Structure of Atom Atom Smallest particle of an












- Slides: 17

Structure of Atom

Atom Smallest particle of an element Atomic Structure – arrangement of smaller particles within an atom

Two Main Regions In Atom Nucleus – very small dense central region protons – positively charged particles neutrons – neutral particles ● Electron Cloud – large region around nucleus electrons – negatively charged particles

Discovery of Electrons JJ Thomson – used Crooke’s Tube (cathode ray tube) Gas under low pressure in a sealed glass tube Has a positive end (Anode) and negative end (Cathode) When anode and cathode are connected to the + and – terminals of a battery, the tube produces a beam of particles called electrons



Thomson’s Atom Plum-pudding model Since total atom is neutral, there must be an equal number of + and - charges Atom is a positively charged area with negative electrons randomly spread throughout

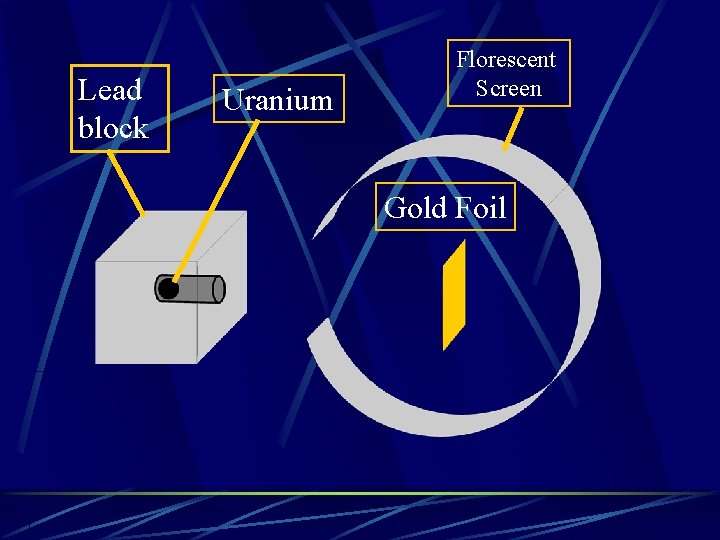
Nucleus Ernest Rutherford – Gold foil experiment Shot positive charged particles at a very thin piece of gold foil

Lead block Uranium Florescent Screen Gold Foil

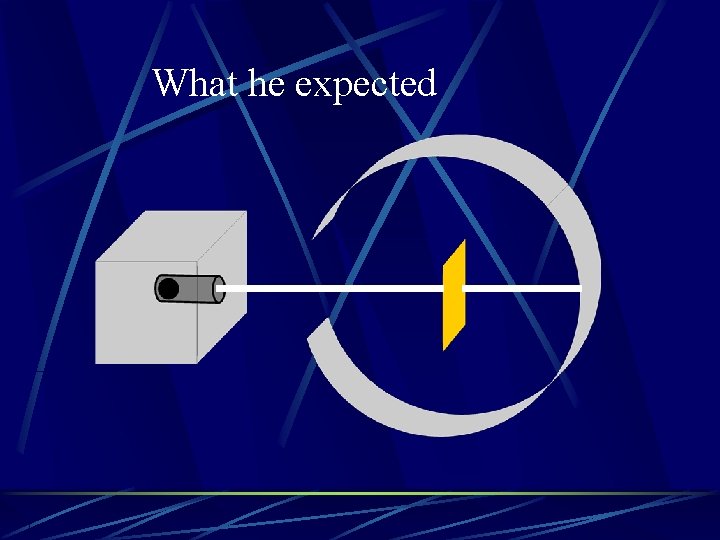
What he expected

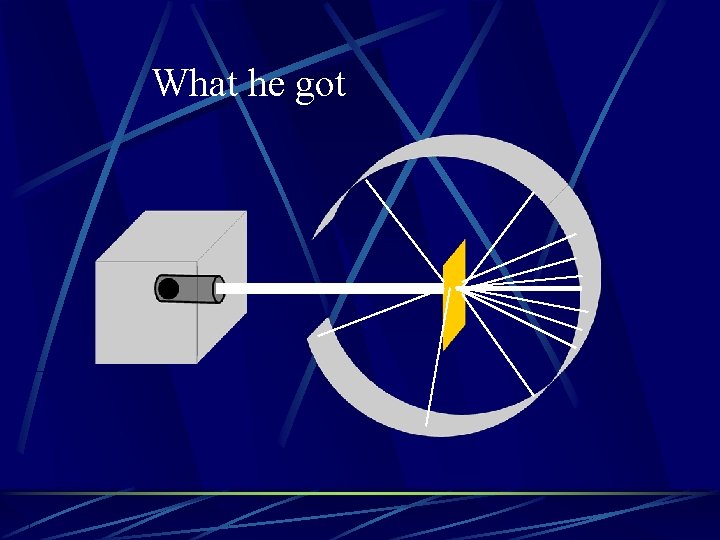
What he got


How he explained it Atom is mostly empty space Small dense, positive piece at center Alpha particles are deflected by it if they get close enough +

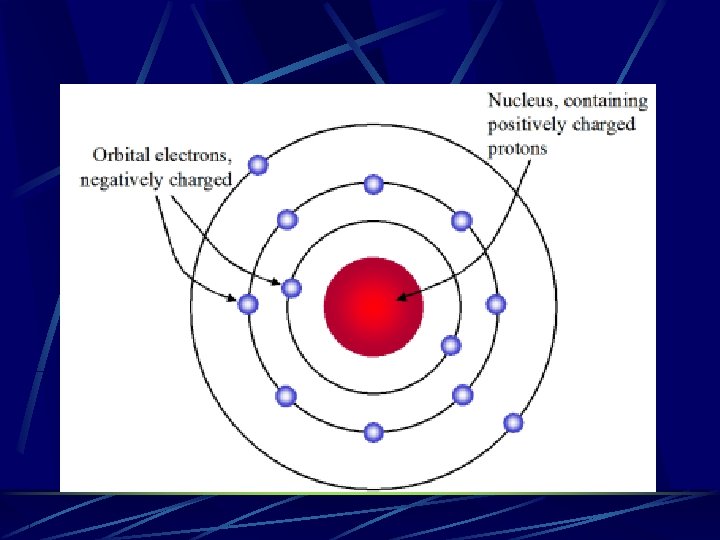
Rutherford’s Model Of Atom Positively charged nucleus containing protons and neutrons with negatively charged particles in orbit around it Electrons stay in orbit due to attraction between + and – charges

Problems With Rutherford Model Charged particle when moving in a curved path give off light so they lose energy If they lose energy, they would gradually get closer and closer to the nucleus and crash into it

Bohr Model of Atom Similar to Rutherford model, but in this model electrons orbit in specific, well defined energy level A gain or loss of energy moves the electron from one energy level to another Positively charged protons and neutral neutrons held together tightly by the Strong Nuclear Force
