A SEMISYNTHETIC STRATEGY TO FUCOSYLATED CHONDROITIN SULFATE POLYSACCHARIDES

![BIOLOGICAL ACTIVITIES f. CS may exhibit activities related to: • Coagulation and thrombosis[1] Pomin, BIOLOGICAL ACTIVITIES f. CS may exhibit activities related to: • Coagulation and thrombosis[1] Pomin,](https://slidetodoc.com/presentation_image_h2/208e6b3c521a4d818417d58bff01db24/image-3.jpg)







- Slides: 20

A SEMI-SYNTHETIC STRATEGY TO FUCOSYLATED CHONDROITIN SULFATE POLYSACCHARIDES FROM MICROBIAL-SOURCED CHONDROITIN Laezza Antonio 1, Iadonisi Alfonso 1, De Castro Cristina 2, De Rosa Mario 3, Schiraldi Chiara 3, Parrilli Michelangelo 4, Bedini Emiliano 1 1 Dipartimento di Scienze Chimiche, Università di Napoli Federico II, Complesso Universitario Monte S. Angelo, via Cintia 4, I-80126 Napoli, Italy 2 Dipartimento di Agraria, Università di Napoli Federico II, via Università 100, I-80055 Portici, Italy 3 Dipartimento di Medicina Sperimentale, Seconda Università di Napoli, via De Crecchio 7, I-80138 Napoli, Italy 4 Dipartimento di Biologia, Università di Napoli Federico II, Complesso Universitario Monte S. Angelo, via Cintia 4, I-80126 Napoli, Italy 4 th EPNOE International Polysaccharide Conference, Warsaw, 19 -22 Oct 2015

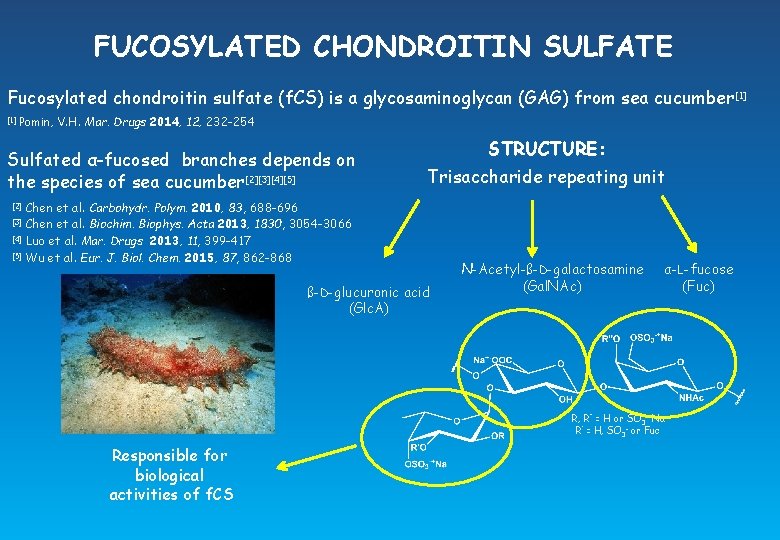
FUCOSYLATED CHONDROITIN SULFATE Fucosylated chondroitin sulfate (f. CS) is a glycosaminoglycan (GAG) from sea cucumber [1] Pomin, V. H. Mar. Drugs 2014, 12, 232 -254 Sulfated α-fucosed branches depends on the species of sea cucumber[2][3][4][5] [2] [3] [4] [5] STRUCTURE: Trisaccharide repeating unit Chen et al. Carbohydr. Polym. 2010, 83, 688 -696 Chen et al. Biochim. Biophys. Acta 2013, 1830, 3054 -3066 Luo et al. Mar. Drugs 2013, 11, 399 -417 Wu et al. Eur. J. Biol. Chem. 2015, 87, 862 -868 ß-D-glucuronic acid (Glc. A) N-Acetyl-ß-D-galactosamine (Gal. NAc) α-L-fucose (Fuc) R, R’’ = H or SO 3 -+Na R’ = H, SO 3 - or Fuc Responsible for biological activities of f. CS
![BIOLOGICAL ACTIVITIES f CS may exhibit activities related to Coagulation and thrombosis1 Pomin BIOLOGICAL ACTIVITIES f. CS may exhibit activities related to: • Coagulation and thrombosis[1] Pomin,](https://slidetodoc.com/presentation_image_h2/208e6b3c521a4d818417d58bff01db24/image-3.jpg)
BIOLOGICAL ACTIVITIES f. CS may exhibit activities related to: • Coagulation and thrombosis[1] Pomin, V. H. Mar. Drugs 2014, 12, 232 -254 • Atherosclerosis[6] Tovar et al. Altherosclerosis 1996, 26, 185 -195 • Cancer metastasis and inflammation[7] Borsig et al. J. Biol. Chem. 2007, 282, 14984 -14991 • Viral infection[8][9] [8] [9] Lian et al. Biochim. Biophys. Acta 2013, 1830, 4681 -4691 Huang et al. Carbohydr. Res. 2013, 380, 64 -69

