9 Fermi Surfaces and Metals Construction of Fermi










![Example: Fermi Surface of Gold d. Hv. A in Au with B // [110]: Example: Fermi Surface of Gold d. Hv. A in Au with B // [110]:](https://slidetodoc.com/presentation_image_h2/805352044c8a26c2fdd128028c7ff0ab/image-33.jpg)
- Slides: 35

9. Fermi Surfaces and Metals • • Construction of Fermi Surfaces Electron Orbits, Hole Orbits, and Open Orbits Calculation of Energy Bands Experimental Methods in Fermi Surface Studies

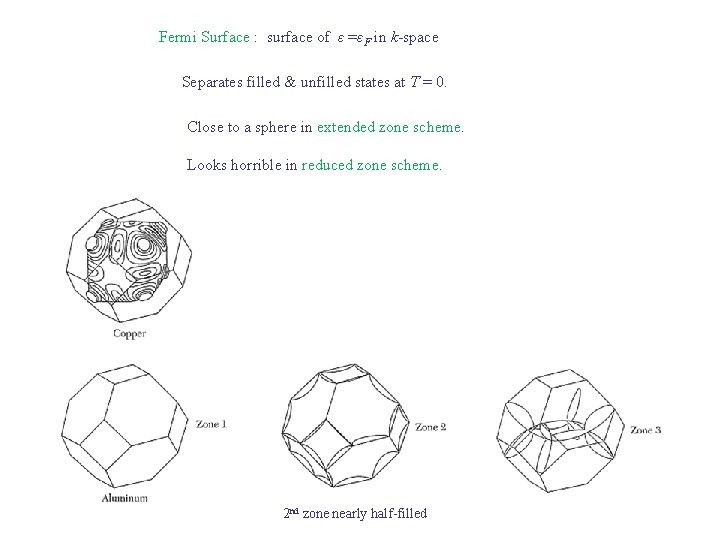
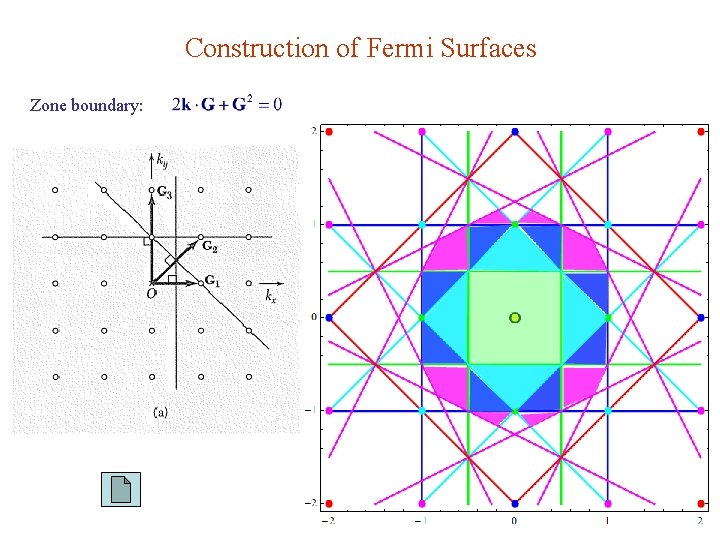
Fermi Surface : surface of ε =εF in k-space Separates filled & unfilled states at T = 0. Close to a sphere in extended zone scheme. Looks horrible in reduced zone scheme. 2 nd zone nearly half-filled

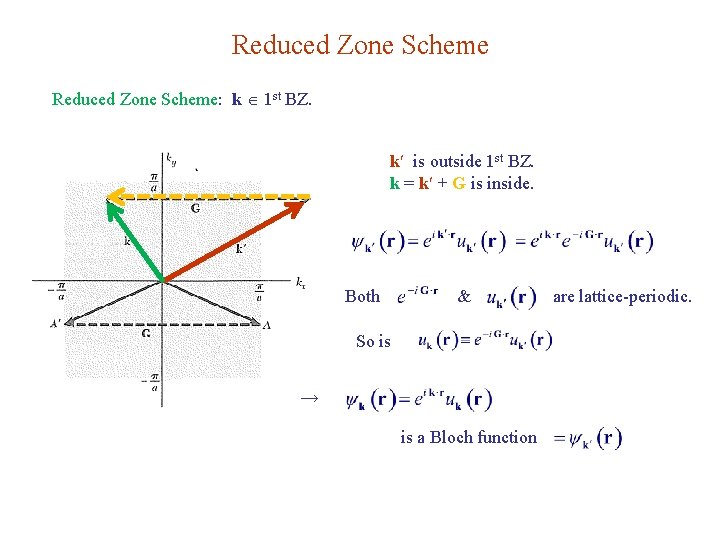
Reduced Zone Scheme: k 1 st BZ. k is outside 1 st BZ. k = k + G is inside. Both & So is → is a Bloch function are lattice-periodic.

