CHAPTER 16 RATE OF A REACTION REACTION RATE














- Slides: 18

CHAPTER 16 RATE OF A REACTION

REACTION RATE The amount of time it takes to use up the reactants (decrease in reactant concentration) OR The amount of time it takes to make products (increase in product concentration)

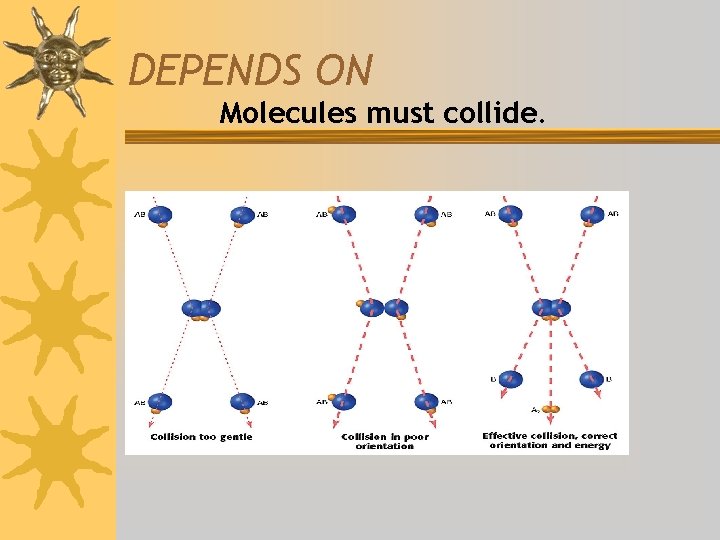
DEPENDS ON Molecules must collide.

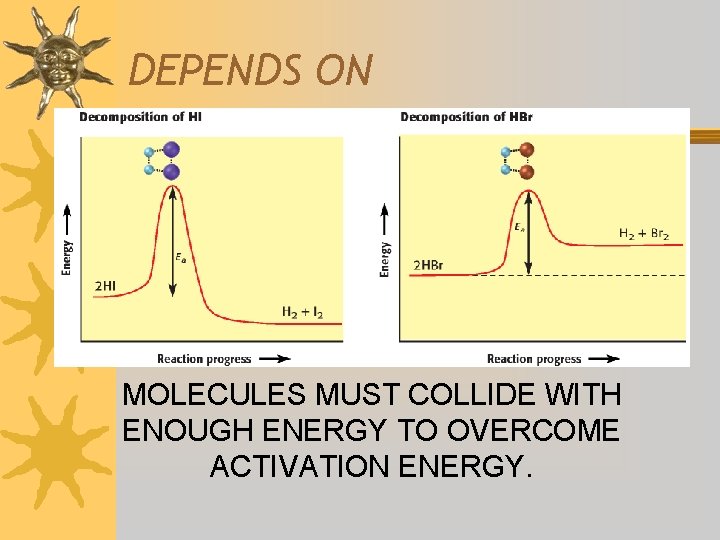
DEPENDS ON MOLECULES MUST COLLIDE WITH ENOUGH ENERGY TO OVERCOME ACTIVATION ENERGY.