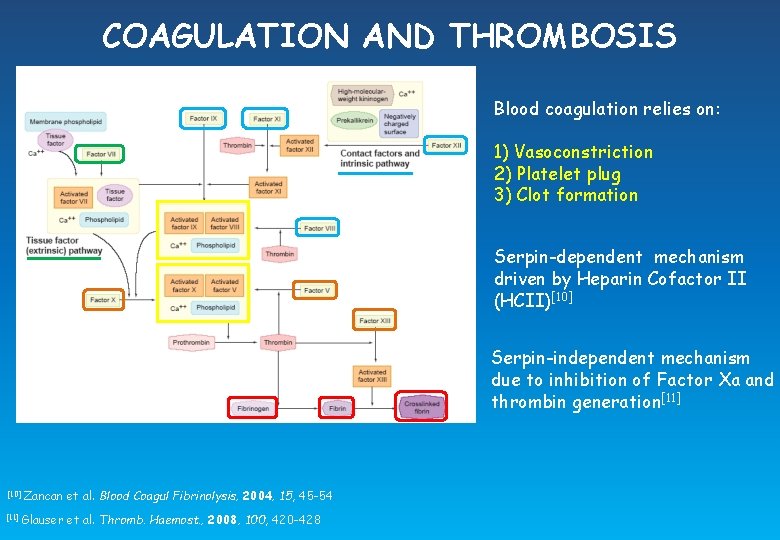
COAGULATION AND THROMBOSIS Blood coagulation relies on: 1) Vasoconstriction 2) Platelet plug 3) Clot formation Serpin-dependent mechanism driven by Heparin Cofactor II (HCII)[10] Serpin-independent mechanism due to inhibition of Factor Xa and thrombin generation[11] [10] [11] Zancan et al. Blood Coagul Fibrinolysis, 2004, 15, 45 -54 Glauser et al. Thromb. Haemost. , 2008, 100, 420 -428

CHONDROITIN DERIVATIZATION Escherichia coli O 5: K 4: H 4 Biotechnological production Polysaccharide purification Defructosylation IN COLLABORATION WITH THE RESEARCH GROUP OF PROF. M. DE ROSA AT THE SECOND UNIVERSITY OF NAPLES (SUN)[12] See OP 133 (E. Bedini) chondroitin purity grade: 89 -94% weight-averaged Mw: 45. 0 k. Da polydispersity: 1. 40 “A MICROBIOLOGICAL-CHEMICAL ACCESS TO CHONDROITIN SULFATE POLYSACCHARIDES” Bedini et al. Angew. Chem. Int. Ed. 2011, 50, 6160 -6163 [12] Cimini et al. Appl. Microbiol. Biotechnol 2010, 85, 1779 -1787

