Atmospheric Chemistry Measurements Organics in Air ATMS 360






















- Slides: 31

Atmospheric Chemistry Measurements – Organics in Air ATMS 360

Organic Chemistry - Carbon Compounds • Carbon - C, atomic number 6, molecular weight 12 • Electron configuration: 1 s 22 p 2 • Tetravalent, covalent bonds – 4 single bonds (sp 3); 2 double bonds (sp 2) one triple (sp) plus one single bond • Other atoms: hydrogen, oxygen, nitrogen, sulfur, halogens (Cl, F, Br)

Compounds • • • Alkanes – Cn. H 2 n+2 (CH 4, C 2 H 6, etc) Alkenes – Cn. H 2 n (C 2 H 4, etc) Alkynes - Cn. H 2 n-2 (C 2 H 2) Aromatic compounds (C 6 H 6, benzene) Polycyclic aromatic hydrocarbons - PAH (naphthalene, C 10 H 8)

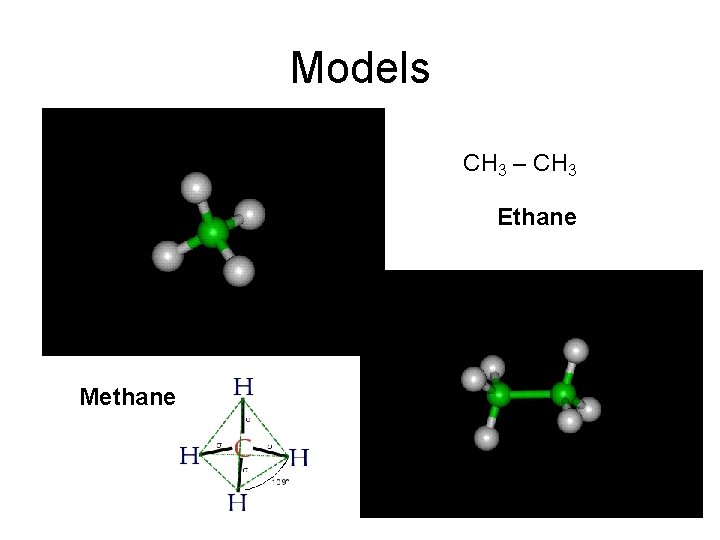
Models CH 3 – CH 3 Ethane Methane

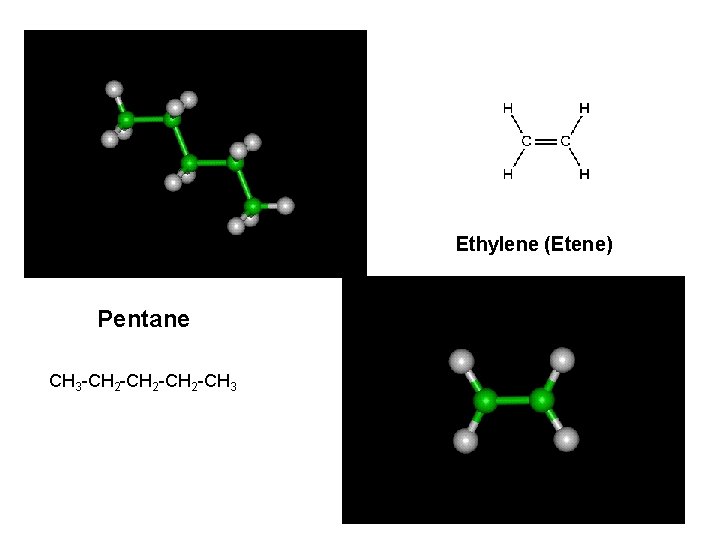
Ethylene (Etene) Pentane CH 3 -CH 2 -CH 3

