The Components of Matter 2 1 The Components






































- Slides: 59

The Components of Matter 2 -1

The Components of Matter • Elements, Compounds, and Mixtures • The Observations That Led to an Atomic View of Matter • Dalton’s Atomic Theory • The Observations That Led to the Nuclear Atom Model • The Atomic Theory Today • Elements: A First Look at the Periodic Table • Compounds: Introduction to Bonding • Formulas, Names, and Masses of Compounds • Mixtures: Classification and Separation 2 -2

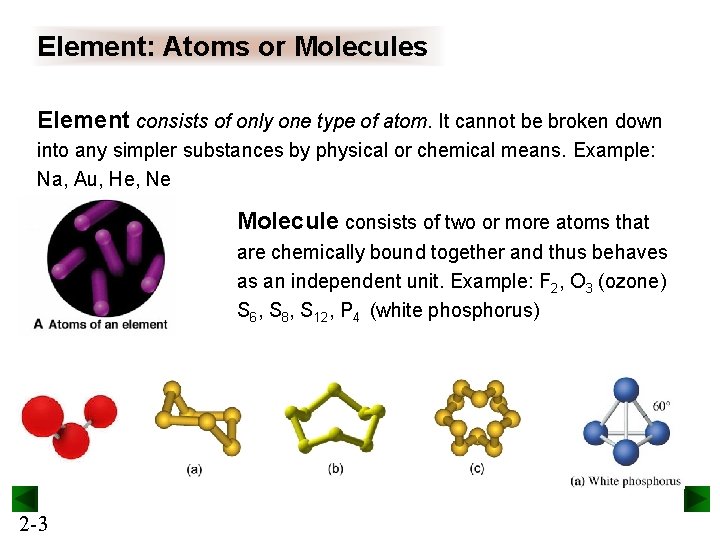
Element: Atoms or Molecules Element consists of only one type of atom. It cannot be broken down into any simpler substances by physical or chemical means. Example: Na, Au, He, Ne Molecule consists of two or more atoms that are chemically bound together and thus behaves as an independent unit. Example: F 2, O 3 (ozone) S 6, S 8, S 12, P 4 (white phosphorus) 2 -3

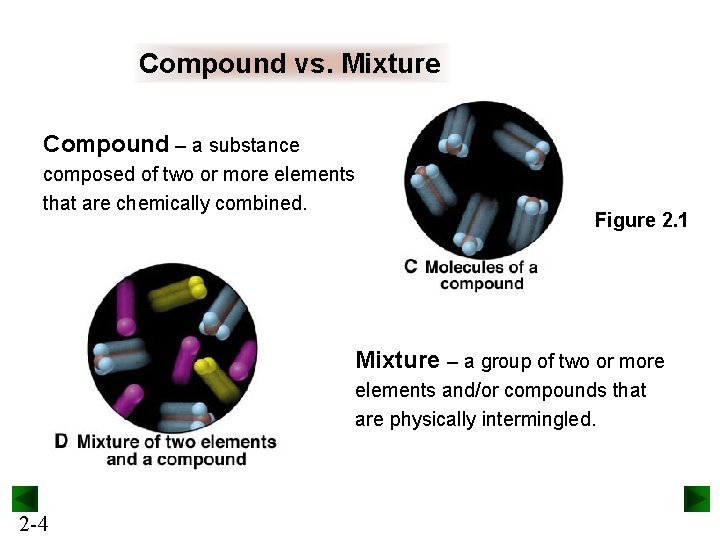
Compound vs. Mixture Compound – a substance composed of two or more elements that are chemically combined. Figure 2. 1 Mixture – a group of two or more elements and/or compounds that are physically intermingled. 2 -4

Law of Multiple Proportions If elements A and B react to form two compounds, the different masses of B that combine with a fixed mass of A can be expressed as a ratio of small whole numbers. Example: Carbon Oxides A & B Carbon Oxide I : 57. 1% oxygen and 42. 9% carbon Carbon Oxide II : 72. 7% oxygen and 27. 3% carbon 2 -5

Assume that you have 100 g of each compound. Carbon Oxide I g oxygen/100 g compound 57. 1 72. 7 g carbon/100 g compound 27. 3 72. 7 = 2. 66 27. 3 g oxygen/g carbon 42. 9 57. 1 = 1. 33 42. 9 2. 66 g O/g C in II 1. 33 g O/g C in I 2 -6 Carbon Oxide II = 2 1

Dalton’s Atomic Theory Dalton postulated that: 1. 2. 3. 4. 2 -7 All matter consists of atoms; tiny indivisible particles of an element that cannot be created or destroyed. Atoms of one element cannot be converted into atoms of another element. Atoms of an element are identical in mass and other properties and are different from the atoms of any other element. Compounds result from the chemical combination of a specific ratio of atoms of different elements.