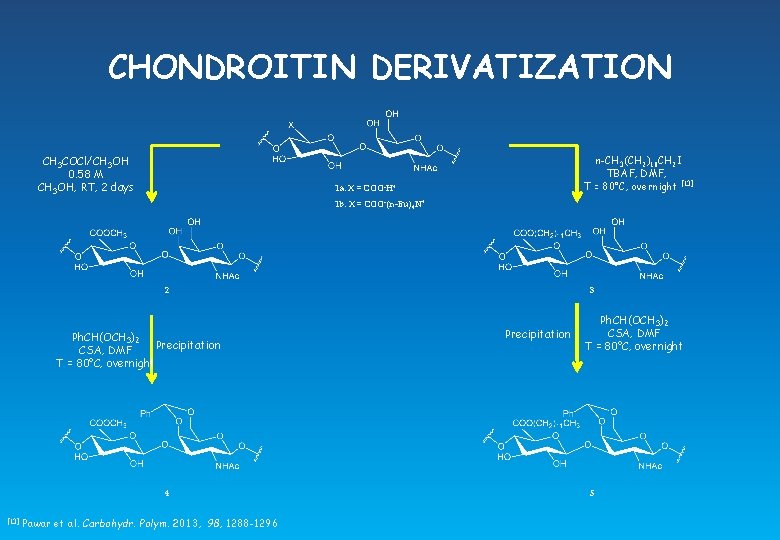
CHONDROITIN DERIVATIZATION CH 3 COCl/CH 3 OH 0. 58 M CH 3 OH, RT, 2 days n-CH 3(CH 2)10 CH 2 I TBAF, DMF, T = 80°C, overnight [13] 1 a: X = COO-H+ 1 b: X = COO-(n-Bu)4 N+ 2 Ph. CH(OCH 3)2 Precipitation CSA, DMF T = 80°C, overnight 4 [13] Pawar et al. Carbohydr. Polym. 2013, 98, 1288 -1296 3 Precipitation Ph. CH(OCH 3)2 CSA, DMF T = 80°C, overnight 5

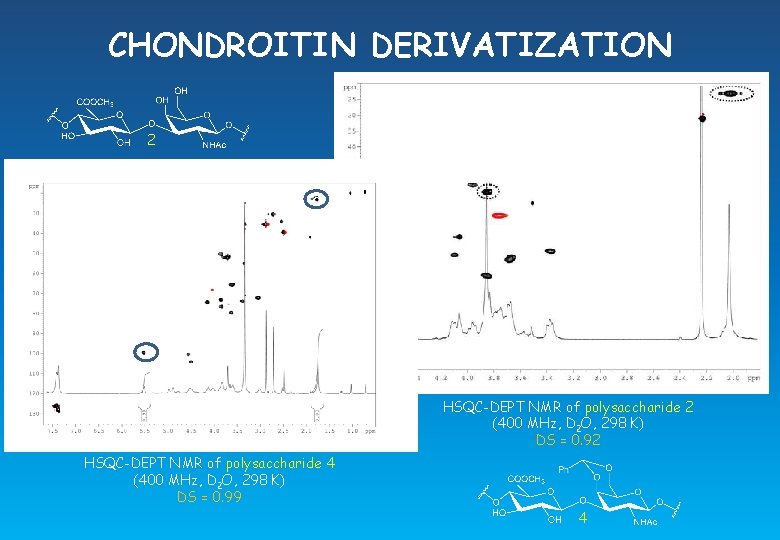
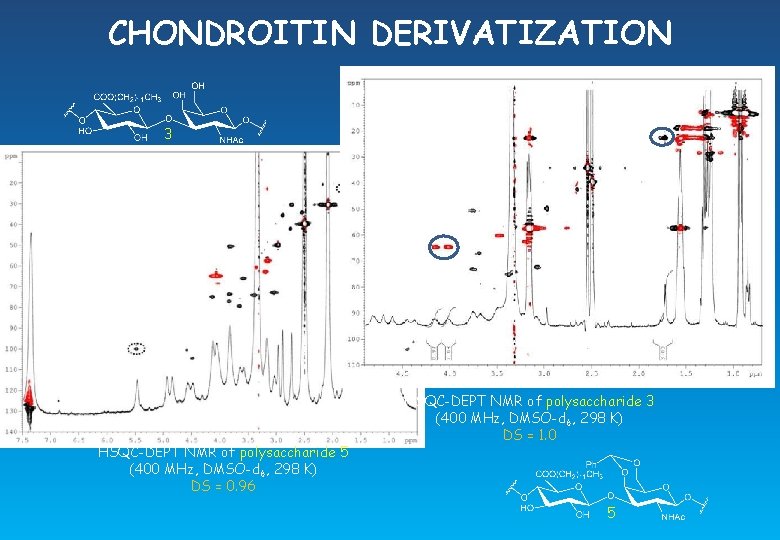
CHONDROITIN DERIVATIZATION 2 c HSQC-DEPT NMR of polysaccharide 2 (400 MHz, D 2 O, 298 K) DS = 0. 92 HSQC-DEPT NMR of polysaccharide 4 (400 MHz, D 2 O, 298 K) DS = 0. 99 4

