Chem 140 Section A Instructor Ken Marr Weekly















































- Slides: 53

Chem 140 Section A Instructor: Ken Marr Weekly Schedule “Lecture” “Lab” 9 -10, MWF in STB-2 8 -10 , Tu in STB-2 8 -10 , Th in STB-5

Chem 140 Section C Instructor: Ken Marr Weekly Schedule “Lecture” “Lab” 10 – 11, MWF in STB-2 10 -12 , Tu in STB-2 10 -12 , Th in STB-5

Chem 140 Section E Instructor: Ken Marr Weekly Schedule “Lecture” “Lab” 1 - 2, MWF in STB-2 1 - 3 , Tu in STB-2 1 - 3 , Th in STB-5

Day 1 Activities l Introduction to Course » Briefly Review Course Outline/Syllabus l Homework Assignments » Reading: See Chem 140 schedule » Lab: Do Prelab assignment for the “Measurement and Density” Lab » Stamped Assignment #1: Chapter 1 HW – due Tues. 10/01/02 ……But start now!!! – Note: 10/2 is a very special day for your instructor!! l Begin Chapter 1 » Alice 1 and 2

CHEMISTRY The Study of Matter and the Changes that Matter Undergoes and The Energy Associated with The Changes

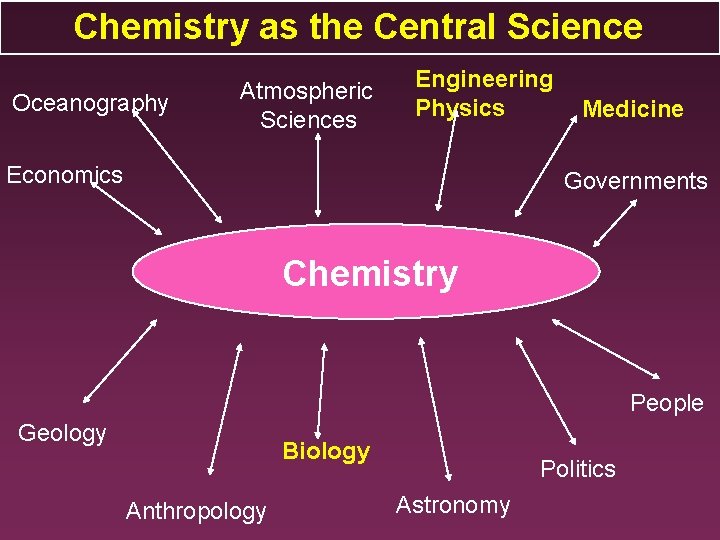
Chemistry as the Central Science Oceanography Atmospheric Sciences Engineering Physics Economics Medicine Governments Chemistry People Geology Biology Anthropology Politics Astronomy

Chapter# 1 : Keys to the Study of Chemistry 1. 1 Some Fundamental Definitions 1. 2 Chemical Arts and the Origins of Modern Chemistry 1. 3 The Scientific Approach: Developing a Model 1. 4 Chemical Problem Solving 1. 5 Measurement in Scientific Study 1. 6 Uncertainty in Measurement: Significant Figures

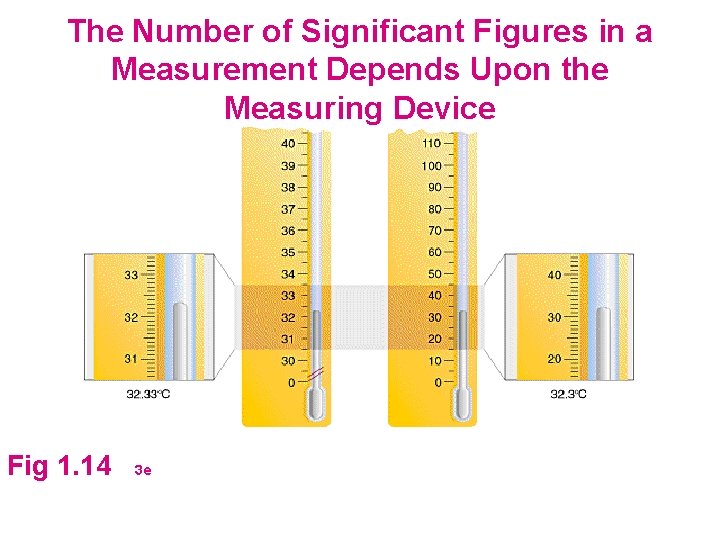
Measurement and Significant Figures l Measured Numbers are Never Exact. . . Why? » Which Graduated Cylinder is the most precise? » How is precision indicated when we record a measurement?