Activation Energy and Chemical Reactions

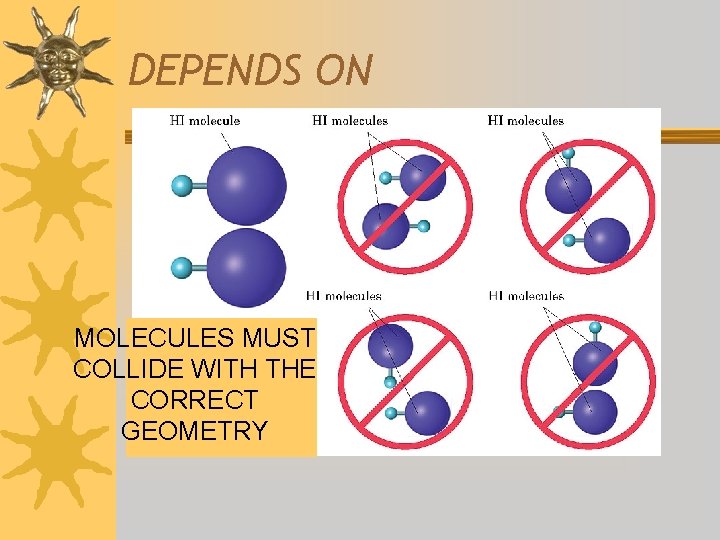
DEPENDS ON MOLECULES MUST COLLIDE WITH THE CORRECT GEOMETRY

FACTORS AFFECTING RATE

WAYS OF INCREASING RATE 1. Types of Reactants – certain bonds are easier to break than others 2. Surface Area – the more surface a molecule can touch → the faster the reaction 3. Temperature – the hotter the molecules → the more they move → the more they collide & change into product

WAYS OF INCREASING RATE 4. Concentration/Molarity – the higher the concentration → the more molecules there are → the more collisions take place creating product

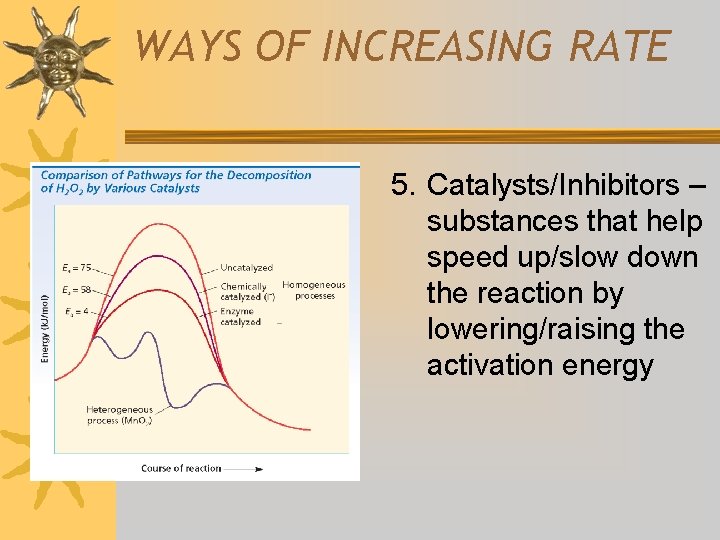
WAYS OF INCREASING RATE 5. Catalysts/Inhibitors – substances that help speed up/slow down the reaction by lowering/raising the activation energy

Multiple Choice 1. To be effective, a collision requires A. enough energy only. B. favorable orientation only. C. enough energy and a favorable orientation. D. a reaction mechanism.

Multiple Choice 1. To be effective, a collision requires A. enough energy only. B. favorable orientation only. C. enough energy and a favorable orientation. D. a reaction mechanism.

Multiple Choice 2. If a collision between molecules is very gentle, the molecules are A. more likely to be oriented favorably. B. less likely to be oriented favorably. C. likely to react. D. likely to rebound without reacting.

Multiple Choice 2. If a collision between molecules is very gentle, the molecules are A. more likely to be oriented favorably. B. less likely to be oriented favorably. C. likely to react. D. likely to rebound without reacting.

Multiple Choice 3. A species that changes the rate of a reaction but is neither consumed nor changed is A. a catalyst. B. an activated complex. C. an intermediate. D. a reactant.

Multiple Choice 3. A species that changes the rate of a reaction but is neither consumed nor changed is A. a catalyst. B. an activated complex. C. an intermediate. D a reactant.

Short Answer 4. Two molecules collide but bounce apart unchanged. What two reasons could account for their failure to react?

Short Answer 4. Two molecules collide but bounce apart unchanged. What two reasons could account for their failure to react? Answer: They had insufficient energy for bonds to break, or they did not collide in a favorable orientation.