CHONDROITIN DERIVATIZATION 3 HSQC-DEPT NMR of polysaccharide 5 (400 MHz, DMSO-d 6, 298 K) DS = 0. 96 HSQC-DEPT NMR of polysaccharide 3 (400 MHz, DMSO-d 6, 298 K) DS = 1. 0 5

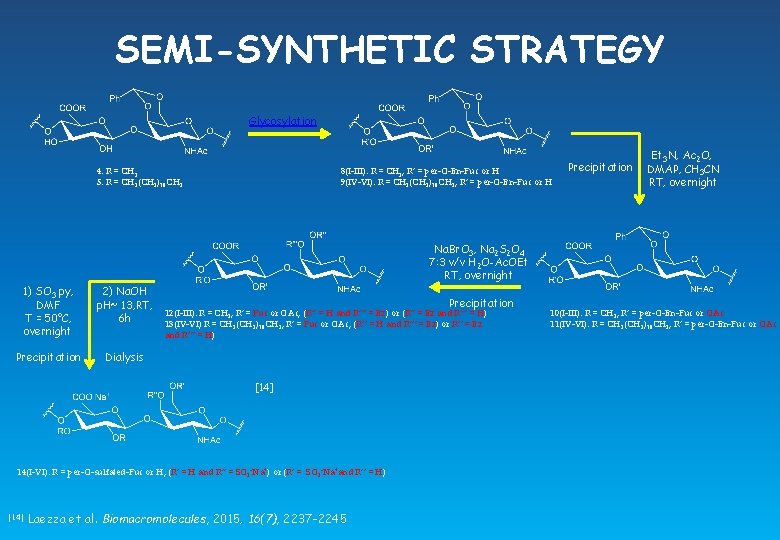
SEMI-SYNTHETIC STRATEGY Glycosylation 4: R = CH 3 5: R = CH 2(CH 2)10 CH 3 8(I-III): R = CH 3, R’ = per-O-Bn-Fuc or H 9(IV-VI): R = CH 2(CH 2)10 CH 3, R’ = per-O-Bn-Fuc or H Precipitation Et 3 N, Ac 2 O, DMAP, CH 3 CN RT, overnight Na. Br. O 3, Na 2 S 2 O 4 7: 3 v/v H 2 O-Ac. OEt RT, overnight 1) SO 3. py, DMF T = 50°C, overnight 2) Na. OH p. H~ 13, RT, 6 h Precipitation Dialysis Precipitation 12(I-III): R = CH 3, R’ = Fuc or OAc, (R’’ = H and R’’’ = Bz) or (R’’ = Bz and R’’’ = H) 13(IV-VI) R = CH 2(CH 2)10 CH 3, R’ = Fuc or OAc, (R’’ = H and R’’’ = Bz) or R’’ = Bz and R’’’ = H) [14] 14(I-VI): R = per-O-sulfated-Fuc or H, (R’ = H and R’’ = SO 3 -Na+) or (R’ = SO 3 -Na+and R’’ = H) [14] Laezza et al. Biomacromolecules, 2015, 16(7), 2237 -2245 10(I-III): R = CH 3, R’ = per-O-Bn-Fuc or OAc 11(IV-VI): R = CH 2(CH 2)10 CH 3, R’ = per-O-Bn-Fuc or OAc