The Number of Significant Figures in a Measurement Depends Upon the Measuring Device Fig 1. 14 3 e

Significant Figures l We use significant figures to indicate the maximum precision of a measurement l Significant Figures » The number of digits that are known with certainty, plus one that is uncertain » Significant figures are used only with measured quantities. » Some numbers are exact and do not have any uncertainty. . . e. g. . . ’s? ?

More Examples Record the exact length in centimeters, cm (T 2 c) l Record the exact amounts for numbers 1 -11 (T 2 d) l

Sig. Fig. Rules to memorize. . . (See page 27 -30 Silberberg 3 e) 1. All nonzero numbers are significant e. g. 2. 3. 23. 8 g, 2345 km, 11 m. L, 5 inches Zeros between nonzero digits are significant i. e. Sandwiched zeros are significant e. g. 509 m, 2001 m. L, 2050. 1 L Zeros preceding the 1 st nonzero digit are not significant. . . . they serve only to locate the decimal point e. g. 0. 083 m, 0. 000306 L – Try converting these numbers to Scientific Notation to prove this!

More Sig. Fig. Rules Involving Zeros 4. Zeros at the end of number that include a decimal point are significant » 5. 0. 800, 11. 40, 10. 00, 400. Zeros at the end of a number without a decimal point are not significant. . . The Greenwater Rule! » 40, 8800, 300, – Use of underlining and decimal points

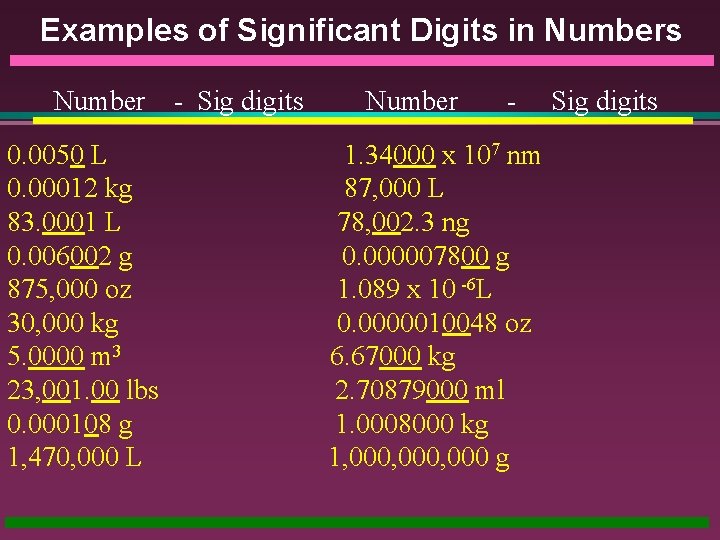
Examples of Significant Digits in Numbers Number 0. 0050 L 0. 00012 kg 83. 0001 L 0. 006002 g 875, 000 oz 30, 000 kg 5. 0000 m 3 23, 001. 00 lbs 0. 000108 g 1, 470, 000 L - Sig digits Number - 1. 34000 x 107 nm 87, 000 L 78, 002. 3 ng 0. 000007800 g 1. 089 x 10 -6 L 0. 0000010048 oz 6. 67000 kg 2. 70879000 ml 1. 0008000 kg 1, 000, 000 g Sig digits

Rounding off Numbers l Rounding off is used to drop non-significant numbers » Rule 1 When the 1 st digit after those you want to retain is 4 of less, that digit and all others to the right are dropped l Round off the following to 3 sig. figs. » 105. 29, 189. 49999, 1. 003, 100. 3, 1001

Rounding off Numbers l Rule 2 When the 1 st digit after those you want to retain is 5 or greater, that digit and all others to the right are dropped and the last digit retained is increased by one l Round off the following to 4 sig. figs. » 10. 87519, 13. 59800, 99. 999, 1042. 5

Sig. Figs. in Calculations l The Central Idea. . . » The result of a calculation based on measurements can not be more precise than the least precise measurement! l Some Rules to, yes, memorize. . .