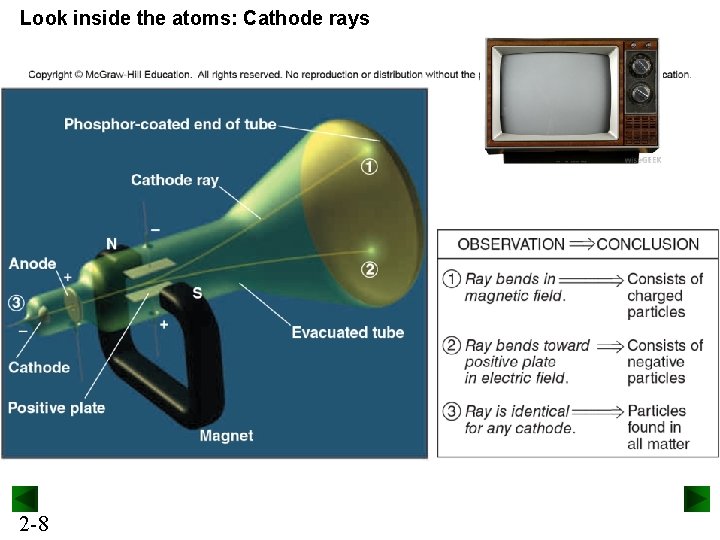
Look inside the atoms: Cathode rays 2 -8

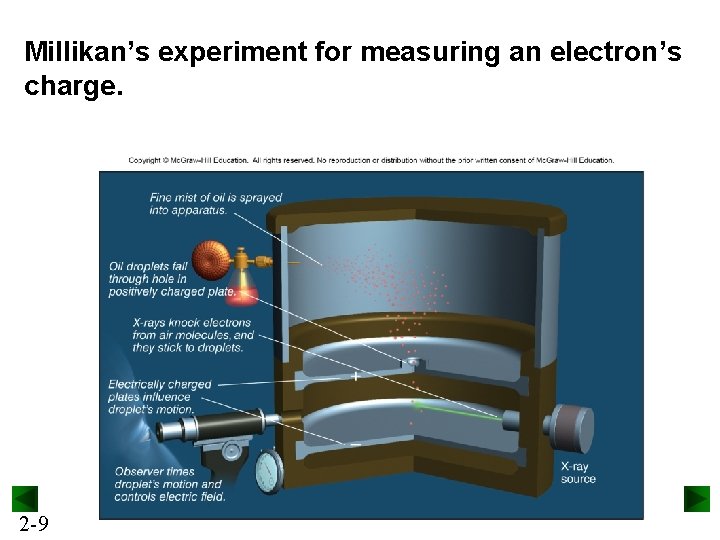
Millikan’s experiment for measuring an electron’s charge. 2 -9

Millikan’s findings were used to calculate the mass of an electron. determined by J. J. Thomson and others mass of electron = mass charge x charge = (– 5. 686 x 10– 12 kg/C) x (– 1. 602 x 10– 19 C) = 9. 109 x 10– 31 kg = 9. 109 x 10– 28 g 2 -10

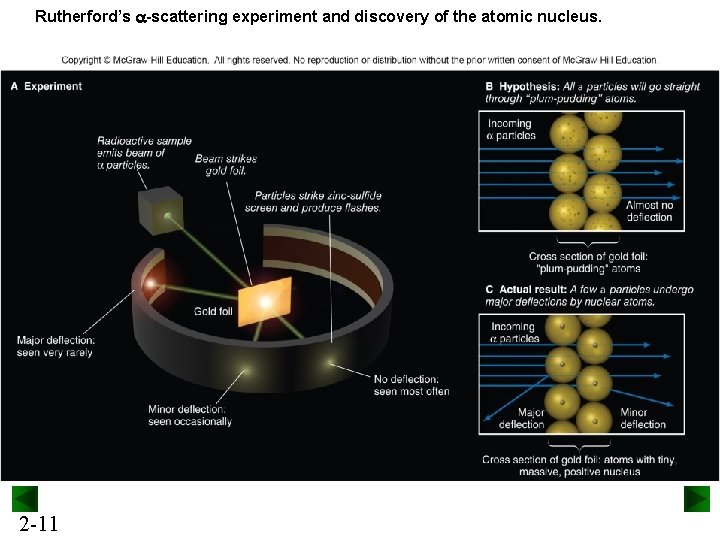
Rutherford’s a-scattering experiment and discovery of the atomic nucleus. 2 -11

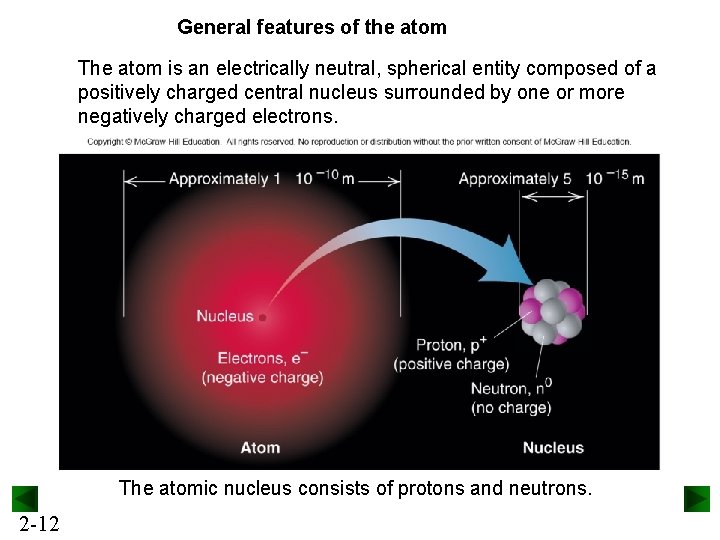
General features of the atom The atom is an electrically neutral, spherical entity composed of a positively charged central nucleus surrounded by one or more negatively charged electrons. The atomic nucleus consists of protons and neutrons. 2 -12