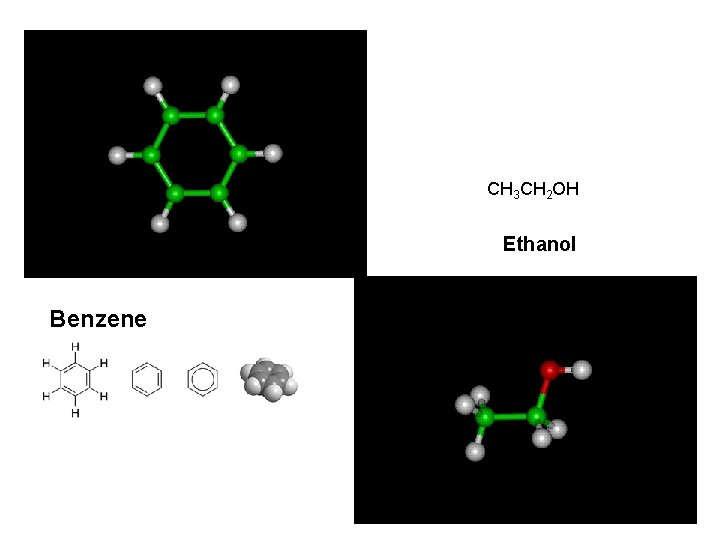
CH 3 CH 2 OH Ethanol Benzene

Sources of Organics in Air • Anthropogenic: – – Incomplete combustion of fossil fuels Biomass burning Industrial processes Cooking • Natural sources – Biogenic emissions (from vegetation) – Volcanic – Evaporation of sea spray • Atmospheric reaction products (from VOC, SVOC), secondary organic aerosol (SOA)

Fossil fuels Complete combustion: Cx. Hy + (x + y/4)O 2 e. g. C 5 H 12 + 8 O 2 x. CO 2 + y/2 H 2 O 5 CO 2 + 6 H 2 O Incomplete: CO, soot, organics and (in air) NOx

Biomass Burning Biomass: cellulose, hemicellulose, lignin, resins

Other sources - testing Residential wood Combustion Meat cooking

Secondary Organic Aerosol (SOA) • SOA processes are studied in photoreactors • European photoreactor (EUPHORE) in Valencia, Spain, is one of the largest (200 m 3) and the bestequipped outdoor simulation chamber in the world • We are studying atmospheric transformation of diesel emissions under the influence of sunlight, ozone, hydroxyl radicals that occur during transport in ambient air

Volatile, Semi-Volatile and Particulate Matter Organic Compounds (VOC, SVOC, PM) Vapor pressure ranges: VOC: > 102 Pa (10 -1 Torr) SVOC: 102 and 10 -6 Pa; (10 -1 and 10 -8 Torr) PM: < 10 -6 Pa (10 -8 Torr)

Organic Aerosol • Organic aerosols are solid or liquid particles suspended in the atmosphere containing organic carbon • Semi-volatile organic compounds (SVOC) distributed between gas and particle phases – reversibly condensable • Particle associated organics – complex mixture, incorporated into/onto particles; includes condensed SVOC and non-volatile organic compounds

Criteria Pollutants – National Ambient Air Quality Standards (NAAQS) • • Particulate Matter (PM) Ozone NOx SO 2 CO Lead (Pb) Ambient standards established by the US EPA and reviewed every 5 years

Why Particulate Matter? • Health effects: particulate matter (fine, PM 2. 5 and to lesser degree, coarse PM 10 -2. 5) has been associated with adverse health effects at low-to-moderate concentrations • NAAQS exist for PM (since 1971): – current (since 1997): PM 2. 5 annual 15 µg/m 3 and 24 -hr 65 µg/m 3; PM 10 annual 50 µg/m 3 and 24 -hr 150 µg/m 3 _ announced in September 2006: PM 2. 5 annual 15 µg/m 3 and 24 -hr 35 µg/m 3; PM 10 annual only • Climate change • Visibility problem (Haze Rule)

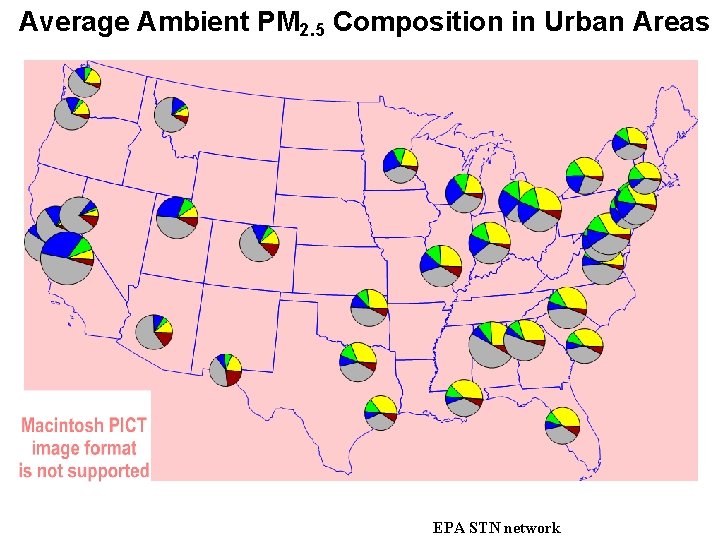
Average Ambient PM 2. 5 Composition in Urban Areas EPA STN network