Sig. Figs. in Multiplication and Division l “The Chain Rule” » Your answer must contain the same number of sig figs as the measurement with the fewest sig figs. . . Some e. g. . . ’s. . . (3. 04) x (2. 2) = 6. 688 = ? ? ? (2. 00) / (0. 3 ) = 6. 666. . . = ? ? ? (18. 4) x (4. 0) = 1. 1117824 = ? ? ? (66. 2)

Sig. Figs. in Addition and Subtraction l “The Decimal Rule” » The answer must have the same precision as the least precise measurement. . . or. . . – Your answer must be expressed to the same number of decimal places as the measurement with the fewest decimal places. l The number of sig figs are not considered, only the number of decimal places are considered!!! » Some examples. .

Sig. Figs. in Addition and Subtraction l Examples. . . » 12. 89 + 12. 1 + 11. 803 + 19 = 55. 793 = ? » 1786 - 130 = 1656 = ? ? ? » 7331 + 0. 495 = 7331. 495 = ? ? ?

Scientific Notation l Scientific Notation » Writing a number as a number between 1 and 10 times a power of 10 » WHY DO IT? ? ? l The Rules. . .

How to Write Numbers in Scientific Notation 1. Move the decimal point in the original number so that it is located after the first nonzero digit » e. g. 5682 ? ? 2. Multiply this number by the proper power of 10 » The power of 10 is equal to the number of places the decimal point was moved. l l POSITIVE IF MOVED TO THE LEFT NEGATIVE IF MOVED TO THE RIGHT

Examples. . l Express the following numbers in scientific notation. . . » 0. 0421 » 150, 000 » 5899 l Express the following in “longhand” » 5. 30 x 10 -4 » 8. 000 x 106

Meaning of Powers of 10 103 l 102 l 101 l 100 l = = 10 -3 = 10 -2 = 10 -1 =

Metric System of measure built around standard or base units l Uses factors of 10 to express larger or smaller numbers of these units l

Table 1. 2 (p. 17, 3 e) SI - Base Units Physical Quantity Unit Name Abbreviation Mass Kilogram kg Length meter m Time second s Temperature Kelvin K Electric current ampere A Amount of substance mole Luminous intensity candela mol cd

Metric Base Units and their Abbreviations Length l Mass l Volume l Temperature l » Prefixes are added to these base units for quantities larger or smaller than the base unit – Prefixes are a multiple of 10

Table 1. 3 Prefix tera giga Mega Kilo hecto deka ----deci centi milli micro nano pico femto Common Decimal Prefixes Used with SI Units. Prefix Symbol Number Word Exponential Notation T 1, 000, 000 trillion G 1, 000, 000 billion M 1, 000 million k 1, 000 thousand h 100 hundred da 10 ten ---1 one d 0. 1 tenth c 0. 01 hundredth m 0. 001 thousandth millionth n 0. 00001 billionth p 0. 0000001 trillionth f 0. 00000001 quadrillionth 1012 109 106 103 102 101 100 10 -1 10 -2 10 -3 10 -6 10 -9 10 -12 10 -15

Common Metric Prefixes l Memorize the Symbol, Numerical Value, and Power of 10 Equivalent for. . . » kilo» centi» milli» micro» nano-

Common Prefix Applications l Length: » km » cm » mm » µm » nm 1 km = ? 1 cm = ? 1 mm = ? 1 µm = ? 1 nm = ? m m m

Common Prefix Applications l Mass » kg » mg » µg 1 kg = ? g 1 mg = ? g 1 µg = ? g

Common Prefix Applications l Volume » m. L » µL 1 m. L = ? L 1 µL = ? L

Important Relationships l Length » 1 m = » 1 cm = ? ? cm ? ? mm ? ? µm ? ? mm

Important Relationships l Mass » 1 g = ? ? mg » 1 kg = ? ? lb. .