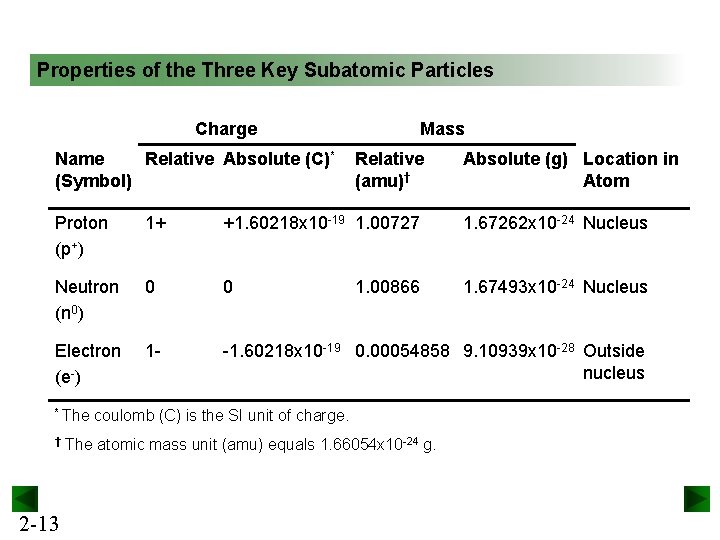
Properties of the Three Key Subatomic Particles Charge Name Relative Absolute (C)* (Symbol) Mass Relative (amu)† Absolute (g) Location in Atom Proton (p+) 1+ +1. 60218 x 10 -19 1. 00727 1. 67262 x 10 -24 Nucleus Neutron (n 0) 0 0 1. 67493 x 10 -24 Nucleus Electron (e-) 1 - -1. 60218 x 10 -19 0. 00054858 9. 10939 x 10 -28 Outside nucleus * The † 2 -13 1. 00866 coulomb (C) is the SI unit of charge. The atomic mass unit (amu) equals 1. 66054 x 10 -24 g.

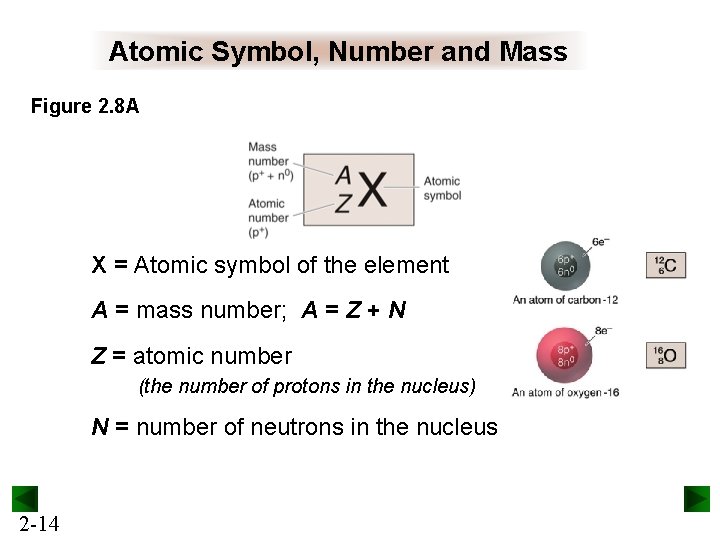
Atomic Symbol, Number and Mass Figure 2. 8 A X = Atomic symbol of the element A = mass number; A = Z + N Z = atomic number (the number of protons in the nucleus) N = number of neutrons in the nucleus 2 -14

Isotopes are atoms of an element with the same number of protons, but a different number of neutrons. Isotopes have the same atomic number, but a different mass number. Figure 2. 8 B 2 -15

Getting familiar with Isotope Symbol PROBLEM: Silicon (Si) has three naturally occurring isotopes: 28 Si, 29 Si, and 30 Si. Determine the number of protons, neutrons, and electrons in 29 Si 2 -16 has 14 p+, 14 e–, and 15 n 0 (29 – 14 = 15)

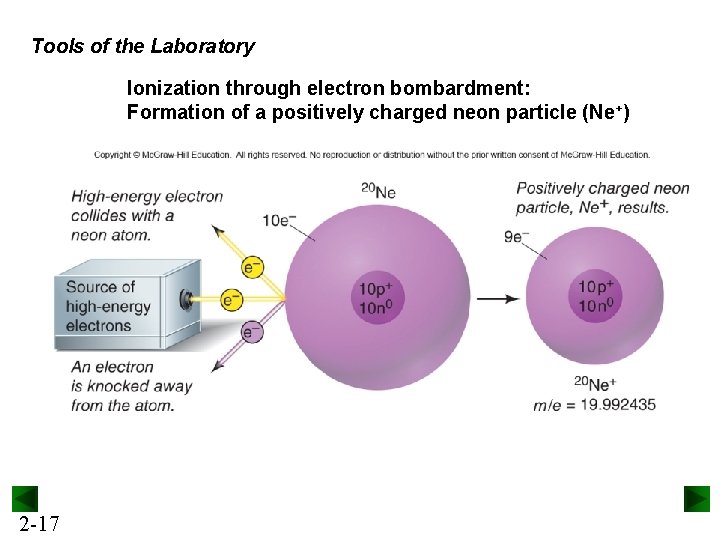
Tools of the Laboratory Ionization through electron bombardment: Formation of a positively charged neon particle (Ne+) 2 -17