Average PM 10 -2. 5, PM 2. 5, and PM 0. 1 composition at EPA “supersite” in Los Angeles, CA, 10/2001 to 9/2002 US EPA OAQPS PM Staff Paper, June 2005

Hazardous Air Pollutants (HAPs) • Full list – 188 compounds, most of them organics • The short list – 33 air toxics, most prevalent in urban area • No ambient standards – regulation of emissions from sources


Measurement Methods • Collection of VOC and aerosol samples followed by off-site laboratory analyses • VOC collection: stainless steel SUMMA canisters, Tedlar bags • PM and SVOC: Filters followed by solid adsorbents. Extraction with organic solvents in the laboratory

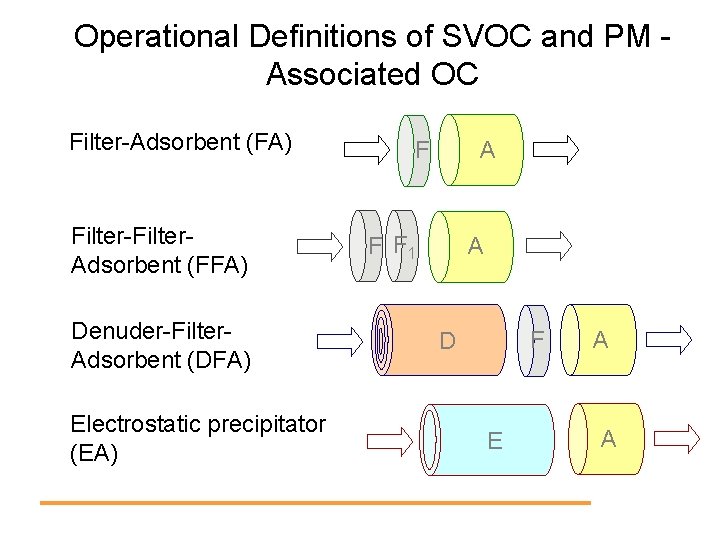
Operational Definitions of SVOC and PM Associated OC Filter-Adsorbent (FA) Filter-Filter. Adsorbent (FFA) Denuder-Filter. Adsorbent (DFA) Electrostatic precipitator (EA) F A F F 1 A F D E A A

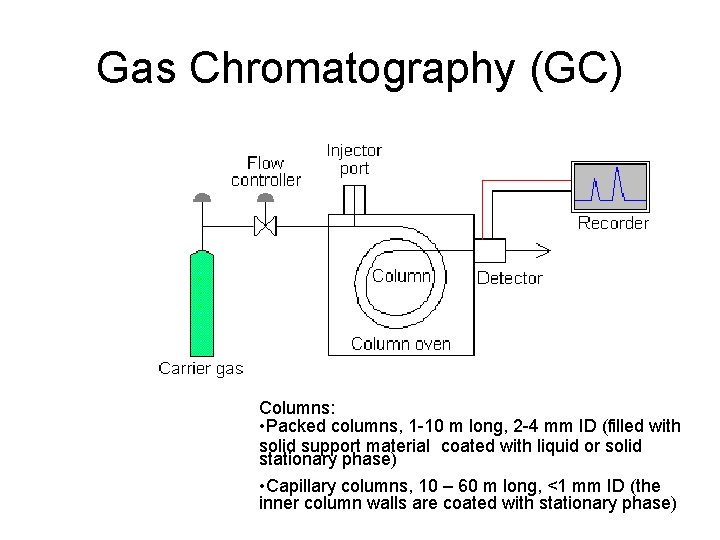
Analysis - Chromatography • Chromatography is a separation method that relies on differences in partitioning behavior between a flowing mobile phase and a stationary phase to separate the components in a mixture • Gas-liquid chromatography (GC) –mobile phase is gas (He, N 2, H 2) • Liquid chromatography (LC) – mobile phase is liquid. High performance liquid chromatography (HPLC) utilizes high-pressure pumps to increase the efficiency of the separation.

