Conformation and Diastereomeric Specific Spectroscopic Investigation of peptides




- Slides: 23

Conformation and Diastereomeric Specific Spectroscopic Investigation of α/β-peptides Ac-Phe-β 3 -h. Ala-NHMe and Ac-β 3 -h. Ala-Phe-NHMe MG 09 William H. James III, Esteban E. Baquero, and Timothy S. Zwier Purdue University, West Lafayette IN 47907 Soo Hyuk Choi and Samuel H. Gellman University of Wisconsin-Madison, Madison WI 53706 June 16, 2008

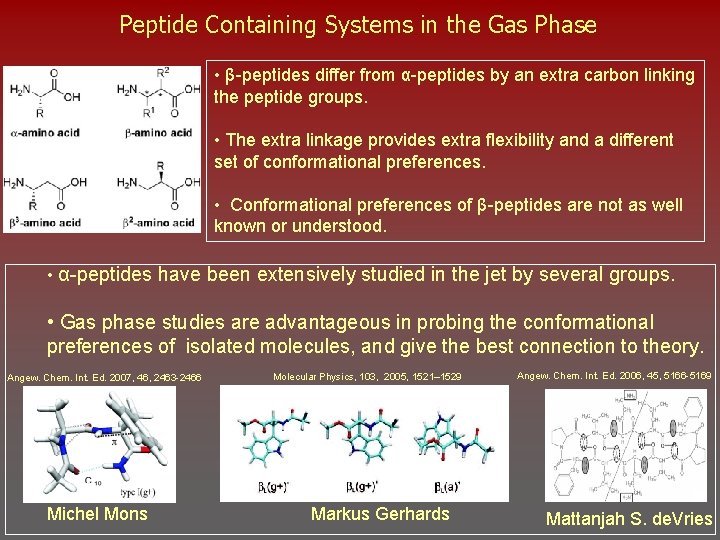
Peptide Containing Systems in the Gas Phase • β-peptides differ from α-peptides by an extra carbon linking the peptide groups. • The extra linkage provides extra flexibility and a different set of conformational preferences. • Conformational preferences of β-peptides are not as well known or understood. • α-peptides have been extensively studied in the jet by several groups. • Gas phase studies are advantageous in probing the conformational preferences of isolated molecules, and give the best connection to theory. Angew. Chem. Int. Ed. 2007, 46, 2463 -2466 Michel Mons Molecular Physics, 103, 2005, 1521– 1529 Markus Gerhards Angew. Chem. Int. Ed. 2006, 45, 5166 -5169 Mattanjah S. de. Vries

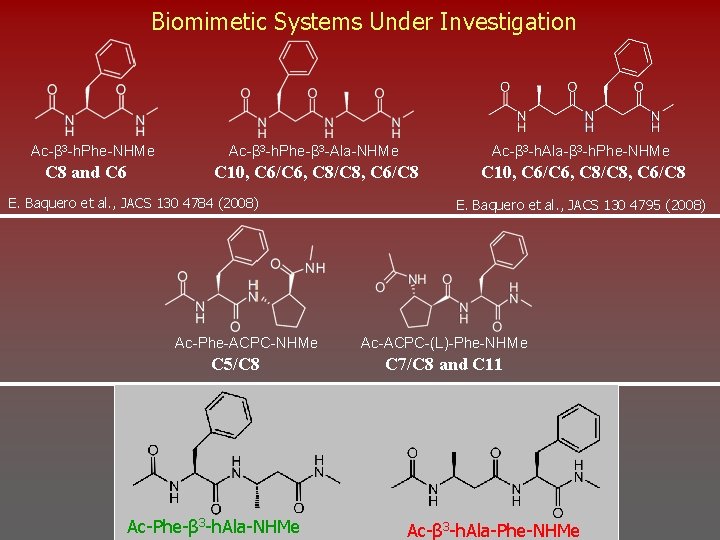
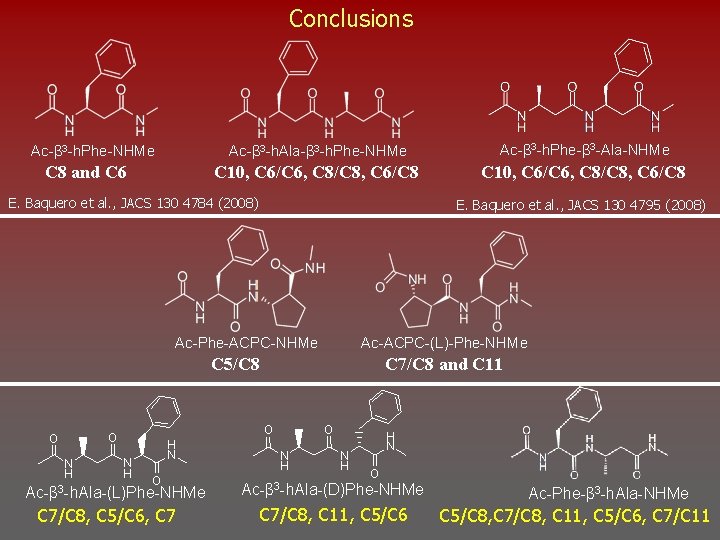
Biomimetic Systems Under Investigation Ac-β 3 -h. Phe-NHMe C 8 and C 6 Ac-β 3 -h. Phe-β 3 -Ala-NHMe Ac-β 3 -h. Ala-β 3 -h. Phe-NHMe C 10, C 6/C 6, C 8/C 8, C 6/C 8 E. Baquero et al. , JACS 130 4784 (2008) Ac-Phe-ACPC-NHMe C 5/C 8 Ac-Phe-β 3 -h. Ala-NHMe E. Baquero et al. , JACS 130 4795 (2008) Ac-ACPC-(L)-Phe-NHMe C 7/C 8 and C 11 Ac-β 3 -h. Ala-Phe-NHMe