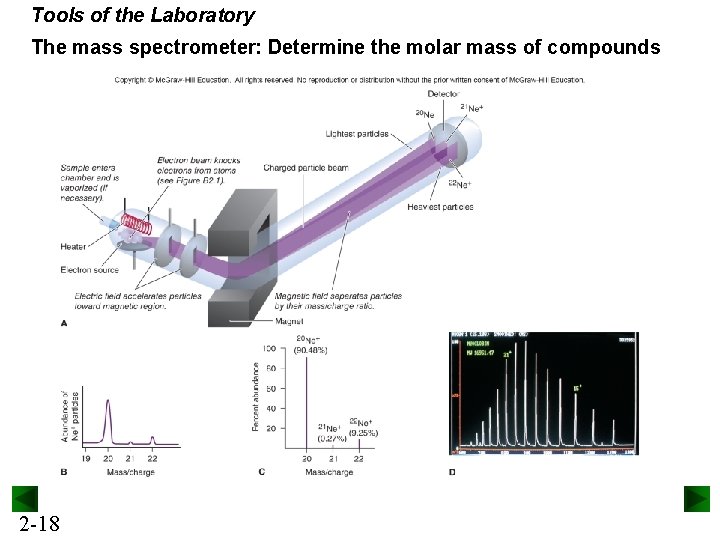
Tools of the Laboratory The mass spectrometer: Determine the molar mass of compounds 2 -18

Abundance of Isotopes 11 B 10 B • Abundance: Percentage of certain isotope among all the isotopes. Example: 10 B 19. 9% vs. 11 B 80. 1% Because of different mass among isotopes, atomic mass is the weighted average among all isotopes. • Average mass = ATOMIC WEIGHT • Atomic mass = S abundance mass number =abundance 1 x #mass 1 + abundance 2 x #mass 2 + … 2 -19

Calculating the Atomic Mass of an Element PROBLEM: Silver (Ag, Z = 47) has two naturally occurring isotopes, 107 Ag and 109 Ag. From the mass spectrometric data provided, calculate the atomic mass of Ag. Isotope 107 Ag 109 Ag Mass (amu) 106. 90509 108. 90476 Abundance (%) 51. 84 48. 16 = 107. 87 amu 2 -20

Practice: Isotopes & Atomic Weight Assuming the mass of isotope equals the mass number in amu: • 6 Li = 7. 5% abundant and 7 Li = – Atomic weight of Li = _______ • 28 Si = 92. 23%, 29 Si = 4. 67%, 30 Si = 3. 10% – Atomic weight of Si = _______ 2 -21 6. 925 28. 11

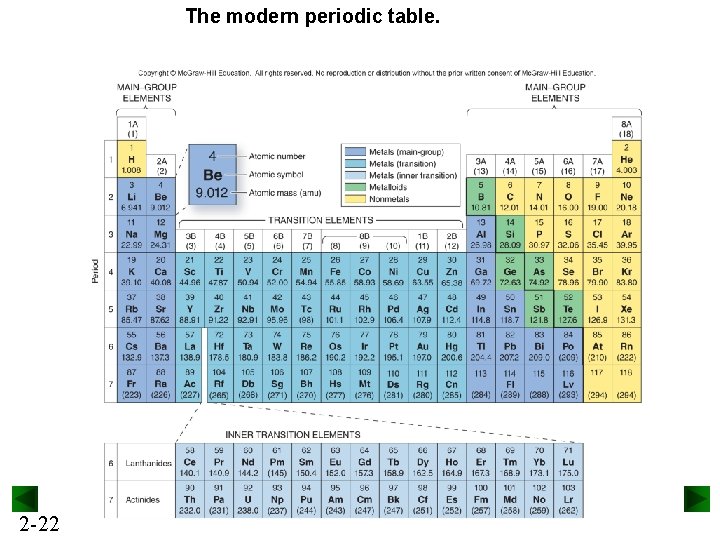
The modern periodic table. 2 -22

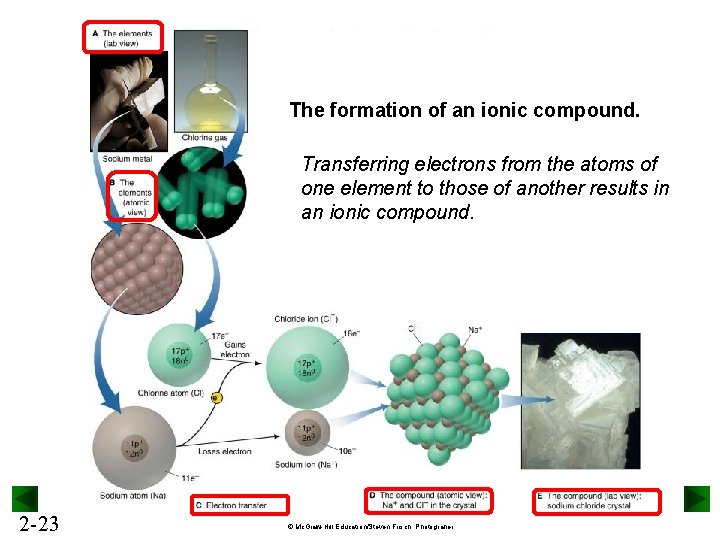
The formation of an ionic compound. Transferring electrons from the atoms of one element to those of another results in an ionic compound. 2 -23 © Mc. Graw-Hill Education/Steven Frisch, Photograher

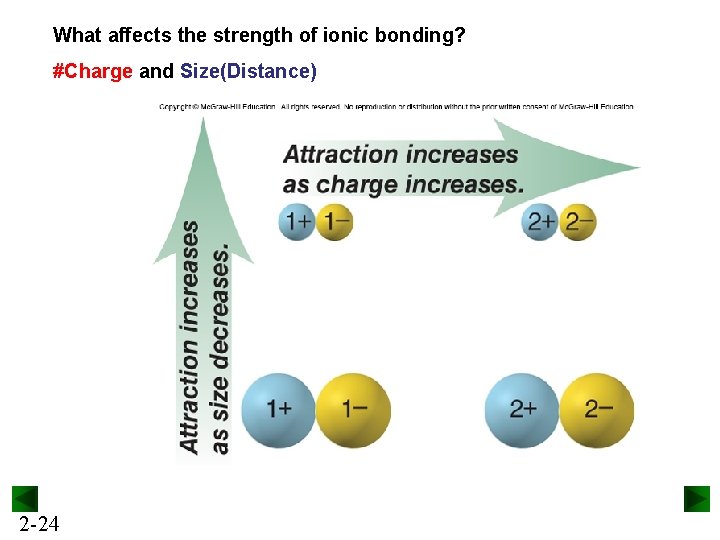
What affects the strength of ionic bonding? #Charge and Size(Distance) 2 -24