SEMI-SYNTHETIC STRATEGY 14(II) 14(I) and 14(III) HSQC-DEPT NMR of polysaccharide 14(I) (600 MHz, D 2 O, 298 K) HSQC-DEPT NMR of polysaccharide 14(III) (600 MHz, D 2 O, 298 K)

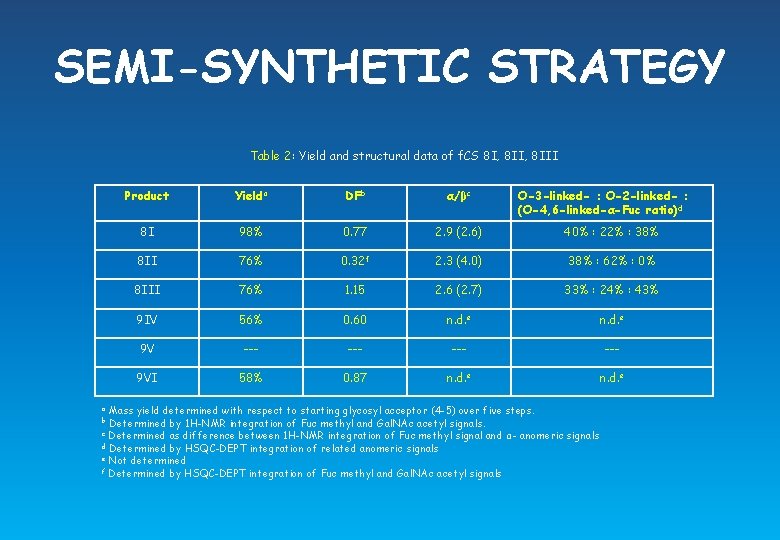
SEMI-SYNTHETIC STRATEGY Table 2: Yield and structural data of f. CS 8 I, 8 III Product Yielda DFb α/βc O-3 -linked- : O-2 -linked- : (O-4, 6 -linked-α-Fuc ratio)d 8 I 98% 0. 77 2. 9 (2. 6) 40% : 22% : 38% 8 II 76% 0. 32 f 2. 3 (4. 0) 38% : 62% : 0% 8 III 76% 1. 15 2. 6 (2. 7) 33% : 24% : 43% 9 IV 56% 0. 60 n. d. e 9 V --- --- 9 VI 58% 0. 87 n. d. e Mass yield determined with respect to starting glycosyl acceptor (4 -5) over five steps. Determined by 1 H-NMR integration of Fuc methyl and Gal. NAc acetyl signals. c Determined as difference between 1 H-NMR integration of Fuc methyl signal and α- anomeric signals d Determined by HSQC-DEPT integration of related anomeric signals e Not determined f Determined by HSQC-DEPT integration of Fuc methyl and Gal. NAc acetyl signals a b

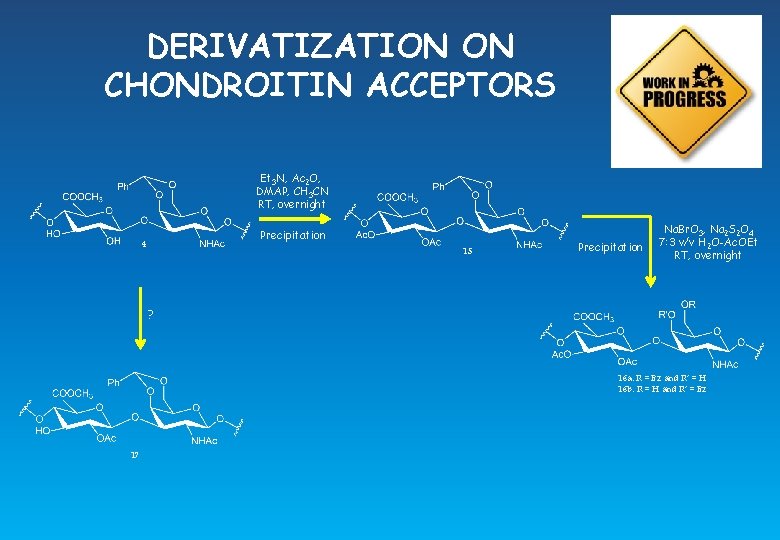
DERIVATIZATION ON CHONDROITIN ACCEPTORS Et 3 N, Ac 2 O, DMAP, CH 3 CN RT, overnight Precipitation 4 15 Precipitation Na. Br. O 3, Na 2 S 2 O 4 7: 3 v/v H 2 O-Ac. OEt RT, overnight ? 16 a: R = Bz and R’ = H 16 b: R = H and R’ = Bz 17