Gas Chromatography (GC) Columns: • Packed columns, 1 -10 m long, 2 -4 mm ID (filled with solid support material coated with liquid or solid stationary phase) • Capillary columns, 10 – 60 m long, <1 mm ID (the inner column walls are coated with stationary phase)

Detectors for GC and HPLC • Gas Chromatography detectors: – – – Flame Ionization (FID)- hydrocarbons Thermal Conductivity (TCD) - universal Electron Capture (ECD) – halogenated organics Photoionization (PID) - aromatics, olefins Fourier Transform Infrared (GC-FTIR) – all organics Mass Spectrometer (GC-MS) – any species • HPLC Detectors: – – UV-VIS absorption spectroscopy Photo diode-array UV-VIS Fluorescence MS (LC-MS)

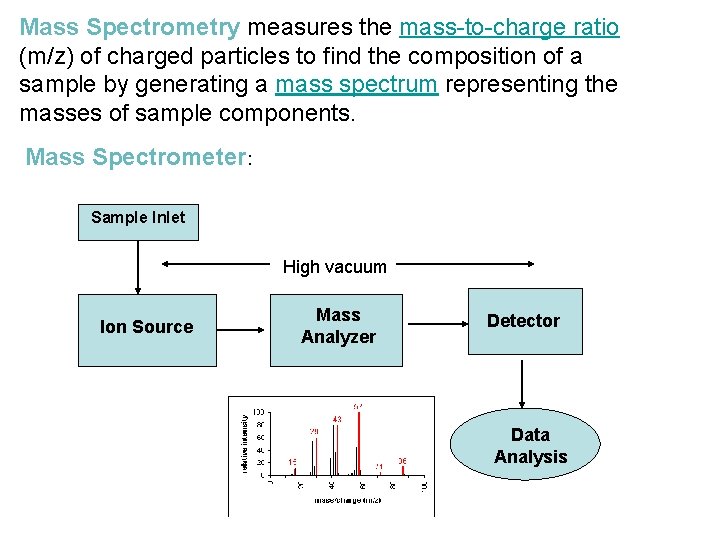
Mass Spectrometry measures the mass-to-charge ratio (m/z) of charged particles to find the composition of a sample by generating a mass spectrum representing the masses of sample components. Mass Spectrometer: Sample Inlet High vacuum Ion Source Mass Analyzer Detector Data Analysis

Ion Source • Ionization methods: Electron Impact (EI), Chemical Ionization (CI), Field Ionization (FI), Field Desorption (FD), Fast Atom Bombardment (FAB), Matrix-Assisted Laser Desorption/ Ionization (MALDI), Electrospray Ionization (ESI), and others. . • EI (unimolecular): bombarding neutral analyte M with high energy (70 e. V) electron beam M + e- M+· + 2 e • CI (bimolecular): M interacts with ions from reagent gas M + [BH]+ [M+H]+ + B (proton transfer) M + X+ [M+X]+ (electrophilic addition) M + X+ M+· + X (charge exchange) M + X +· [M-A]+ + AX (anion abstraction)

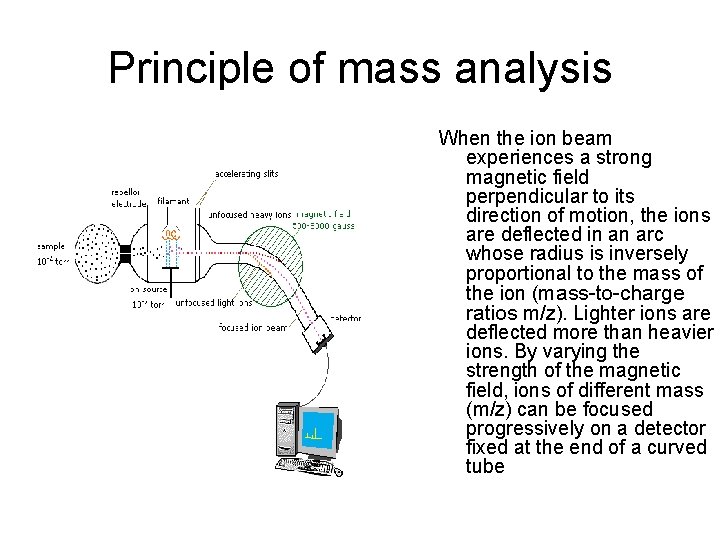
Principle of mass analysis When the ion beam experiences a strong magnetic field perpendicular to its direction of motion, the ions are deflected in an arc whose radius is inversely proportional to the mass of the ion (mass-to-charge ratios m/z). Lighter ions are deflected more than heavier ions. By varying the strength of the magnetic field, ions of different mass (m/z) can be focused progressively on a detector fixed at the end of a curved tube