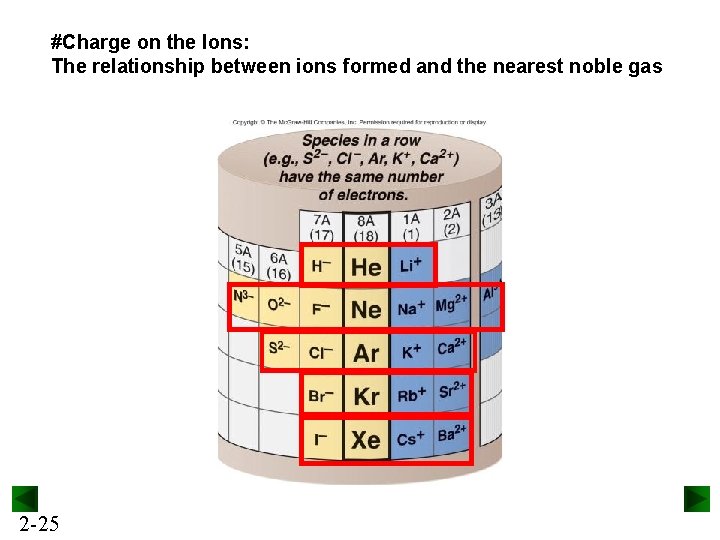
#Charge on the Ions: The relationship between ions formed and the nearest noble gas 2 -25

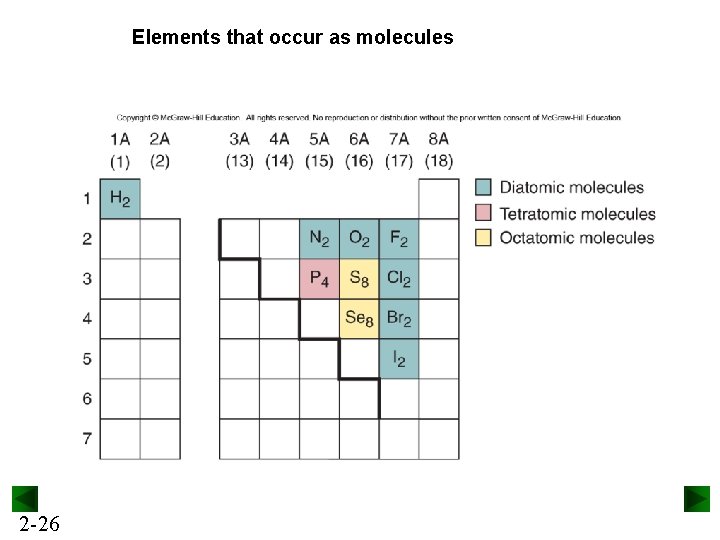
Elements that occur as molecules 2 -26

Chemical Formulas • A chemical formula consists of – element symbols with numerical subscripts. • The chemical formula indicates the – type and number of each atom present in the smallest unit of a substance. – Example: One unit of (NH 4)3 PO 4 = 3 N atoms + 12 H atoms + 1 P atom + 4 O atoms 2 -27

Binary Molecular Compounds: Two Nonmetals (such as CO 2) 1. Name first element in formula first – use the full name of the element 2. Name the second element in the formula with an -ide – as if it were an anion, however, remember these compounds do not contain ions! 3. Use a prefix in front of each name to indicate the number of atoms a) Never use the prefix mono- on the first element 2 -28 28

Subscript - Prefixes • 1 = mono-; – not used on first nonmetal • 2 = di • 3 = tri • 4 = tetra- 2 -29 • • • 5 = penta 6 = hexa 7 = hepta 8 = octadrop last “a” if name begins with vowel 29

Practice: Naming Molecular Compounds • • 2 -30 CO Cl. O 3 SO 2 P 2 O 5 N 2 O 4 IF 7 SF 6 30

Ionic Compounds • Made of Cation (+) and Anion (-) • Name: Cation Anion example: Na. Cl Sodium Chloride – Cation: • Type I metal • Type II metal • Polyatomic ion: ammonium NH 4+ – Anion: • Nonmetal: Chloride Cl-, Oxide O 2 • Polyatomic ion: SO 42 - , OH- , NO 32 -31 31

Metal Cations: Type I (Groups IA, IIA, AZA) – only have one possible charge • Groups IA, IIA, Ag+, Zn 2+, Al 3+ – Charge by position on the Periodic Table • IA = +1, IIA = +2, • Ag+ (IB), Zn 2+(IIB) Al 3+(IIIA) How do you know a metal cation is Type II? its not Type I !!! 2 -32 32

Metal Cations: Type II: Metal ions that are other than Type I Common Examples: Fe 2+/3+, Cu+/2+, Cr 3+/6+, Mn 2+/4+, Pb 2+/4+, Sn 2+/4+, etc ) – have more than one possible charge – determine charge by charge on anion 2 -33 How do you know a metal cation is Type II? its not Type I !!! 33