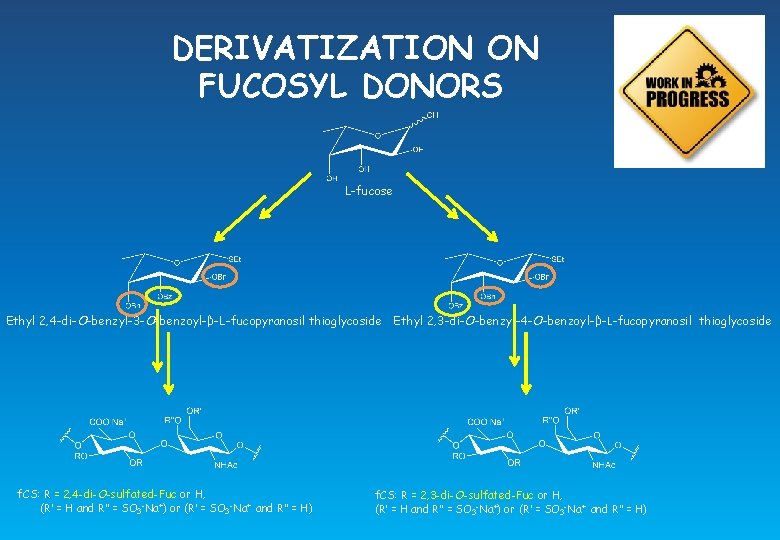
DERIVATIZATION ON FUCOSYL DONORS L-fucose Ethyl 2, 4 -di-O-benzyl-3 -O-benzoyl-β-L-fucopyranosil thioglycoside Ethyl 2, 3 -di-O-benzyl-4 -O-benzoyl-β-L-fucopyranosil thioglycoside f. CS: R = 2, 4 -di-O-sulfated-Fuc or H, (R’ = H and R’’ = SO 3 -Na+) or (R’ = SO 3 -Na+ and R’’ = H) f. CS: R = 2, 3 -di-O-sulfated-Fuc or H, (R’ = H and R’’ = SO 3 -Na+) or (R’ = SO 3 -Na+ and R’’ = H)

CONCLUSIONS First seven-step semi-synthetic strategy from microbial source chondroitin to fucosylated chondroitin sulfate First O-glycosylation of secondary hydroxyls of polysaccharides • HIGH GLOBAL YIELD • CHEAPNESS OF THE USED REAGENTS • RESEMBLANCE WITH NATURAL f. CS

FUTURE PERSPECTIVES Different protection patterns on Fuc donors Different protection patterns on chondroitin intermediates Synthesis of a library of f. CS polysaccharides Anticoagulant tests on f. CS polysaccharides

ACKNOWLEDGMENT University of Naples “Federico II” - Prof. M. Parrilli - Dr. E. Bedini -Prof. A. Iadonisi -Prof. C. De Castro Second University of Naples (SUN) - Prof. M. De Rosa - Prof. C. Schiraldi MIUR L. 297 project “Produzione biotecnologica di condroitina” Bio. Tek. Net