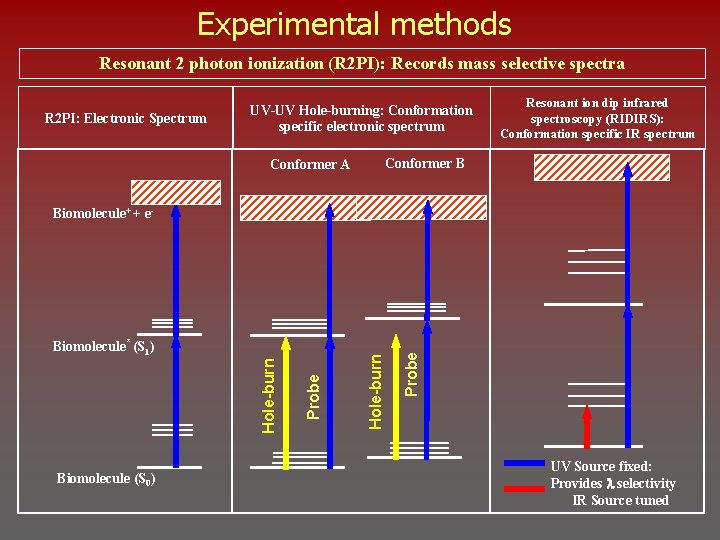
Experimental methods Resonant 2 photon ionization (R 2 PI): Records mass selective spectra R 2 PI: Electronic Spectrum UV-UV Hole-burning: Conformation specific electronic spectrum Resonant ion dip infrared spectroscopy (RIDIRS): Conformation specific IR spectrum Conformer B Conformer A Biomolecule (S 0) Probe Hole-burn Biomolecule* (S 1) Hole-burn Biomolecule+ + e- UV Source fixed: Provides l selectivity IR Source tuned

Experimental Methods Supersonic Expansion Cools gas phase molecules to effective temperatures of a few degrees Kelvin BC A D C A E B B B A A B Boltzmann distribution of the vibrational population prior to expansion UV D C C B* E B* B A C B* A Collisional cooling to zero-point vibrational level • The solid peptide sample is enclosed in a glass insert inside a stainless steel sample holder to lessen decomposition and heated to ~245 °C, ~20 °C below its melting point, prior to supersonic expansion.