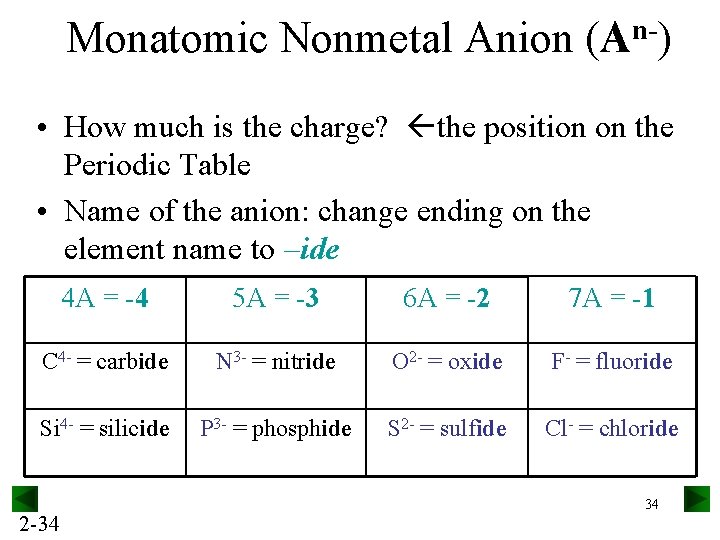
Monatomic Nonmetal Anion n(A ) • How much is the charge? the position on the Periodic Table • Name of the anion: change ending on the element name to –ide 4 A = -4 5 A = -3 6 A = -2 7 A = -1 C 4 - = carbide N 3 - = nitride O 2 - = oxide F- = fluoride Si 4 - = silicide P 3 - = phosphide S 2 - = sulfide Cl- = chloride 2 -34 34

Type I Binary Ionic Compounds Binary: only two kinds of ions in one UNIT Example: Mg. O, Ca. Cl 2 • Metal listed first in formula & name 1. Metal Cation Nonmetal Anion 2. Cation name <= Metal name: Magnesium, Calcium 3. Nonmetal anion <= Nonmetal name ends with –ide: Oxide, Chloride 2 -35 35

Type II Binary Ionic Compounds Metal listed first in formula & name 1. Metal cation Nonmetal anion 2. metal cation Metal(Roman Numeral): to indicate its charge. Iron(II), Copper(I) – determine charge from anion charge – Common Type II cations in Table 5. 5 3. Nonmetal anion Nonmetal name ended with –ide: Chloride, Oxide Example: Iron(II) chloride, Copper(I) oxide 2 -36 36

How to find the charge on Type II metal ions? • Example: Name Compound Fe 2 O 3 Since the sum of all charges equals zero, the charge on iron ions are unknown and oxide ion each has – 2 charge, then we have 2 x Fe + 3 x (-2) = 0 Fe = +3, each iron ion has a charge of +3 Name: iron(III) oxide Key: knowing the charge on ANIONs! 2 -37 37

Practice: Naming Ionic compounds • • 2 -38 Hg. F 2 Cu. I 2 Ca. Cl 2 Fe 2 S 3 Sn. Cl 4 Mg 3 N 2 Ag 2 S 38

• • 2 -39 Answer key: names of ionic compounds Hg. F 2 = Mercury(II) fluoride Cu. I 2 = copper(II) iodide Ca. Cl 2 = calcium chloride Fe 2 S 3 = Iron(III) sulfide Sn. Br 4 = tin(IV) bromide Mg 3 N 2 = magnesium nitride Ag 2 S = silver sulfide 39

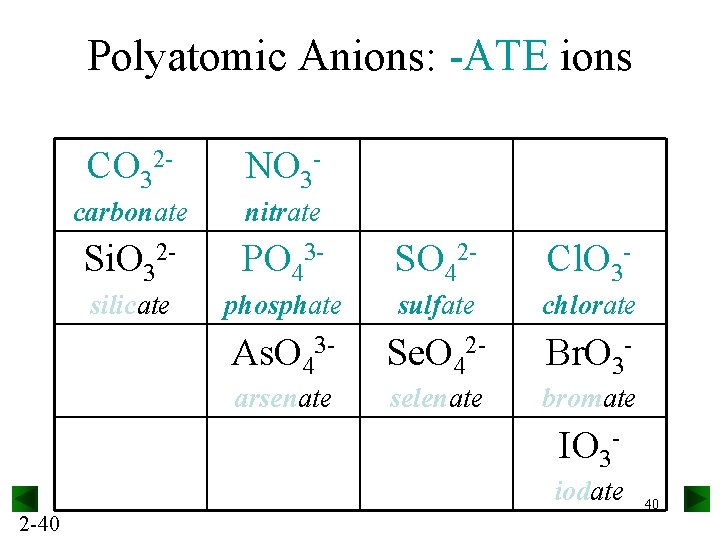
Polyatomic Anions: -ATE ions CO 32 - NO 3 - carbonate nitrate Si. O 32 - PO 43 - SO 42 - Cl. O 3 - silicate phosphate sulfate chlorate As. O 43 - Se. O 42 - Br. O 3 - arsenate selenate bromate IO 3 iodate 2 -40 40