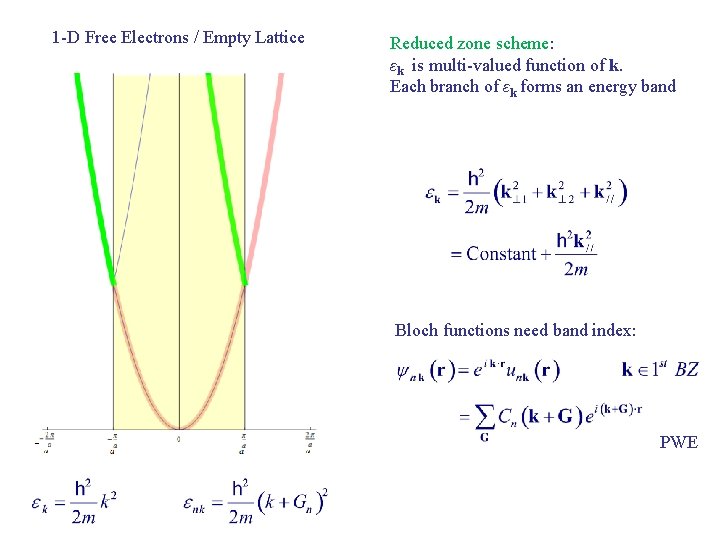
1 -D Free Electrons / Empty Lattice Reduced zone scheme: εk is multi-valued function of k. Each branch of εk forms an energy band Bloch functions need band index: PWE

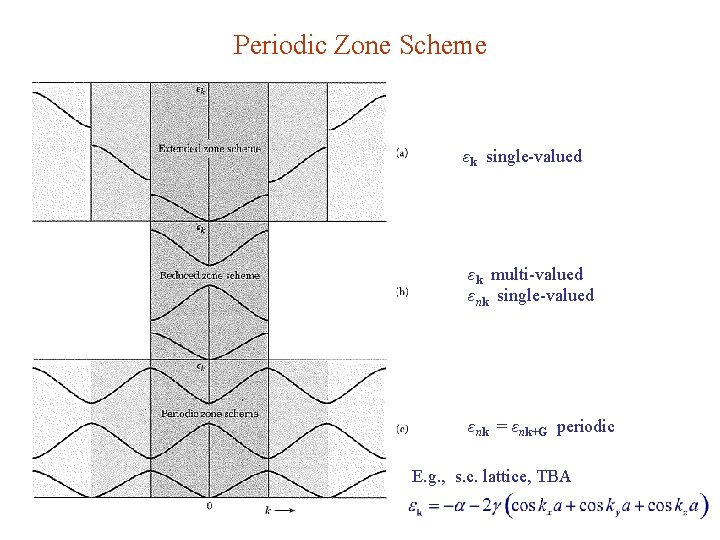
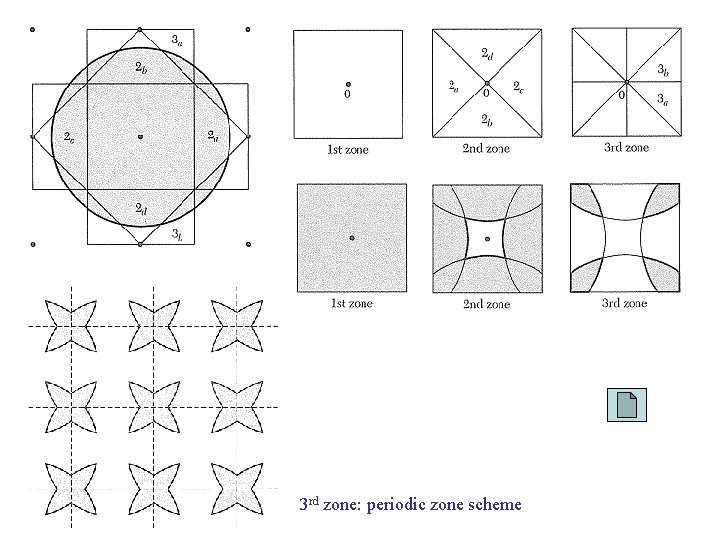
Periodic Zone Scheme εk single-valued εk multi-valued εnk single-valued εnk = εnk+G periodic E. g. , s. c. lattice, TBA

Construction of Fermi Surfaces Zone boundary:

3 rd zone: periodic zone scheme

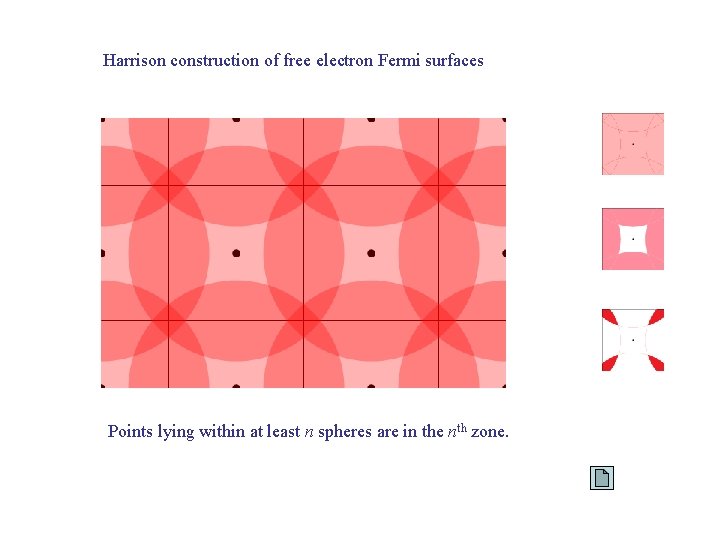
Harrison construction of free electron Fermi surfaces Points lying within at least n spheres are in the nth zone.

Nearly free electrons: Energy gaps near zone boundaries → Fermi surface edges “rounded”. Fermi surfaces & zone boundaries are always orthogonal.

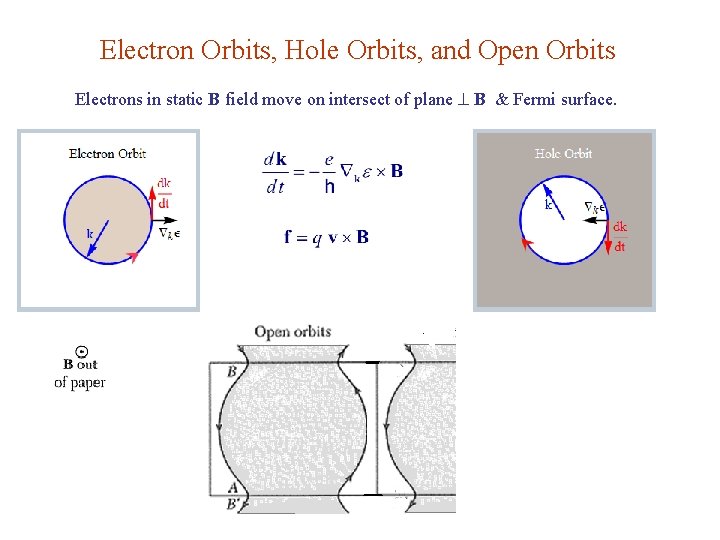
Electron Orbits, Hole Orbits, and Open Orbits Electrons in static B field move on intersect of plane B & Fermi surface.

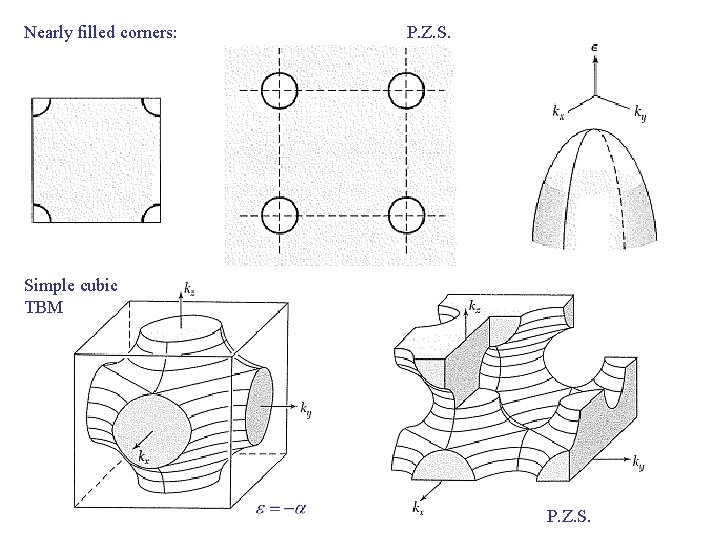
Nearly filled corners: P. Z. S. Simple cubic TBM P. Z. S.

Calculation of Energy Bands • • Tight Binding Method for Energy Bands Wigner-Seitz Method Cohesive Energy Pseudopotential Methods

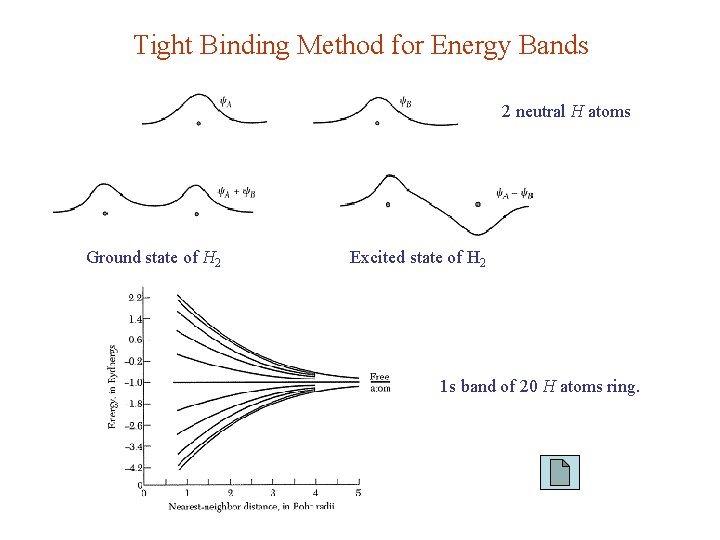
Tight Binding Method for Energy Bands 2 neutral H atoms Ground state of H 2 Excited state of H 2 1 s band of 20 H atoms ring.