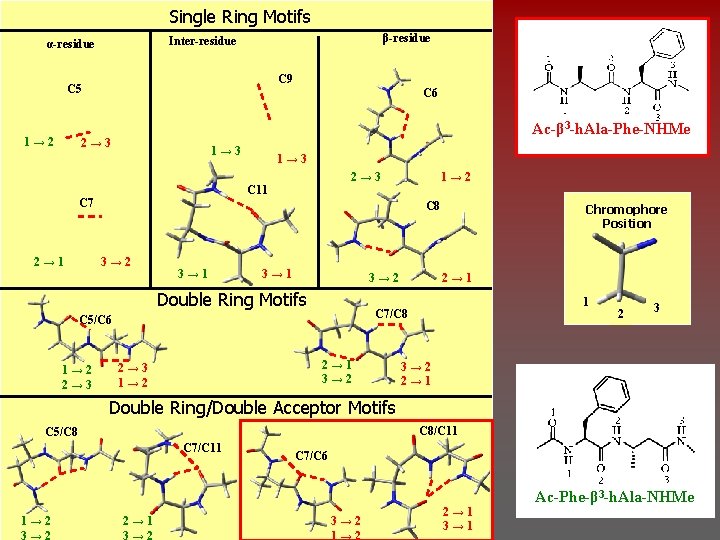
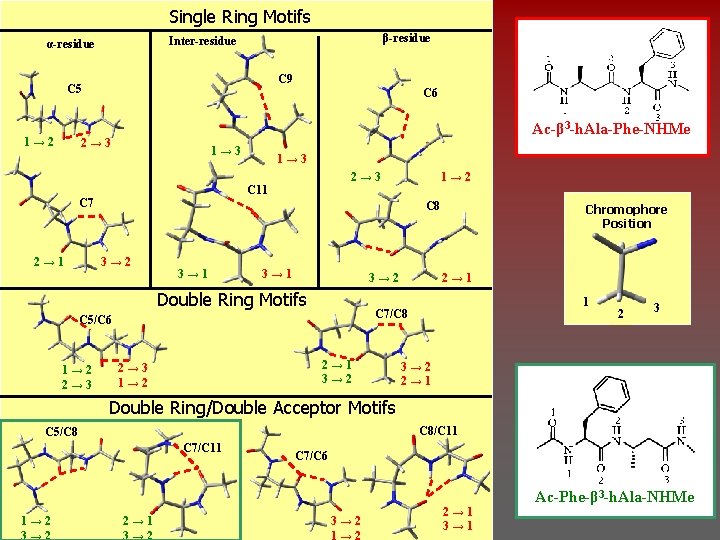
Single Ring Motifs β-residue Inter-residue α-residue C 9 C 5 1→ 2 C 6 Ac-β 3 -h. Ala-Phe-NHMe 2→ 3 1→ 3 2→ 3 C 11 C 7 2→ 1 1→ 3 C 8 3→ 2 3→ 1 3→ 2 Double Ring Motifs 2→ 1 3→ 2 2→ 3 1→ 2 Chromophore Position 2→ 1 1 C 7/C 8 C 5/C 6 1→ 2 2→ 3 1→ 2 2 3 3→ 2 2→ 1 Double Ring/Double Acceptor Motifs C 8/C 11 C 5/C 8 C 7/C 11 1→ 2 3→ 2 2→ 1 3→ 2 C 7/C 6 3→ 2 1→ 2 2→ 1 3→ 1 Ac-Phe-β 3 -h. Ala-NHMe

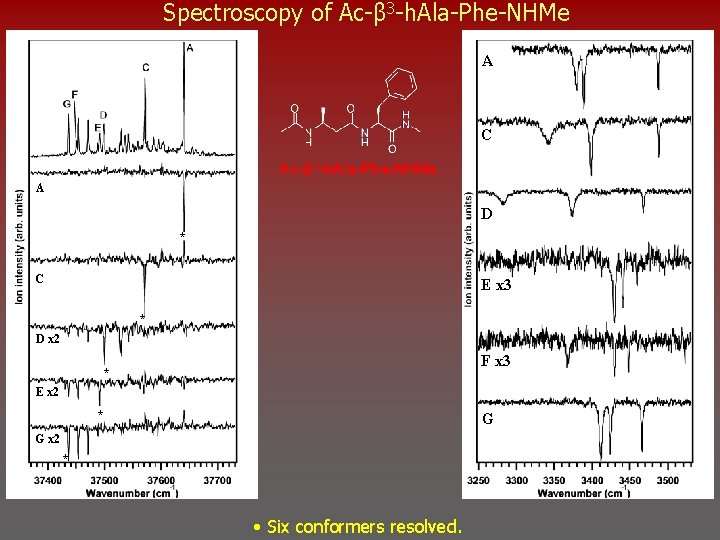
Spectroscopy of Ac-β 3 -h. Ala-Phe-NHMe A C Ac-β 3 -h. Ala-Phe-NHMe A D * C E x 3 * D x 2 F x 3 * E x 2 * G G x 2 * • Six conformers resolved.