Mass Analyzers • Types of mass analyzers: – magnetic sector (deflection of ion beam, separation by momentum); – linear quadrupole (4 rod electrodes, the pair of opposite rods are each held at the same potential composed of DC and AC component; a mass spectrum is obtained by monitoring the ions passing through the quadrupole filter as the voltages on the rods are varied); – quadrupole ion trap (three-dimensional RF quadrupole field to store ions within defined boundaries); – time-of-flight (TOF, uses the differences in transit time through a drift region to separate ions of different masses ).

Advances in Mass Spectrometry • "Aerosol -MS" is the measurement in real-time of the aerosol composition using a mass spectrometer. Almost always the particle size is measured simultaneously with the composition. Two approaches: – Single particle MS –Laser desorption-ionization MS. Example: Aerosol Time of Flight MS (ATOFMS, available commercially from TSI, Inc. ) – Thermal desorption aerosol MS. Example Aerodyne Aerosol MS, available commercially (http: //cires. colorado. edu/~jjose/ams. html#Info_Aerosol. MS).

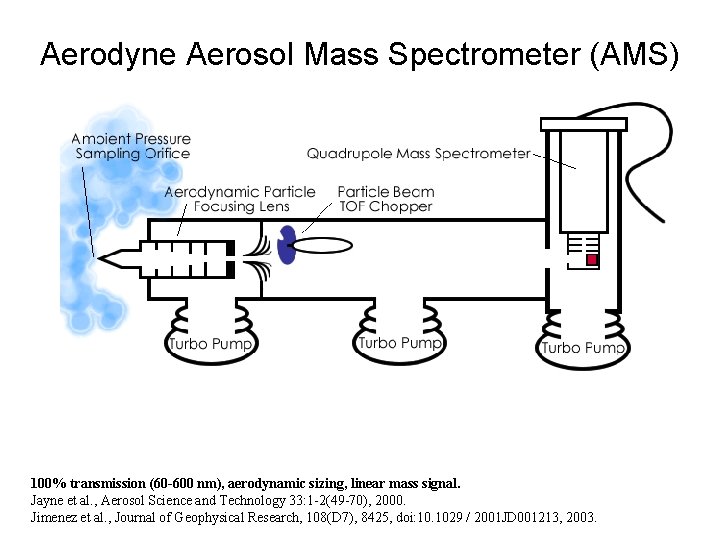
Aerodyne Aerosol Mass Spectrometer (AMS) Particle Beam Generation Aerodynamic Sizing Particle Composition Quadrupole Mass Spectrometer Chopper Thermal Vaporization & Electron Impact Ionization TOF Region Aerodynamic Lens (2 Torr) Particle Inlet (1 atm) Turbo Pump 100% transmission (60 -600 nm), aerodynamic sizing, linear mass signal. Jayne et al. , Aerosol Science and Technology 33: 1 -2(49 -70), 2000. Jimenez et al. , Journal of Geophysical Research, 108(D 7), 8425, doi: 10. 1029 / 2001 JD 001213, 2003.

Aerodyne Aerosol Mass Spectrometer (AMS) 100% transmission (60 -600 nm), aerodynamic sizing, linear mass signal. Jayne et al. , Aerosol Science and Technology 33: 1 -2(49 -70), 2000. Jimenez et al. , Journal of Geophysical Research, 108(D 7), 8425, doi: 10. 1029 / 2001 JD 001213, 2003.