TBM / LCAO approximation Good for valence bands, less so for conduction bands. α = s, p, d, … Bravais lattice , s-orbital only: ψk is a Bloch function since 1 st order energy: Keep only on site & nearest neighbor terms: j runs over the basis atoms

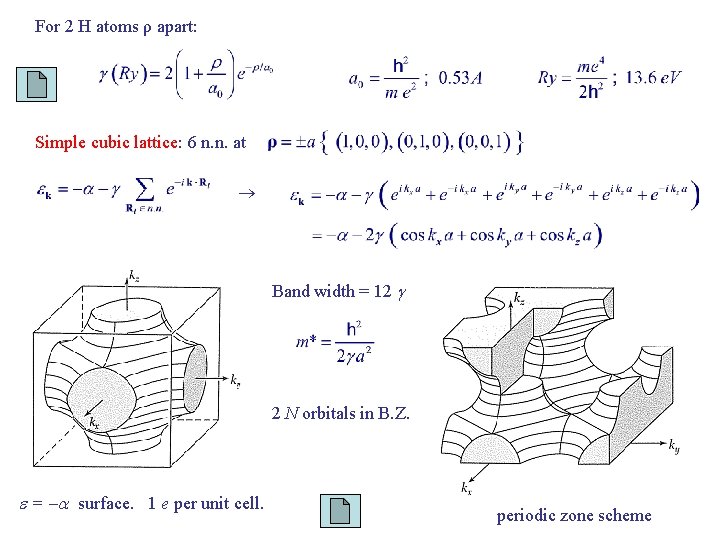
For 2 H atoms ρ apart: Simple cubic lattice: 6 n. n. at Band width = 12 2 N orbitals in B. Z. = surface. 1 e per unit cell. periodic zone scheme

Fcc lattice: 12 n. n. at surface Band width = 24

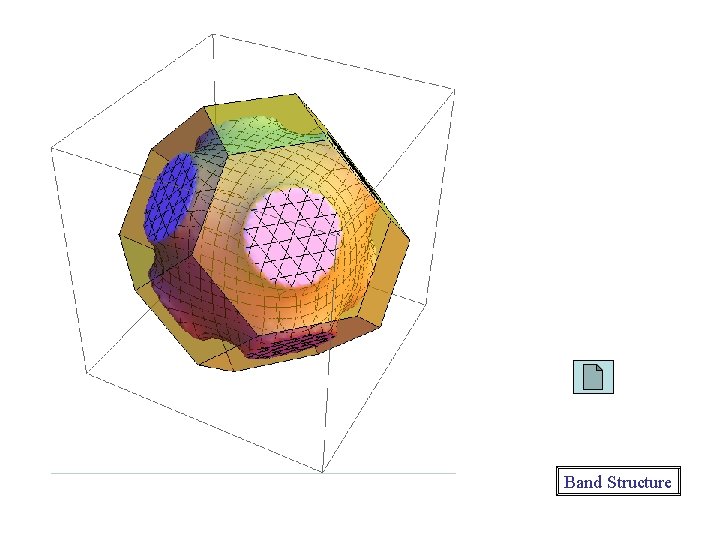
Band Structure

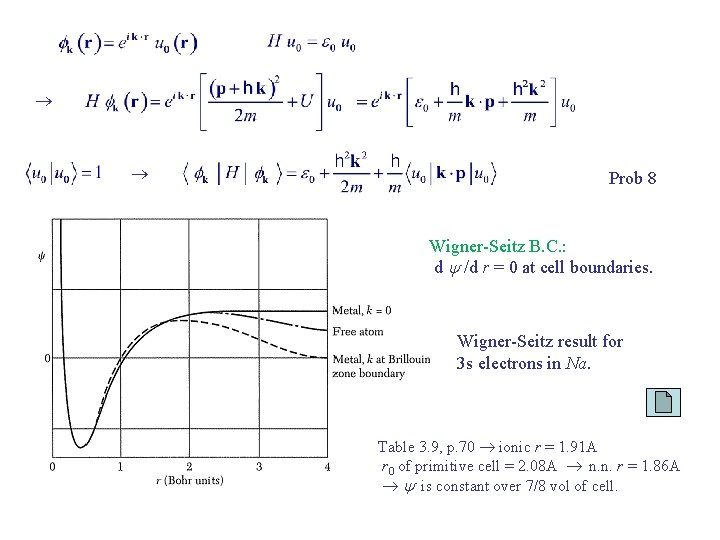
Wigner-Seitz Method Bloch function: Schrodinger eq. : For k = 0, we have u 0 is periodic in R l. is a Bloch function; can serve as an approximate solution of the Schrodinger eq. for k 0.

