Electronic spectra of transition metal complexes B Sc

Electronic spectra of transition metal complexes B. Sc IIIrd year Prof. Sonu Sharma Chemistry Department D. A. V. College, Hoshiarpur

Term symbol �Symbols which are used to represent the electronic configuration and the resultant angular momentum of an atom. 2 S+1 L J S= total spin angular quantum number L= Total orbital angular quantum number J= Total angular momentum quantum number 2 S+1 = spin multiplicity of the state

Rules to find ground state term – Hund rules �State having the highest S value is most stable. �The state with the highest value of L is the most stable state. �The state with the smallest J value is most stable if the sub shell is less than half filled and the state with highest J value is most stable if the subshell is more than half filled.
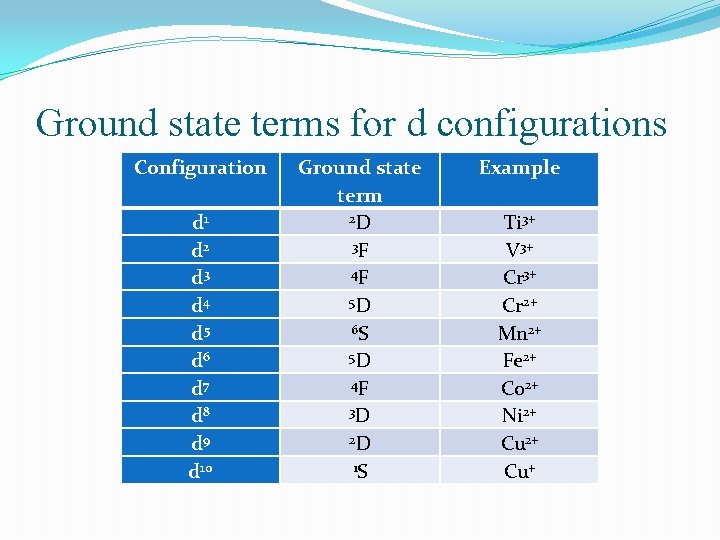
Ground state terms for d configurations Configuration d 1 d 2 d 3 d 4 d 5 d 6 d 7 d 8 d 9 d 10 Ground state term 2 D 3 F 4 F 5 D 6 S 5 D 4 F 3 D 2 D 1 S Example Ti 3+ V 3+ Cr 2+ Mn 2+ Fe 2+ Co 2+ Ni 2+ Cu+

Correlation of spectroscopic terms into Mulliken symbol Spectroscopic Term Mulliken symbol in Octahedral field S P D F G A 1 g T 1 g Eg+T 2 g A 2 g+T 1 g+T 2 g A 1 g+Eg+T 1 g+T 2 g

Orgel Diagrams �Orgel diagrams are the correlation diagrams which shows the variation of energy levels of spectroscopic states of different symmetry as a function of field strength. �These diagrams are named after their creator Leslie Orgel.

Some basic rules 1. dn and dn+5 have same orgel diagrams. 2. dn octahedral diagram is reverse of dn tetrahedral diagram. 3. dn is reverse of d 10 -n.
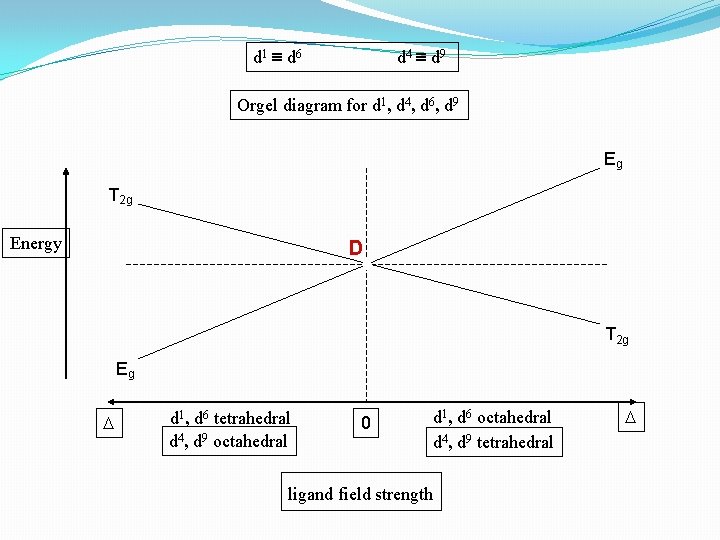
d 1 d 6 d 4 d 9 Orgel diagram for d 1, d 4, d 6, d 9 Eg T 2 g Energy D T 2 g Eg D d 1, d 6 tetrahedral d 4, d 9 octahedral 0 ligand field strength d 1, d 6 octahedral d 4, d 9 tetrahedral D
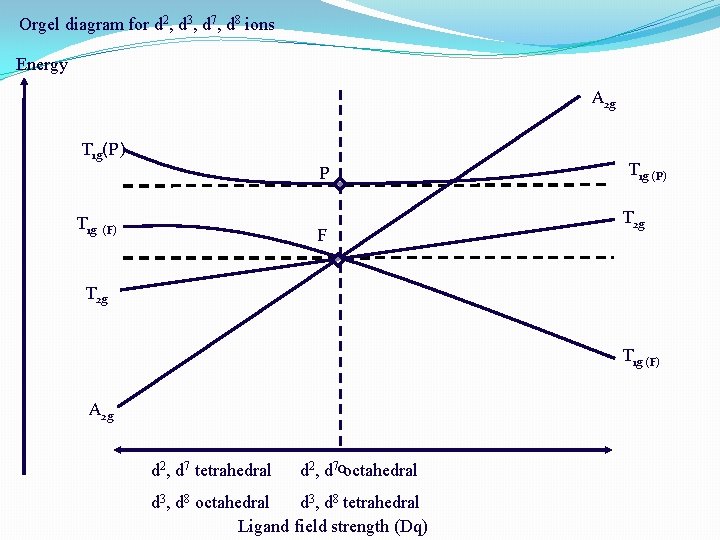
Orgel diagram for d 2, d 3, d 7, d 8 ions Energy A 2 g T 1 g(P) T 1 g P (F) F T 1 g (P) T 2 g T 1 g (F) A 2 g d 2, d 7 tetrahedral d 2, d 70 octahedral d 3, d 8 tetrahedral Ligand field strength (Dq)

In this diagram, the two T 1 g states, one from P state and other from F state are slightly curved lines. This is because the two T 1 g terms have same symmetry and hence repel each other.

Limitations of Orgel Diagrams 1. These diagrams are qualitative in nature. 2. These diagrams consider only high spin (weak field) cases. 3. These diagrams takes into account spin allowed transitions only.
- Slides: 11