1 Atom The smallest particle of an element









- Slides: 16

1. Atom • The smallest particle of an element that still has the properties of that element

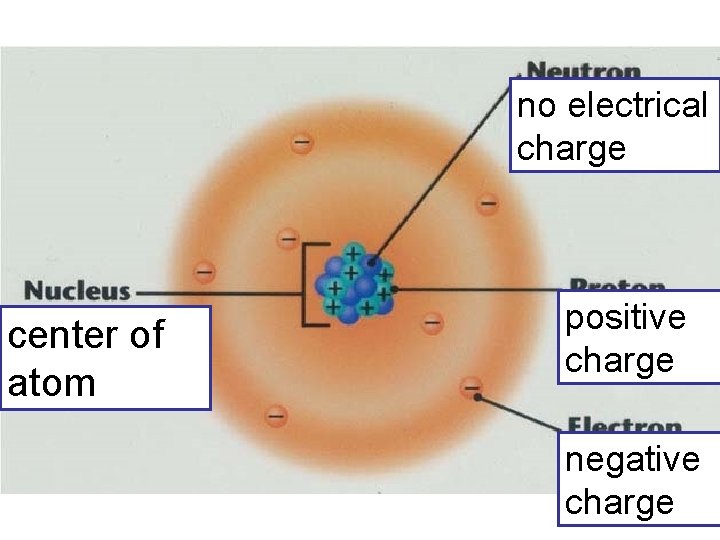
no electrical charge center of atom positive charge negative charge

2. Compound • Made of combination of elements • Properties of compounds often differ from properties of elements that make them up

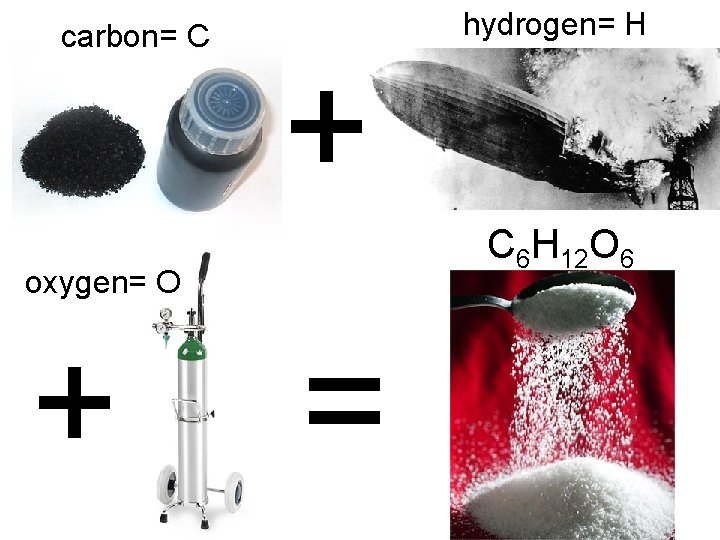
carbon= C hydrogen= H + C 6 H 12 O 6 oxygen= O + =

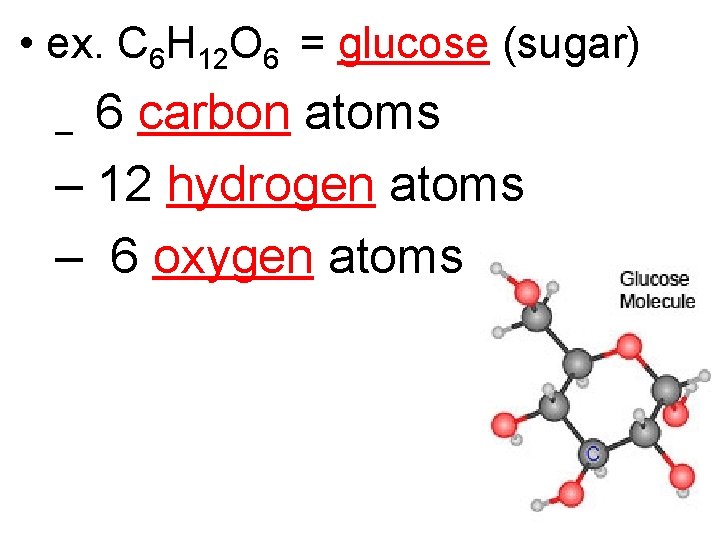
• ex. C 6 H 12 O 6 = glucose (sugar) 6 carbon atoms – 12 hydrogen atoms – 6 oxygen atoms –

3. Atomic Theory – the beginning • Democritus believed that the tiniest particle was the atom, which was the smallest thing that could ________________. • He also thought that atoms were made of a _________________ that formed into different shapes and sizes. be divided to single material Atomos indivisible

4. John Dalton’s Atomic Theory • Based on scientific _____________ through _________, John Dalton published a theory in 1803. His theory states these ideas: evidence experiments

4. John Dalton’s Atomic Theory • All substances are made of ________. Atoms are small particles that cannot be ________, _________, or _____________. • Atoms of the same element are ________________. and the atoms of different elements are ______________. • Atoms join with other atoms to make _______________. atoms created divided destroyed exactly alike different. new substances