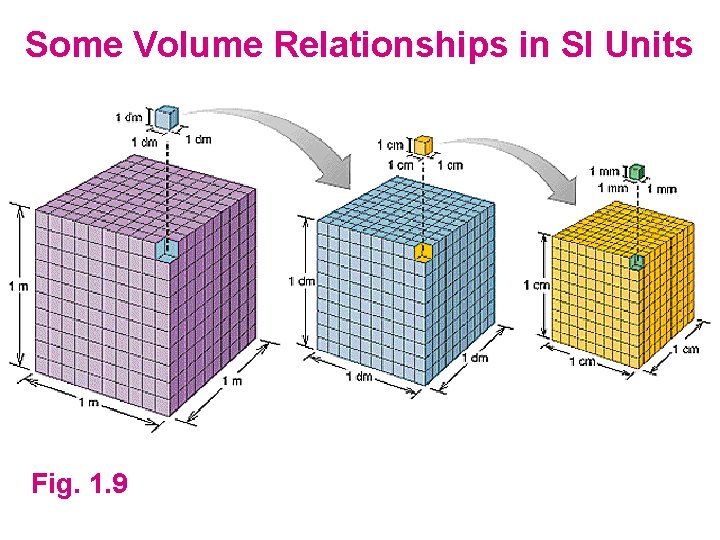
Some Volume Relationships in SI Units Fig. 1. 9

Important Relationships l Volume » 1 L = ? ? m. L » 1 m. L = ? ? cm 3 » 1 L = 1. 057 qt.

Solving Chemistry Problems Develop a Plan Carryout Plan Check Answer 1. Developing a Plan: Read the problem carefully! 1. Clarify the know and unknown: 1. 2. What information is given? What are you trying to find? 2. Think about how to solve the problem before you start to juggle numbers 1. 2. 3. Suggest steps from the known to unknown Determine principles involved and the relationships needed Use sample problems as a guides 3. Map out the strategy you will follow

Solving Chemistry Problems (cont. ) 2. Solve the problem: Carry out your plan » Set up problem in a neat, organized, and logical way! » Unwanted units should cancel to give the desired unit of measure » Make a rough estimate of the answer before using your calculator » Round off to correct number of sig. figs. » Answer must have correct units

Solving Chemistry Problems (cont. ) 3. Check your answer » Is it reasonable? » Correct nits? » Same “ballpark” as a rough estimate? » Makes chemical sense?

Problem Solving: Some Examples 1. How many hours would it take a pump to remove the water from a flooded basement that is about 30 feet wide and 50 feet long with water at a depth of about 2 feet? The pump has a capacity of 80 liters per minute. See Table 1. 4, Common SI-English Equivalent Quantities, page 18 Silberberg 3 e. 1062 min = 17. 7 hours = 20 hours

Metric Conversion Factors l Be able to do conversions within the metric system involving the common metric prefixes » kilo» centi» milli» micro» nano– e. g. #32 on page 43

Metric - English Conversions Given metric - English conversion factors, be able to convert between these two systems l You do not have to memorize metric to English conversions factors l

Measurement of Temperature l Heat vs. Temperature » Temperature (SI unit: Kelvin, K) – A measure of how hot or cold an object is relative to another object – Also measured in degrees Celsius, o. C » Heat (SI unit: joule, J) – The energy transferred between objects at different temperatures – A form of energy associated with the motion of atoms and molecules (the small particles of matter) – Also measured in calories, cal

Application: Heat vs. Temperature l Which contains more heat. . . » 1 m. L of water at 90 o. C or 1 liter of water at 90 o. C ? » 1 burning match or 10 burning matches?