Prob 8 Wigner-Seitz B. C. : d /d r = 0 at cell boundaries. Wigner-Seitz result for 3 s electrons in Na. Table 3. 9, p. 70 ionic r = 1. 91 A r 0 of primitive cell = 2. 08 A n. n. r = 1. 86 A is constant over 7/8 vol of cell.

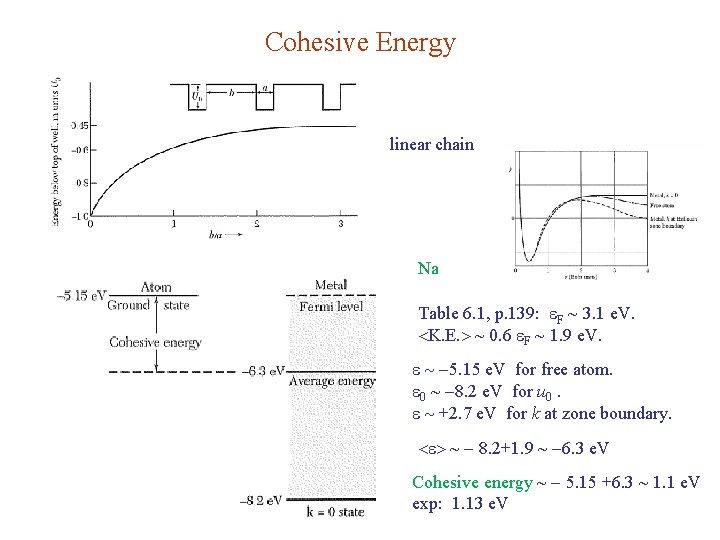
Cohesive Energy linear chain Na Table 6. 1, p. 139: F ~ 3. 1 e. V. K. E. ~ 0. 6 F ~ 1. 9 e. V. 5. 15 e. V for free atom. 0 ~ 8. 2 e. V for u 0. +2. 7 e. V for k at zone boundary. ~ 8. 2+1. 9 ~ 6. 3 e. V Cohesive energy ~ 5. 15 +6. 3 ~ 1. 1 e. V exp: 1. 13 e. V

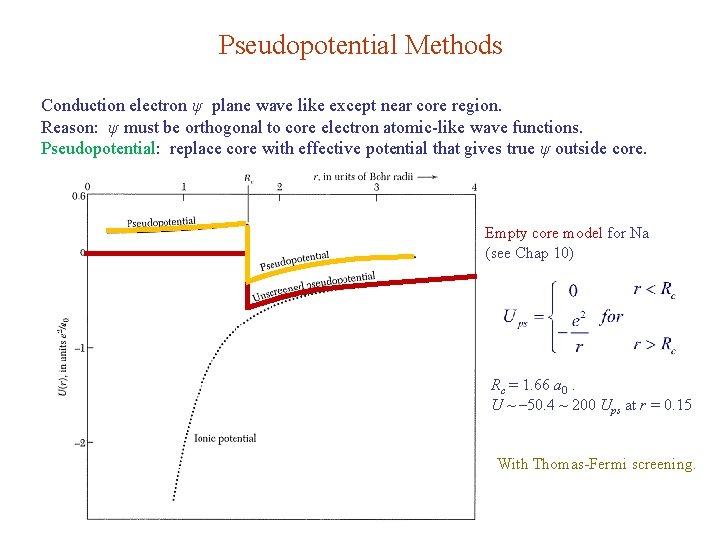
Pseudopotential Methods Conduction electron ψ plane wave like except near core region. Reason: ψ must be orthogonal to core electron atomic-like wave functions. Pseudopotential: replace core with effective potential that gives true ψ outside core. Empty core model for Na (see Chap 10) Rc = 1. 66 a 0. U ~ – 50. 4 ~ 200 Ups at r = 0. 15 With Thomas-Fermi screening.