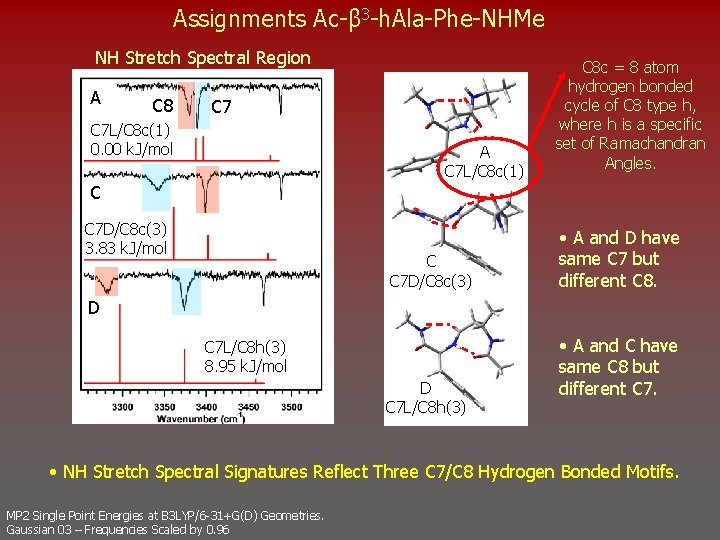
Assignments Ac-β 3 -h. Ala-Phe-NHMe NH Stretch Spectral Region A C 8 C 7 L/C 8 c(1) 0. 00 k. J/mol A C 7 L/C 8 c(1) C 8 c = 8 atom hydrogen bonded cycle of C 8 type h, where h is a specific set of Ramachandran Angles. C C 7 D/C 8 c(3) 3. 83 k. J/mol C C 7 D/C 8 c(3) • A and D have same C 7 but different C 8. D C 7 L/C 8 h(3) 8. 95 k. J/mol D C 7 L/C 8 h(3) • A and C have same C 8 but different C 7. • NH Stretch Spectral Signatures Reflect Three C 7/C 8 Hydrogen Bonded Motifs. MP 2 Single Point Energies at B 3 LYP/6 -31+G(D) Geometries. Gaussian 03 – Frequencies Scaled by 0. 96

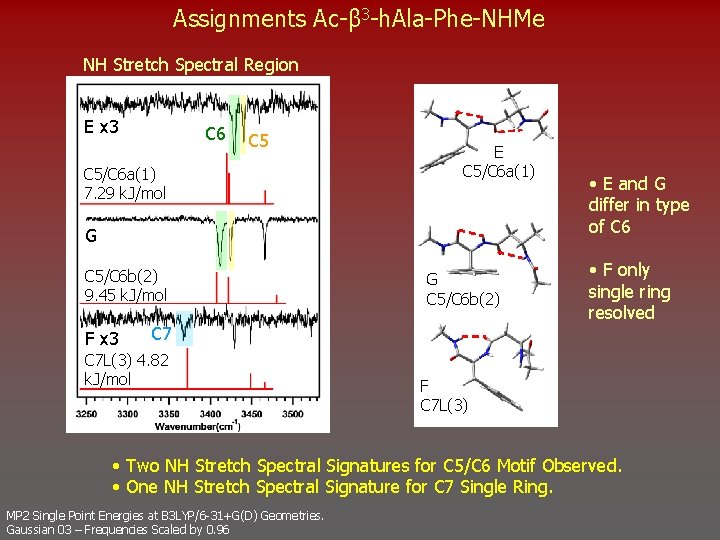
Assignments Ac-β 3 -h. Ala-Phe-NHMe NH Stretch Spectral Region E x 3 C 6 C 5/C 6 a(1) 7. 29 k. J/mol E C 5/C 6 a(1) G C 5/C 6 b(2) 9. 45 k. J/mol F x 3 G C 5/C 6 b(2) C 7 L(3) 4. 82 k. J/mol • E and G differ in type of C 6 • F only single ring resolved F C 7 L(3) • Two NH Stretch Spectral Signatures for C 5/C 6 Motif Observed. • One NH Stretch Spectral Signature for C 7 Single Ring. MP 2 Single Point Energies at B 3 LYP/6 -31+G(D) Geometries. Gaussian 03 – Frequencies Scaled by 0. 96

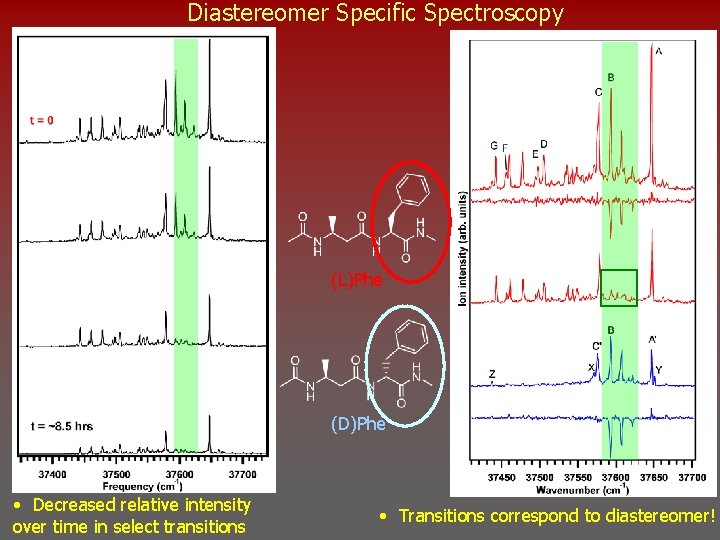
Diastereomer Specific Spectroscopy (L)Phe (D)Phe • Decreased relative intensity over time in select transitions • Transitions correspond to diastereomer!

