Lab 5 Atmospheric Moisture Relative Humidity Sling Psychrometers

Lab 5: Atmospheric Moisture

Relative Humidity • Sling Psychrometers: measures Relative Humidity • Dry bulb temp • Web bulb temp DB – WB = Wet bulb depression DB vs. WB: Big difference = dry air Small difference = moist air

Relative Humidity • Swing for ~ 60 seconds • If you have a fraction, change to a whole number • Record DB & WB temps • Individuals or groups • 4 locations: • Inside • Outside

Measuring Relative Humidity Dry Bulb Temp Saturation Mixing Ratio (SMR) Based on DB temp; TABLE 2 Wet Bulb Temp Wet-Bulb Depression Relative Humidity (RH) Mixing Ratio (MR) DB – WB Based on DB temp & WBD; TABLE 4 SMR * RH (RH is a %!)

Atmospheric Moisture: Latent Heat • • • Water comes in three phases: – Solid – Liquid – Vapor • Unique to this atmospheric component *** Latent heat transfer: process of water changing phase – Energy is consumed or released – Sensible heat: heat we can feel & measure – Latent heat: energy in the form of heat Sensible heat: potential energy in the form of thermal energy or heat Latent heat: amount of energy in the form of heat released or absorbed by a chemical substance during a change of state. Kelvin is the most reliable measurement of temperature because zero kelvin represents the temperature at which all molecular motion stops.
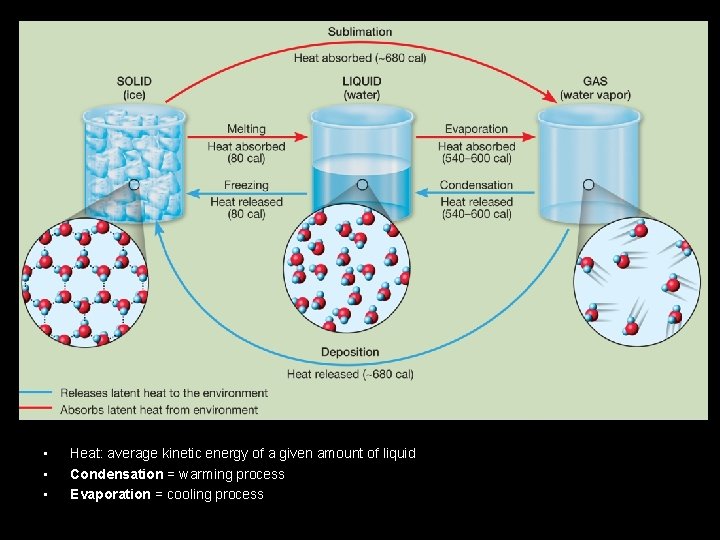
• • • Heat: average kinetic energy of a given amount of liquid Condensation = warming process Evaporation = cooling process

Atmospheric Moisture: Latent Heat Phase change Energy Examples Ice → (liquid) water Consumed Ice cubes melting in a glass; melting of falling snowflake Water → water vapor Consumed Clothes drying; evaporation off a water surface Ice → water vapor Consumed Solid air freshner; sublimation of snow/ice Water vapor → water Released Dew; condensation on a cold can of pop; contrail Water vapor → ice Released Frost on grass or on a window Water → ice Released Freezing ice cubes

• Amount of water vapor in the air depends on amount of energy available to change liquid → gas. – As temperature increases, the liquid water molecules start moving faster…it’s more likely to evaporate • Energy NOT used to boil the water is used to change the state of the water from liquid to gas

• Two ways to measure atmospheric moisture: 1. Vapor pressure – § § Water vapor molecules exert pressure proportional to their concentration in the atmosphere Maximum is called saturation vapor pressure • SVP increases with temperature

2. Mixing Ratio – – Mass of water vapor in the air – Ratio of water vapor mass to the mass of dry air • Units of grams of water vapor per kilogram of dry air – saturation mixing ratio: air is saturated • Depends on temperature (↑ with temperature) • Represents max weight of water vapor/kilogram of dry air

Relative Humidity: measures how close the air sample is to saturation • It is a ratio of actual water vapor in the air to the saturation level at a given temperature. • The greater the difference between air temperature & the dew point, the lower the RH (dry air) • RH is temperature dependant
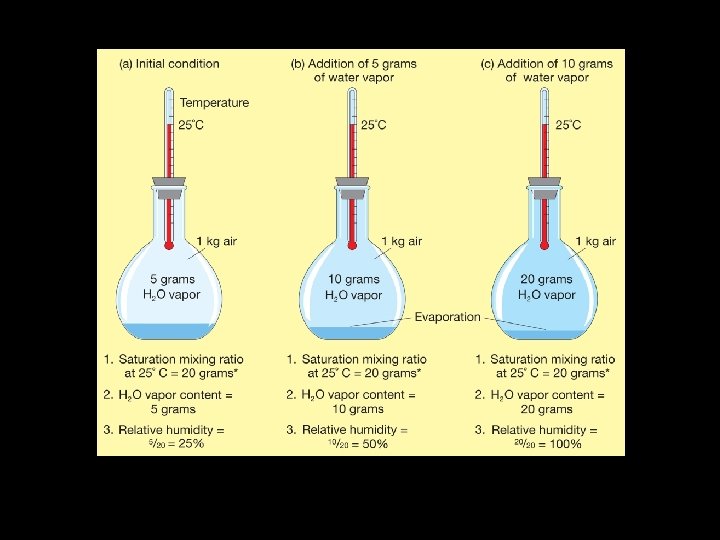

Winter? Warming up already dry air will DECREASE Relative Humidity! • Does winter indicate air that would be dry or moist? In the winter, air is generally rather dry. Cold air has less capacity to hold water vapor than warm air. • Heat in your home – becomes dry


Summer? • In the summer, air is generally moist. Warm air can hold greater amounts of water vapor than cold air. • Cooling already moist air will INCREASE Relative Humidity!

Dew Point • Dew point: temperature to which air must be cooled to reach saturation (RH = 100%) – Generally seen as dew in the morning on the grass.

Dew Point 1. Find MR of 17 2. Create a STRAIGHT line to the dew point line 3. Create another STRAIGHT line down to the temperature 4. Read the temperature. A 30° air sample would have to cool to ~ 22° to reach saturation (100% humidity)
- Slides: 17