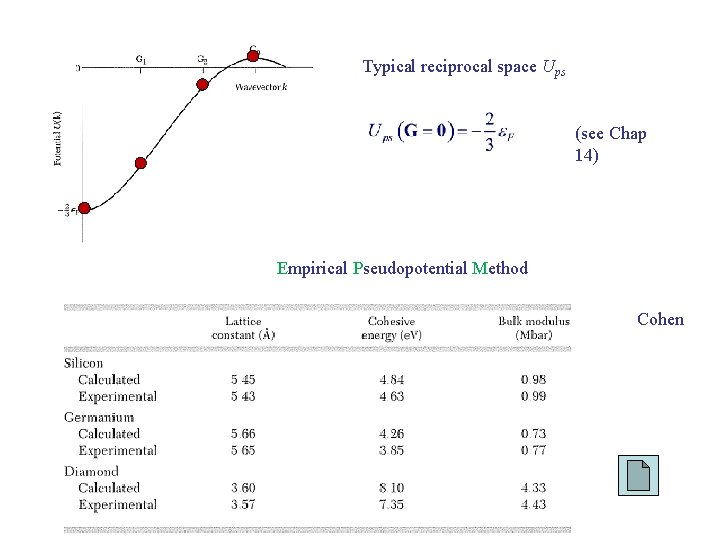
Typical reciprocal space Ups (see Chap 14) Empirical Pseudopotential Method Cohen

Experimental Methods in Fermi Surface Studies • • • Quantization of Orbits in a Magnetic Field De Haas-van Alphen Effect Extremal Orbits Fermi Surface of Copper Example: Fermi Surface of Gold Magnetic Breakdown

Experimental methods for determining Fermi surfaces: • Magnetoresistance • Anomalous skin effect • Cyclotron resonance • Magneto-acoustic geometric effects • Shubnikov-de Haas effect • de Haas-van Alphen effect Experimental methods for determining momentum distributions: • Positron annihilation • Compton scattering • Kohn effect Metal in uniform B field → 1/B periodicity

Quantization of Orbits in a Magnetic Field q = –e for electrons Bohr-Sommerfeld quantization rule: Phase corrector γ = ½ for free electrons B = const → Flux quantization Dirac flux quantum

→ For Δr B : Let A = Area in r-space, S = Area in k-space. → Hence Area of orbit in k-space is quantized If then Properties that depend on S are periodic functions of 1/B.

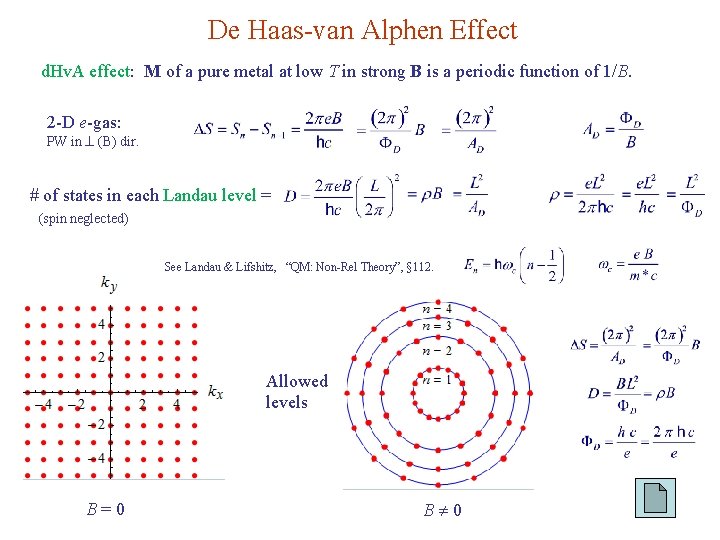
De Haas-van Alphen Effect d. Hv. A effect: M of a pure metal at low T in strong B is a periodic function of 1/B. 2 -D e-gas: PW in (B) dir. # of states in each Landau level = (spin neglected) See Landau & Lifshitz, “QM: Non-Rel Theory”, § 112. Allowed levels B=0 B 0

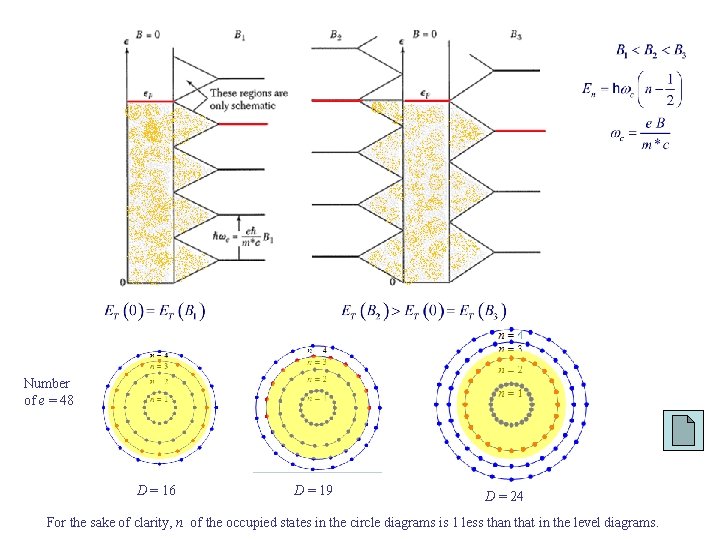
Number of e = 48 D = 16 D = 19 D = 24 For the sake of clarity, n of the occupied states in the circle diagrams is 1 less than that in the level diagrams.