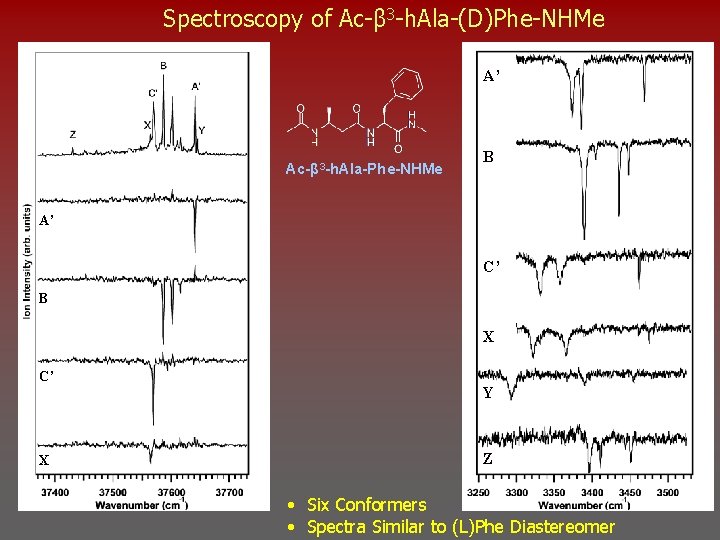
Spectroscopy of Ac-β 3 -h. Ala-(D)Phe-NHMe A’ Ac-β 3 -h. Ala-Phe-NHMe B A’ C’ B X C’ X Y Z • Six Conformers • Spectra Similar to (L)Phe Diastereomer

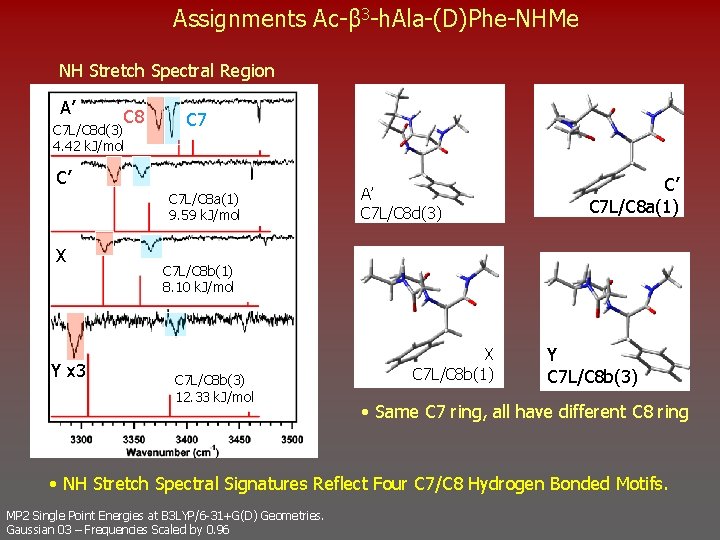
Assignments Ac-β 3 -h. Ala-(D)Phe-NHMe NH Stretch Spectral Region A’ C 8 C 7 L/C 8 d(3) 4. 42 k. J/mol C 7 C’ C 7 L/C 8 a(1) 9. 59 k. J/mol X Y x 3 A’ C 7 L/C 8 d(3) C’ C 7 L/C 8 a(1) C 7 L/C 8 b(1) 8. 10 k. J/mol C 7 L/C 8 b(3) 12. 33 k. J/mol X C 7 L/C 8 b(1) Y C 7 L/C 8 b(3) • Same C 7 ring, all have different C 8 ring • NH Stretch Spectral Signatures Reflect Four C 7/C 8 Hydrogen Bonded Motifs. MP 2 Single Point Energies at B 3 LYP/6 -31+G(D) Geometries. Gaussian 03 – Frequencies Scaled by 0. 96