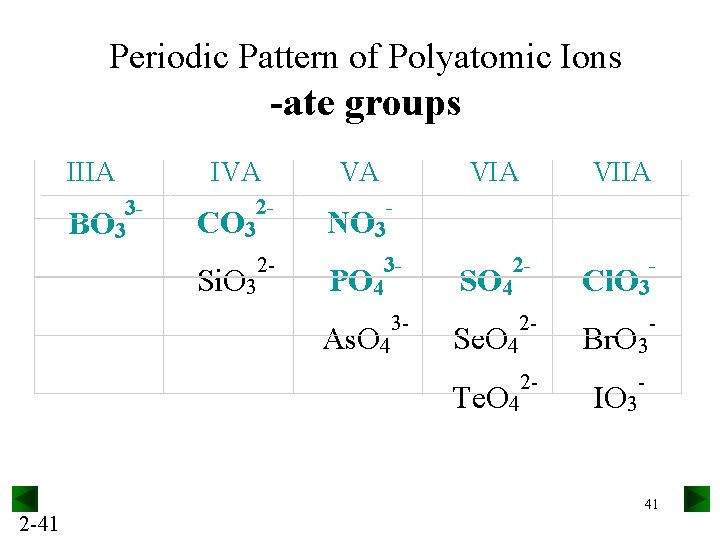
Periodic Pattern of Polyatomic Ions -ate groups IIIA 3 BO 3 2 -41 IVA VA VIIA 2 CO 3 NO 3 2 Si. O 3 3 PO 4 2 SO 4 Cl. O 3 3 As. O 4 2 Se. O 4 Br. O 3 2 Te. O 4 IO 3 41

Patterns for Polyatomic Ions 1. elements in the same Group form similar polyatomic ions – same number of O’s and same charge Cl. O 3 - = chlorate (-1 charge) Br. O 3 - = bromate (-1 charge) 2. if the polyatomic ion starts with H, the name adds hydrogen- prefix before name and add 1 to the charge CO 32 - = carbonate HCO 3 - = hydrogen carbonate 2 -42 42

Patterns for Polyatomic Ions -ate ion – chlorate = Cl. O 3 - • -ate ion + 1 O same charge, per- prefix – perchlorate = Cl. O 4 - • -ate ion – 1 O same charge, -ite suffix – chlorite = Cl. O 2 - • -ate ion – 2 O same charge, hypo- prefix, -ite suffix – hypochlorite = Cl. O 2 -43 43

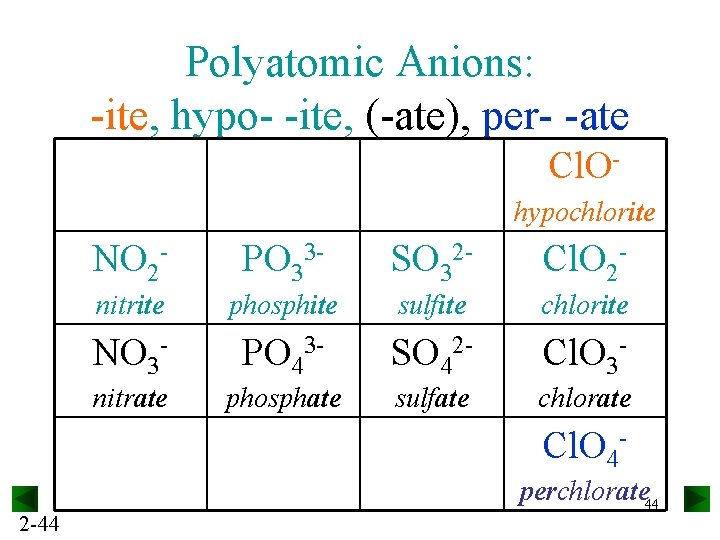
Polyatomic Anions: -ite, hypo- -ite, (-ate), per- -ate Cl. Ohypochlorite NO 2 - PO 33 - SO 32 - Cl. O 2 - nitrite phosphite sulfite chlorite NO 3 - PO 43 - SO 42 - Cl. O 3 - nitrate phosphate sulfate chlorate Cl. O 42 -44 perchlorate 44

Polyatomic Ions to Remember Name Formula acetate C 2 H 3 O 2 – hypochlorite Cl. O– carbonate CO 32– chlorite Cl. O 2– hydrogen carbonate (aka Bicarbonate) HCO 3 chlorate Cl. O 3– hydroxide OH– perchlorate Cl. O 4– nitrate NO 3– sulfate SO 42– nitrite NO 2– Hydrogen sulfate (aka Bisulfate) HSO 4– permanganate Mn. O 4– sulfite SO 32– chromate Cr. O 42– dichromate Cr 2 O 7 Hydrogen sulfite (aka Bisulfite) HSO 3– ammonium NH 4+ cyanide CN– 2 -45 – 2– 45

Practice: Naming Ionic compounds • • 2 -46 Hg 2 SO 4 Cu. Cl. O 3 Zn(NO 3)2 Fe. CO 3 Sn(SO 3)2 Co. PO 4 Al(Cl. O 4)3 46

Naming Acids All names have acid at end • Binary Acids (Hn. X) = hydro prefix + stem of the name of the nonmetal + ic suffix Example: HCl (Hydrochloric acid) • Oxyacids (Hn. XOm : H 2 CO 3 , H 2 SO 4) – if polyatomic ion ends in –ate = name of polyatomic ion with –ic suffix : H 2 SO 4 (Sulfuric acid); H 2 CO 3 (Carbonic acid); HNO 3 (Nitric acid); H 3 PO 4 (Phosphoric acid) – if polyatomic ion ends in –ite = name of polyatomic ion with –ous suffix 2 -47 47

Naming Binary Acids – HF 1. First of all, it is binary acid HX 2. Identify the anion F F-, fluoride because Group 7 A 2. Name the anion with an –ic suffix F- = fluoride fluoric 3. Add a hydro- prefix to the anion name hydrofluoric 4. Add the word acid to the end hydrofluoric acid 2 -48 48

