % yield) (24 hrs) 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 29
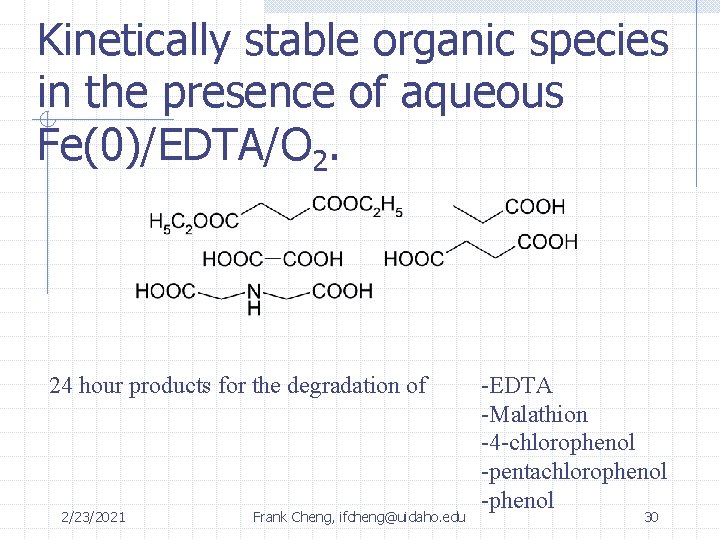
Kinetically stable organic species in the presence of aqueous Fe(0)/EDTA/O 2. 24 hour products for the degradation of 2/23/2021 Frank Cheng, ifcheng@uidaho. edu -EDTA -Malathion -4 -chlorophenol -pentachlorophenol -phenol 30
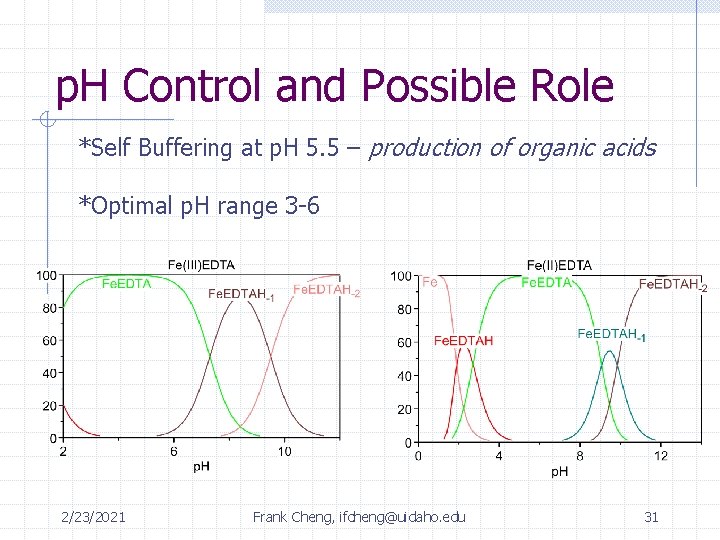
p. H Control and Possible Role *Self Buffering at p. H 5. 5 – production of organic acids *Optimal p. H range 3 -6 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 31

Technical Summary Ability to degrade organic pollutants • Under room temperature, atmospheric pressure conditions • Inexpensive Reagents – Iron particles, water, air & EDTA • Unspecialized reactors • Process is easily transportable, iron particles & EDTA • Strong possibility of scale-up • Example of a “Green Oxidant” 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 32

Scientific Summary • First example of abiotic RTP activation of O 2 able to oxidize destructively organics • Control experiments indicate process is dependent on Fe(0), EDTA, air, and water 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 33

Future Investigations • Mechanisms – Understanding the process, C balance • Kinetics – Speeding up the process • Homogeneous Systems • Search for an oxidatively stable iron chelate/organic solvent • Basic organic chemistry – oxidizable and nonoxidizable functional groups • Application & Scale-up 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 34

Desorption Studies Step 1 Starting Mixture Step 2 Step 3 0. 5 -g 40 -70 mesh Fe(0) 0. 72 m. M EDTA 0. 54 m. M 4 -chlorophenol Aerobic conditions Corrosion products observed Reaction Time 4 hours No detectable 4 -chlorophenol by GC-FID Begin N 2 purge Addition of 0. 54 m. M 4 -chlorophenol Step 5 a Aerobic Conditions Addition of 0. 72 m. M EDTA Reaction time 4 hours No 4 -chlorophenol detected by GC-FID Step 5 b Aerobic Conditions Reaction time 4 hours Approx. 50% of 4 -chlorophenol detected by GC-FID 2/23/2021 Frank Cheng, ifcheng@uidaho. edu Step 4 N 2 purge Reaction time 4 hours 0. 54 m. M 4 -chlorophenol detected by GC-FID Step 5 c N 2 purge Reaction time 4 hours 0. 54 m. M 4 -chlorophenol detected by GCFID 35
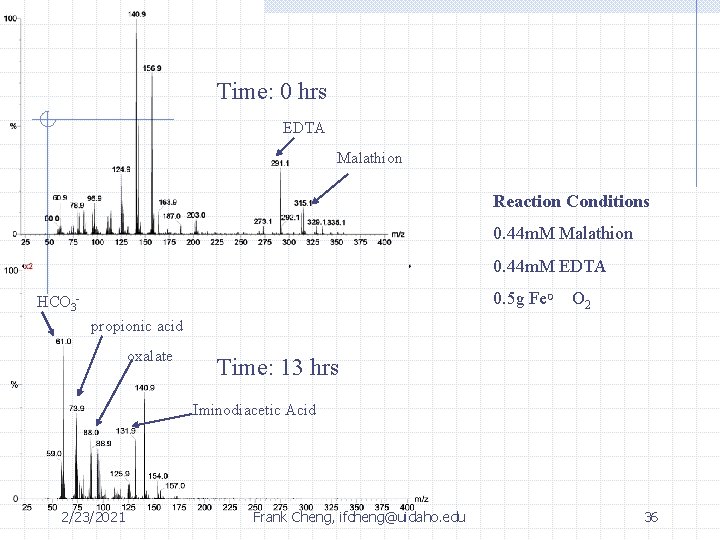
Time: 0 hrs EDTA Malathion Reaction Conditions 0. 44 m. M Malathion 0. 44 m. M EDTA 0. 5 g Fe. O O 2 HCO 3 propionic acid oxalate Time: 13 hrs Iminodiacetic Acid 2/23/2021 Frank Cheng, ifcheng@uidaho. edu 36
- Slides: 36