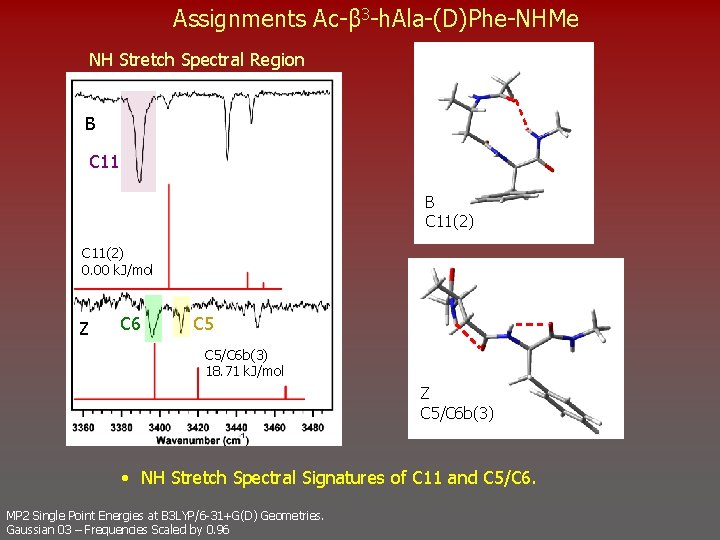
Assignments Ac-β 3 -h. Ala-(D)Phe-NHMe NH Stretch Spectral Region B C 11(2) 0. 00 k. J/mol Z C 6 C 5/C 6 b(3) 18. 71 k. J/mol Z C 5/C 6 b(3) • NH Stretch Spectral Signatures of C 11 and C 5/C 6. MP 2 Single Point Energies at B 3 LYP/6 -31+G(D) Geometries. Gaussian 03 – Frequencies Scaled by 0. 96

Single Ring Motifs β-residue Inter-residue α-residue C 9 C 5 1→ 2 C 6 Ac-β 3 -h. Ala-Phe-NHMe 2→ 3 1→ 3 2→ 3 C 11 C 7 2→ 1 1→ 3 C 8 3→ 2 3→ 1 3→ 2 Double Ring Motifs 2→ 1 3→ 2 2→ 3 1→ 2 Chromophore Position 2→ 1 1 C 7/C 8 C 5/C 6 1→ 2 2→ 3 1→ 2 2 3 3→ 2 2→ 1 Double Ring/Double Acceptor Motifs C 8/C 11 C 5/C 8 C 7/C 11 1→ 2 3→ 2 2→ 1 3→ 2 C 7/C 6 3→ 2 1→ 2 2→ 1 3→ 1 Ac-Phe-β 3 -h. Ala-NHMe

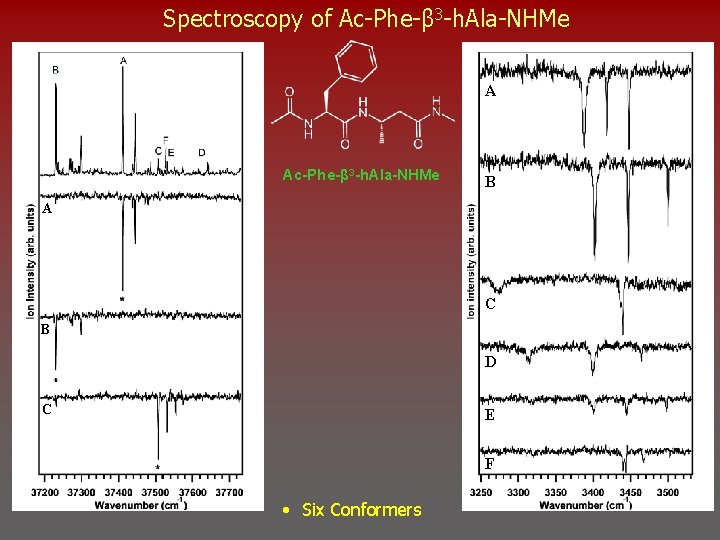
Spectroscopy of Ac-Phe-β 3 -h. Ala-NHMe A Ac-Phe-β 3 -h. Ala-NHMe B A C B D C E F • Six Conformers

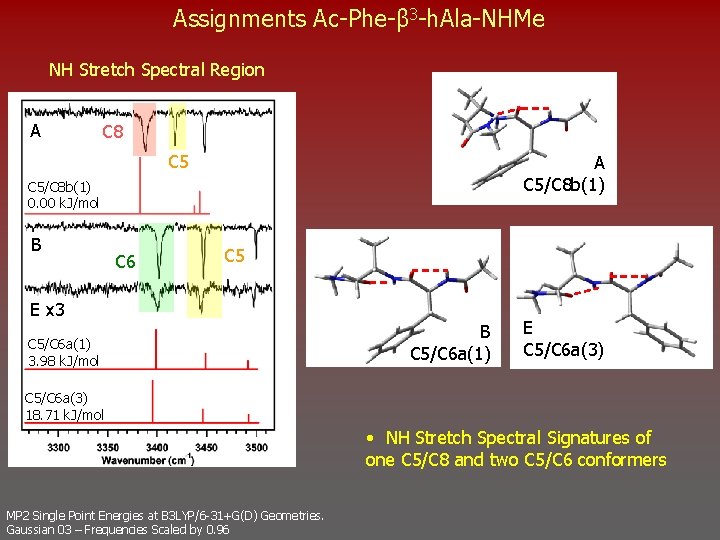
Assignments Ac-Phe-β 3 -h. Ala-NHMe NH Stretch Spectral Region A C 8 C 5 A C 5/C 8 b(1) 0. 00 k. J/mol B C 6 C 5 E x 3 C 5/C 6 a(1) 3. 98 k. J/mol B C 5/C 6 a(1) E C 5/C 6 a(3) 18. 71 k. J/mol • NH Stretch Spectral Signatures of one C 5/C 8 and two C 5/C 6 conformers MP 2 Single Point Energies at B 3 LYP/6 -31+G(D) Geometries. Gaussian 03 – Frequencies Scaled by 0. 96