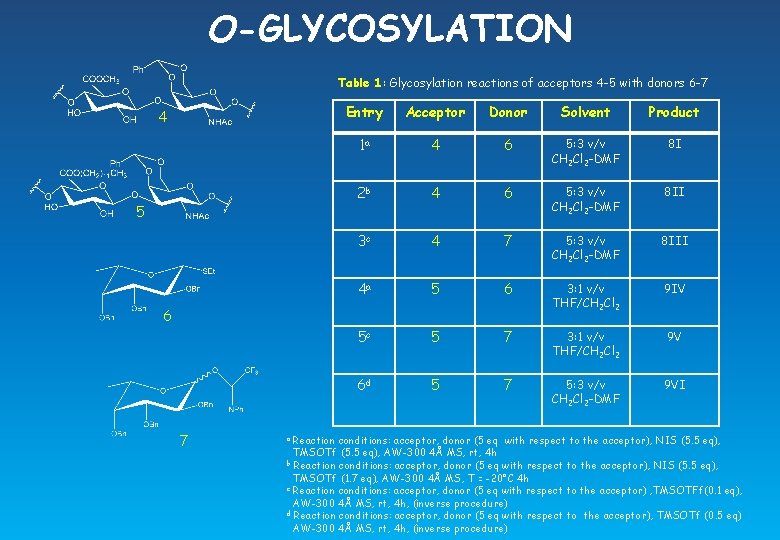
O-GLYCOSYLATION Table 1: Glycosylation reactions of acceptors 4 -5 with donors 6 -7 4 5 Entry Acceptor Donor Solvent Product 1 a 4 6 5: 3 v/v CH 2 Cl 2 -DMF 8 I 2 b 4 6 5: 3 v/v CH 2 Cl 2 -DMF 8 II 3 c 4 7 5: 3 v/v CH 2 Cl 2 -DMF 8 III 4 a 5 6 3: 1 v/v THF/CH 2 Cl 2 9 IV 5 c 5 7 3: 1 v/v THF/CH 2 Cl 2 9 V 6 d 5 7 5: 3 v/v CH 2 Cl 2 -DMF 9 VI 6 7 Reaction conditions: acceptor, donor (5 eq with respect to the acceptor), NIS (5. 5 eq), TMSOTf (5. 5 eq), AW-300 4Å MS, rt, 4 h b Reaction conditions: acceptor, donor (5 eq with respect to the acceptor), NIS (5. 5 eq), TMSOTf (1. 7 eq), AW-300 4Å MS, T = -20°C 4 h c Reaction conditions: acceptor, donor (5 eq with respect to the acceptor) , TMSOTFf(0. 1 eq), AW-300 4Å MS, rt, 4 h, (inverse procedure) d Reaction conditions: acceptor, donor (5 eq with respect to the acceptor), TMSOTf (0. 5 eq) AW-300 4Å MS, rt, 4 h, (inverse procedure) a

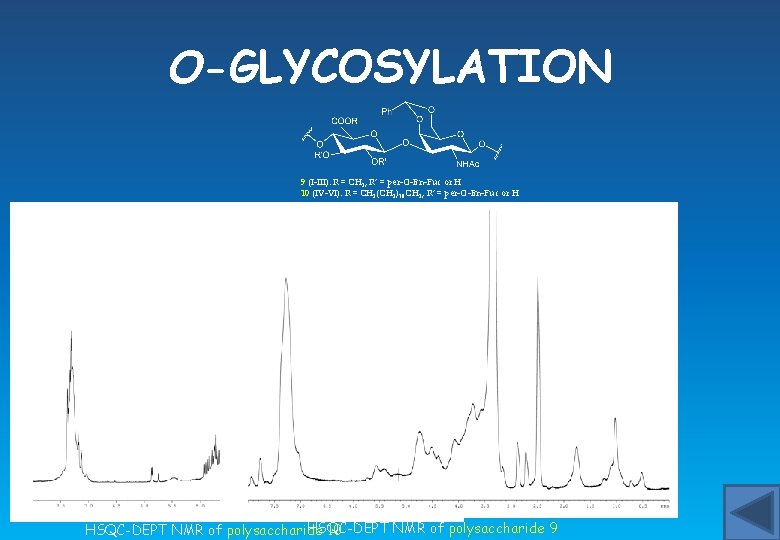
O-GLYCOSYLATION 9 (I-III): R = CH 3, R’ = per-O-Bn-Fuc or H 10 (IV-VI): R = CH 2(CH 2)10 CH 3, R’ = per-O-Bn-Fuc or H HSQC-DEPT NMR of polysaccharide 9 HSQC-DEPT NMR of polysaccharide 10