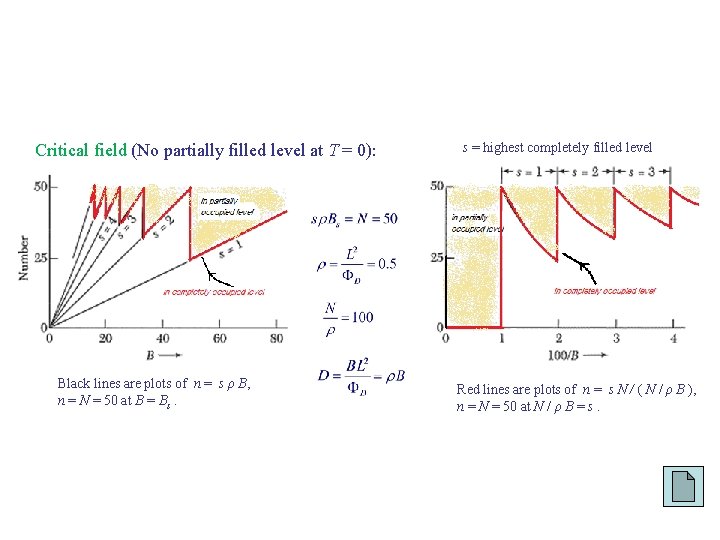
Critical field (No partially filled level at T = 0): Black lines are plots of n = s ρ B, n = N = 50 at B = Bs. s = highest completely filled level Red lines are plots of n = s N / ( N / ρ B ), n = N = 50 at N / ρ B = s.

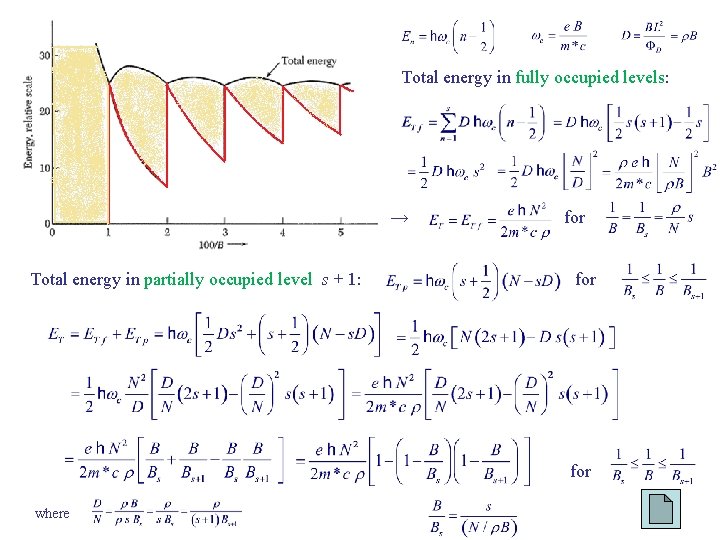
Total energy in fully occupied levels: → Total energy in partially occupied level s + 1: for for where

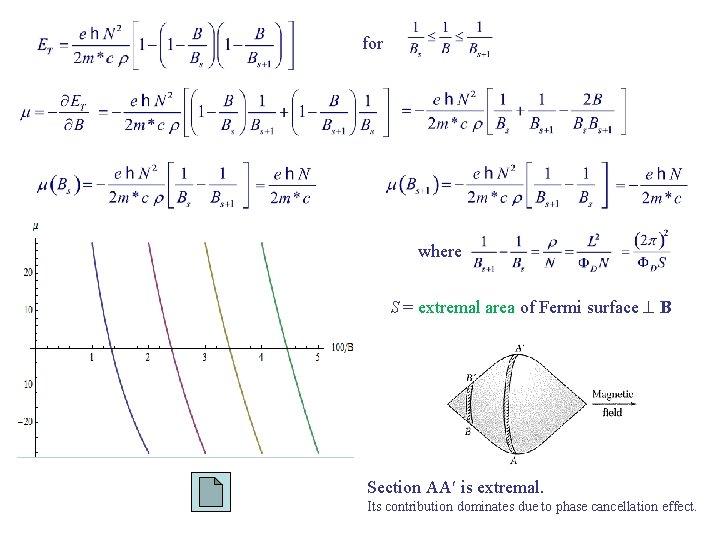
for where S = extremal area of Fermi surface B Section AA is extremal. Its contribution dominates due to phase cancellation effect.