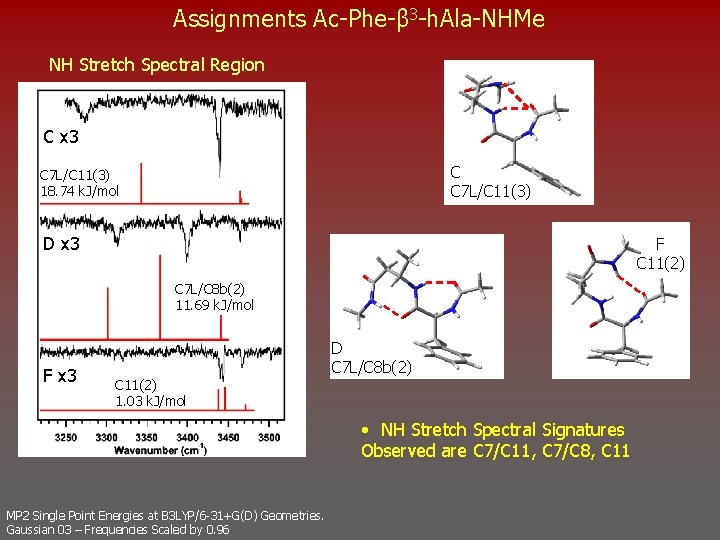
Assignments Ac-Phe-β 3 -h. Ala-NHMe NH Stretch Spectral Region C x 3 C C 7 L/C 11(3) 18. 74 k. J/mol D x 3 F C 11(2) C 7 L/C 8 b(2) 11. 69 k. J/mol F x 3 C 11(2) 1. 03 k. J/mol D C 7 L/C 8 b(2) • NH Stretch Spectral Signatures Observed are C 7/C 11, C 7/C 8, C 11 MP 2 Single Point Energies at B 3 LYP/6 -31+G(D) Geometries. Gaussian 03 – Frequencies Scaled by 0. 96

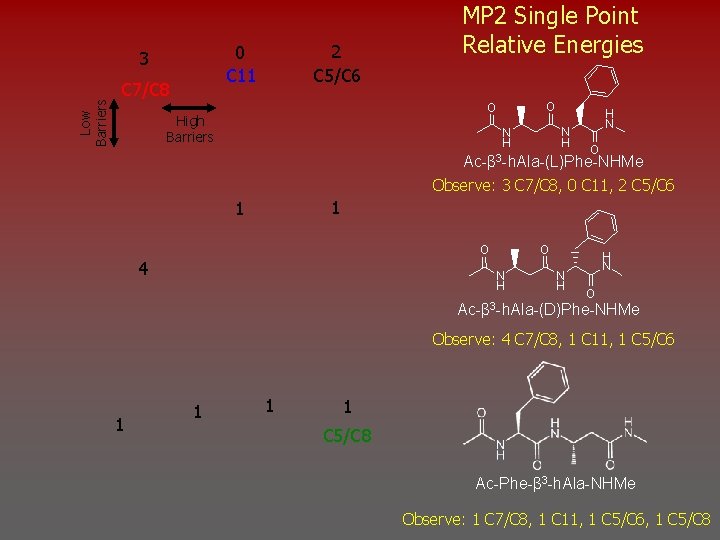
Low Barriers 2 C 5/C 6 0 C 11 3 C 7/C 8 MP 2 Single Point Relative Energies O O High Barriers N H H N O Ac-β 3 -h. Ala-(L)Phe-NHMe Observe: 3 C 7/C 8, 0 C 11, 2 C 5/C 6 1 1 O 4 O N H H N O Ac-β 3 -h. Ala-(D)Phe-NHMe Observe: 4 C 7/C 8, 1 C 11, 1 C 5/C 6 1 1 C 5/C 8 Ac-Phe-β 3 -h. Ala-NHMe Observe: 1 C 7/C 8, 1 C 11, 1 C 5/C 6, 1 C 5/C 8