Temperature Conversions The boiling point of Liquid Nitrogen is - 195. 8 o. C, what is the temperature in Kelvin and degrees Fahrenheit? T (in K) = T (in o. C) + 273. 15 T (in K) = -195. 8 + 273. 15 = 77. 35 K = 77. 4 K T (in o. F) = 9/5 T (in o. C) + 32 T (in o. F) = 9/5 ( -195. 8 o. C) +32 = -320. 4 o. F The normal body temperature is 98. 6 o. F, what is it in Kelvin and degrees Celsius? T (in o. C) = [ T (in o. F) - 32] 5/9 T (in o. C) = [ 98. 6 o. F - 32] 5/9 = 37. 0 o. C T (in K) = T (in o. C) + 273. 15 T (in K) = 37. 0 o. C + 273. 15 = 310. 2

Density l Density = mass (g) / Volume (m. L or cm 3 or L) » Physical characteristic of a substance » Aids in identification of a substance » Calculated by. . . – divide the mass of a substance by the volume occupied by that mass – Units l l mass in grams volume » Solids and Liquids: m. L or cm 3 » Gasses: L

Density l Densities vary with temperature! » Why? ? l Would you expect densities to increase or decrease as the temperature increases?

Density l Immiscible liquids and solids separate into layers according to their densities » List the order from top to bottom when the following are mixed – Hg (13. 5525 g/m. L) – Carbon Tetrachloride (1. 59525 g/m. L) – Mg (1. 7425 g/m. L) – Water (1. 004 g/m. L) l What do the superscripts mean next to each density listed above?

Calculations Involving Density l Be able to calculate the density, mass, or volume of a substance » Use the plug and chug method or use density as a conversion factor l Practice makes perfect. .

Specific Gravity l Compares the density of a liquid or solid to that of water. . . Units? ? ? » Sp. Gravity = dsolid or liquid / dwater – Usually use dwater @ 4 o. C = 1. 000 g/m. L l Compares the density of a gas to that of air. . . Units? ? ? » Sp. Gravity = dgas/ dair

Mass vs. Weight l Mass » Amount of matter in an object » Independent of location » Measure with a balance by comparison with other known masses

Mass vs. Weight l Weight » Measures earth’s gravitational attraction on an object » Measure with a scale – measures force against a spring » Depends on – position relative to earth – motion of object w. r. t. the earth

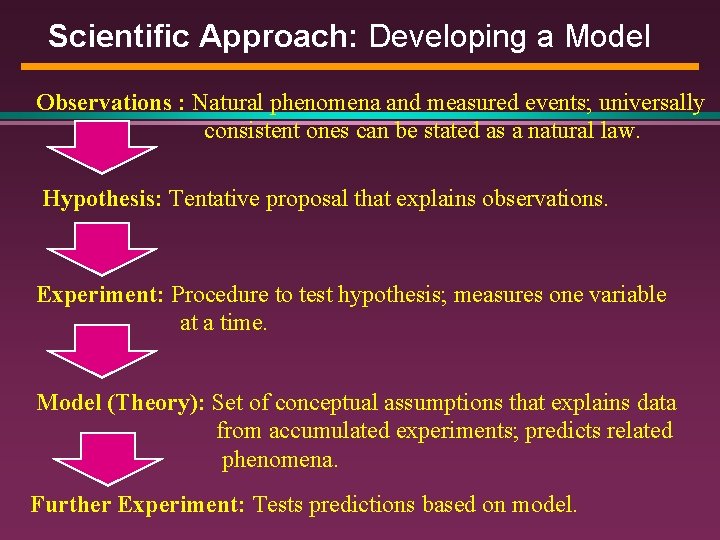
Scientific Approach: Developing a Model Observations : Natural phenomena and measured events; universally consistent ones can be stated as a natural law. Hypothesis: Tentative proposal that explains observations. Experiment: Procedure to test hypothesis; measures one variable at a time. Model (Theory): Set of conceptual assumptions that explains data from accumulated experiments; predicts related phenomena. Further Experiment: Tests predictions based on model.