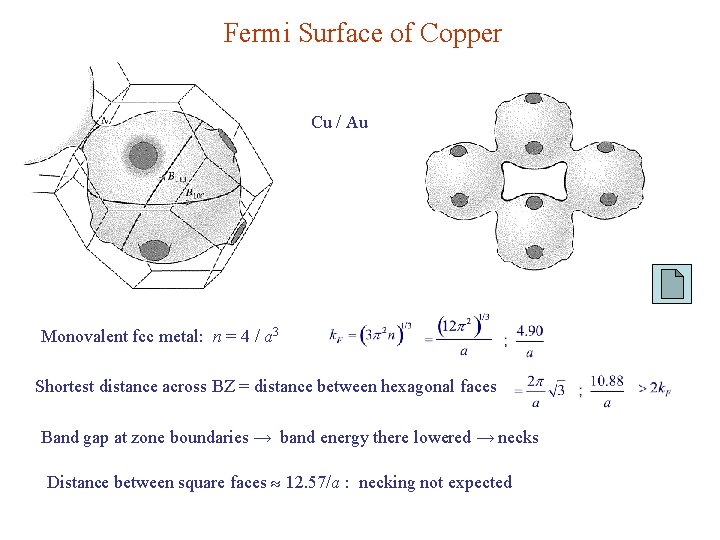
Fermi Surface of Copper Cu / Au Monovalent fcc metal: n = 4 / a 3 Shortest distance across BZ = distance between hexagonal faces Band gap at zone boundaries → band energy there lowered → necks Distance between square faces 12. 57/a : necking not expected
![Example Fermi Surface of Gold d Hv A in Au with B 110 Example: Fermi Surface of Gold d. Hv. A in Au with B // [110]:](https://slidetodoc.com/presentation_image_h2/805352044c8a26c2fdd128028c7ff0ab/image-33.jpg)
Example: Fermi Surface of Gold d. Hv. A in Au with B // [110]: Dogbone μ has period 2 10– 9 gauss– 1 for most directions → Table 6. 1: → Period along [111] is 6 10– 8 gauss– 1 → Dogbone area ~ 0. 4 of belly area → neck

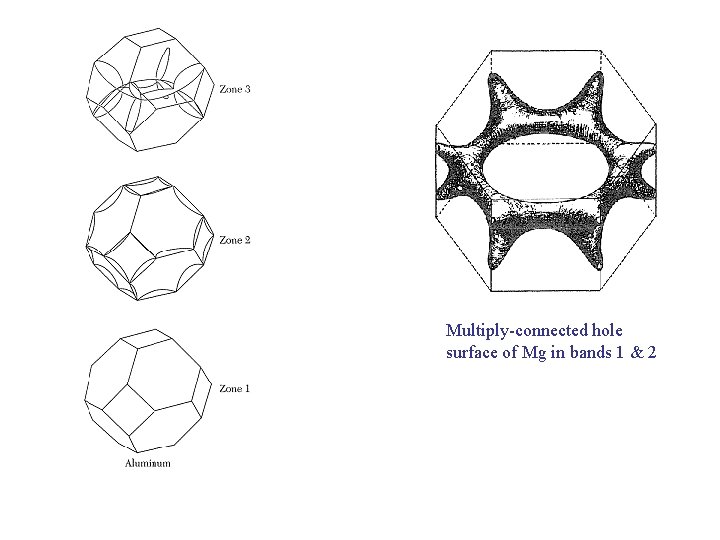
Multiply-connected hole surface of Mg in bands 1 & 2

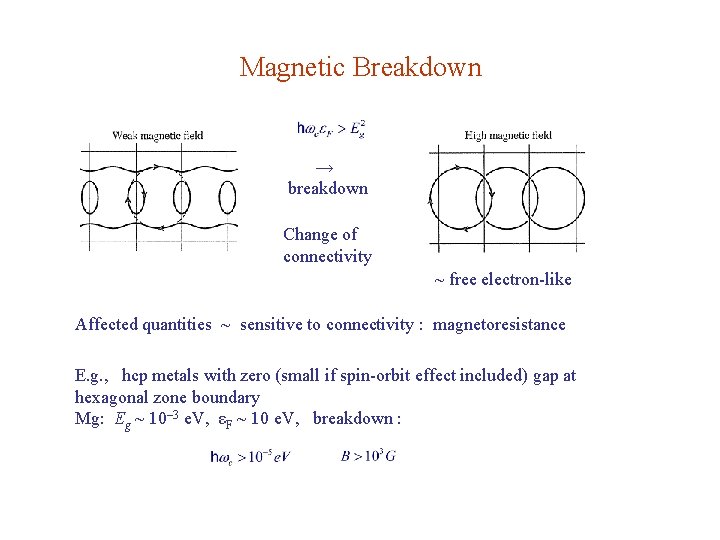
Magnetic Breakdown → breakdown Change of connectivity ~ free electron-like Affected quantities ~ sensitive to connectivity : magnetoresistance E. g. , hcp metals with zero (small if spin-orbit effect included) gap at hexagonal zone boundary Mg: Eg ~ 10– 3 e. V, εF ~ 10 e. V, breakdown :