Naming Oxyacids: H 2 SO 4 1. Identify the anion SO 4 = SO 42 - = sulfate 2. If the anion has –ate suffix, change it to –ic. If the anion has –ite suffix, change it to -ous SO 42 - = sulfate sulfuric 3. Write the name of the anion followed by the word acid sulfuric acid (kind of an exception, to make it sound nicer!) 2 -49 49

• • 2 -50 Practice: Naming Acids first: what is the anion? HNO 3 HCl. O 3 HBr H 2 CO 3 H 2 SO 3 H 3 PO 4 HCl. O 4 nitrate chlorate bromide carbonate sulfite phosphate perchlorate 50

Review: Naming Compounds • • 2 -51 Cu. SO 3 Ag. Cl. O N 2 O 5 H 2 S Fe. I 2 Sn(NO 3)4 Ba 3(PO 4)2 (NH 4)2 S 1. Common exceptions? H 2 O, NH 3, CH 4, C 12 H 22 O 11 2. Identify as Molecular or Ionic? 3. Identify • • Binary molecular Type I or II metal ion 51

Review: Naming Compounds • • 2 -52 Cu. SO 3 Ag. Cl. O N 2 O 5 H 2 S Fe. I 2 Sn(NO 3)4 Ba 3(PO 4)2 (NH 4)2 S copper(II) sulfite silver hypochlorite dinitrogen pentoxide hydrosulfuric acid iron(II) iodide tin(IV) nitrate barium phosphate ammonium sulfide 52

Write Chemical Formula using the charge of known ions • Example: Compound between Ca 2+ and PO 43, the number of ions of each needs to be 3 and 2, so that the combined charge = 3 x (+2) + 2 x (-3) = 0 Therefore the formula for the compound is Ca 3(PO 4)2 2 -53 53

Write Chemical Formula using the charge of known ions “Criss. Cross-Simplify”: • The charge of an ion turns into the subscript (the number) of the counterpart ion Pb 4+ O 2 - Pb 2 O 4 • Since the subscripts in an ionic compound represents the RATIO among the ions, the subscripts need to be simplified when there is common denominator Pb 2 O 4 Pb. O 2 2 -54 54

Writing formulae • • 2 -55 copper(II) sulfate aluminum perchlorate hydroiodic acid iron(III) bromide Diphosphorus pentoxide lead(IV) nitride zinc carbonate helium gas 55

Tools of the Laboratory Basic Separation Techniques Filtration: Separates components of a mixture based upon differences in particle size. Filtration usually involves separating a precipitate from solution. Crystallization: Separation is based upon differences in solubility of the components in a mixture. Distillation: Separation is based upon differences in volatility. Extraction: Separation is based upon differences in solubility in different solvents (major material). Chromatography: Separation is based upon differences in solubility in a solvent versus a stationary phase. 2 -56

Tools of the Laboratory Distillation: Separation through Difference in Boiling Temperatures 2 -57

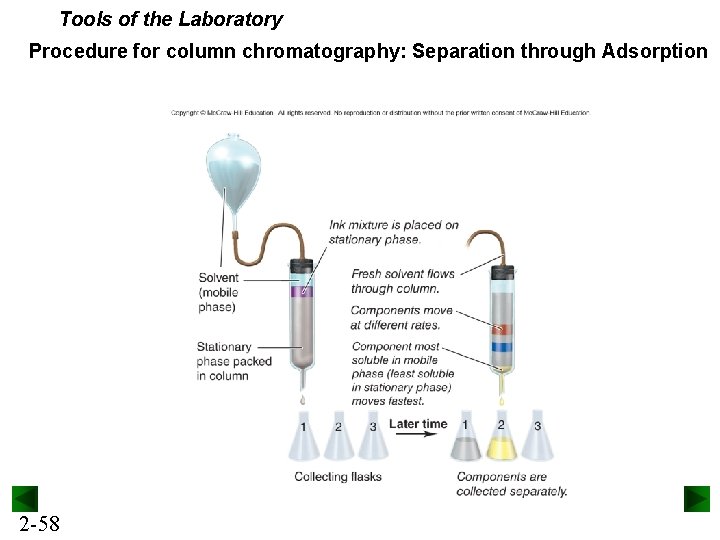
Tools of the Laboratory Procedure for column chromatography: Separation through Adsorption 2 -58

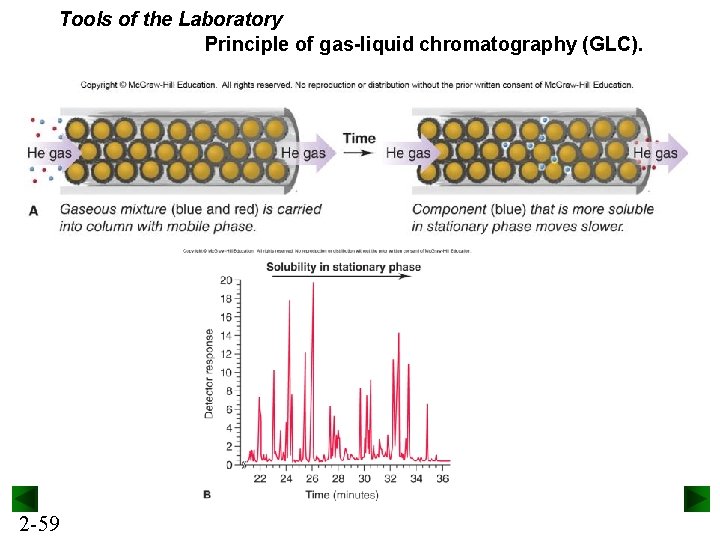
Tools of the Laboratory Principle of gas-liquid chromatography (GLC). 2 -59