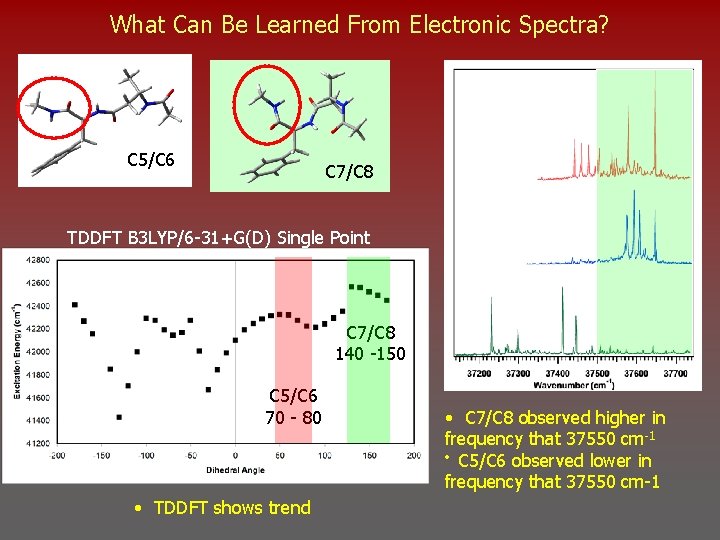
What Can Be Learned From Electronic Spectra? C 5/C 6 C 7/C 8 TDDFT B 3 LYP/6 -31+G(D) Single Point C 7/C 8 140 -150 C 5/C 6 70 - 80 • TDDFT shows trend • C 7/C 8 observed higher in frequency that 37550 cm-1 • C 5/C 6 observed lower in frequency that 37550 cm-1

Conclusions Ac-β 3 -h. Phe-NHMe C 8 and C 6 Ac-β 3 -h. Ala-β 3 -h. Phe-NHMe Ac-β 3 -h. Phe-β 3 -Ala-NHMe C 10, C 6/C 6, C 8/C 8, C 6/C 8 E. Baquero et al. , JACS 130 4784 (2008) E. Baquero et al. , JACS 130 4795 (2008) Ac-Phe-ACPC-NHMe Ac-ACPC-(L)-Phe-NHMe C 5/C 8 O O O N H C 7/C 8 and C 11 N H H N O Ac-β 3 -h. Ala-(L)Phe-NHMe C 7/C 8, C 5/C 6, C 7 O N H H N O Ac-β 3 -h. Ala-(D)Phe-NHMe C 7/C 8, C 11, C 5/C 6 Ac-Phe-β 3 -h. Ala-NHMe C 5/C 8, C 7/C 8, C 11, C 5/C 6, C 7/C 11

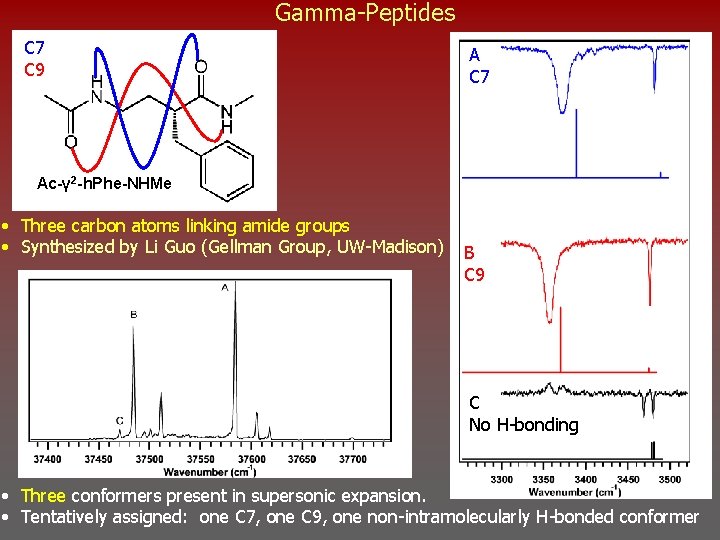
Gamma-Peptides C 7 C 9 A C 7 Ac-γ 2 -h. Phe-NHMe • Three carbon atoms linking amide groups • Synthesized by Li Guo (Gellman Group, UW-Madison) B C 9 C No H-bonding • Three conformers present in supersonic expansion. • Tentatively assigned: one C 7, one C 9, one non-intramolecularly H-bonded conformer

Acknowledgements Professor Samuel H. Gellman Soo Hyuk Choi Li Guo Professor Timothy S. Zwier Current Group Members: Dr. Christian Müller Tracy Le. Greve Nathan Pillsbury Joshua Newby Chirantha Rodrigo Joshua Sebree Evan Buchanan Former Group Members: Dr. Jaime Stearns Dr. Talitha Selby Dr. Jasper Clarkson Dr. Ching-Ping Liu Dr. Esteban Baquero Dr. V. Alvin Shubert FUNDING Computational Resources