5. Disagreeing with Dalton • As scientists were able to gather new data, more discoveries about the atom showed some mistakes with Dalton’s theory. • • J. J. Thompson discovered with a _____________ that atoms contained __________-charged particles called __________. He came up with the _______________ model. cathode-ray negatively electrons. Plum-pudding

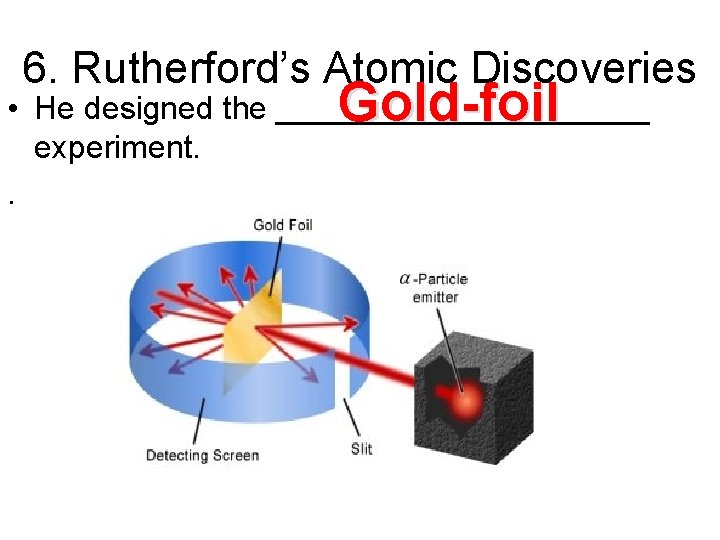
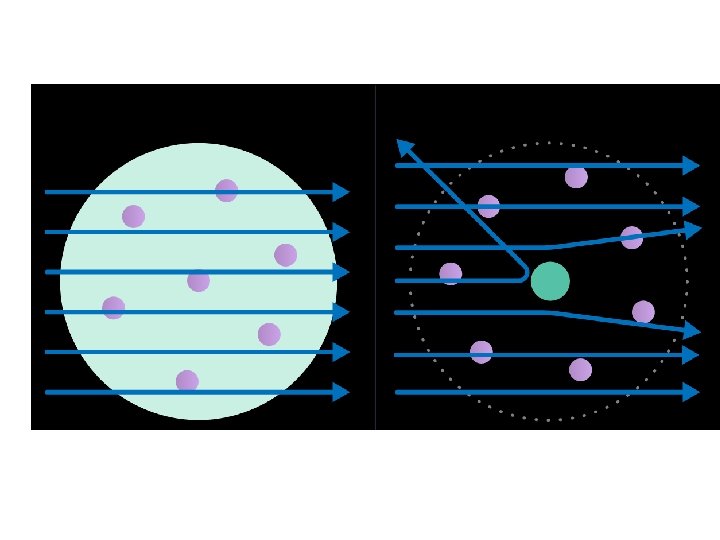
6. Rutherford’s Atomic Discoveries • Later, one of Thompson’s students named Ernest Rutherford discovered the ____________-charged particles in the center of the atom. . positively

6. Rutherford’s Atomic Discoveries Gold-foil • He designed the ___________ experiment. .


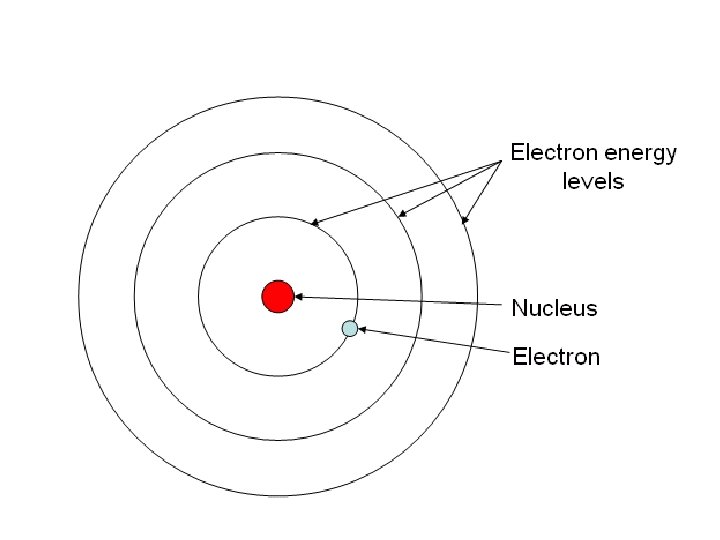
7. Bohr’s Model and Modern Theory • With more experiments and evidence, Niels Bohr developed his model named after himself, called the ________ model. • This model represents a small, positivelycharged _________ Bohr nucleus surrounded by ___________ electrons that travel in circular orbits _________________ around the nucleus.


7. Bohr’s Model and Modern Theory • Modern Theory developed by scientists, especially Schrödinger and Heisenberg, states that electrons are found in probable _______________ called regions Electron clouds _________________.

Now let’s draw the models that evolved throughout history.