SECTION 1 STRUCTURE OF THE ATOM Chapter 17
















- Slides: 19

SECTION 1: STRUCTURE OF THE ATOM Chapter 17: Properties of Atoms

LEARNING GOALS Identify the names and symbols of common elements. Identify quarks as subatomic particles of matter. Describe the electron cloud model of the atom. Explain how electrons are arranged in an atom.

WARM-UP: Agree or Disagree

SCIENTIFIC SHORTHAND Each element can be symbolized with a chemical symbol. �A short or abbreviated way to write the name of an element. �Consist of one capital letter plus one or two small letters.

SCIENTIFIC SHORTHAND Some elements use the first letter or letters of their name to symbolize the element: �Aluminum (Al) �Calcium (Ca) �Carbon (C) �Nitrogen (N)

SCIENTIFIC SHORTHAND The symbols of other elements are derived from Latin: �Sodium (Na) �Silver (Ag) �Gold (Au) �Potassium (K)

ATOMIC COMPONENTS An element is composed of one type of atom. �Atom: the smallest piece of matter that still retains the properties of that element.

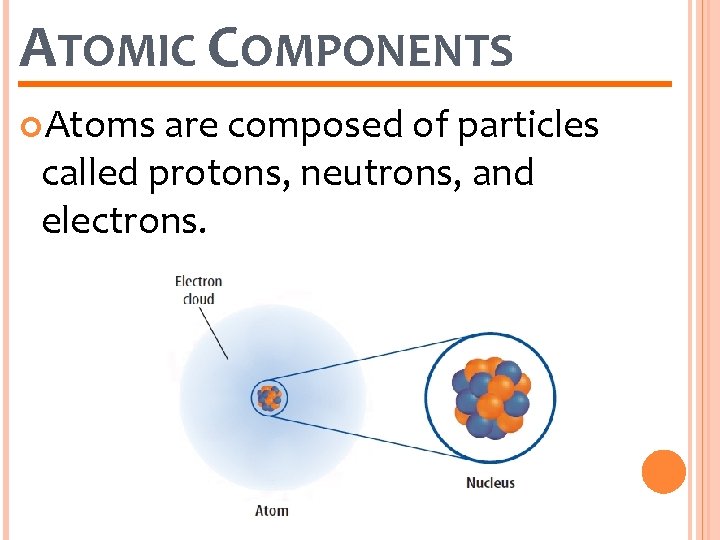
ATOMIC COMPONENTS Atoms are composed of particles called protons, neutrons, and electrons.

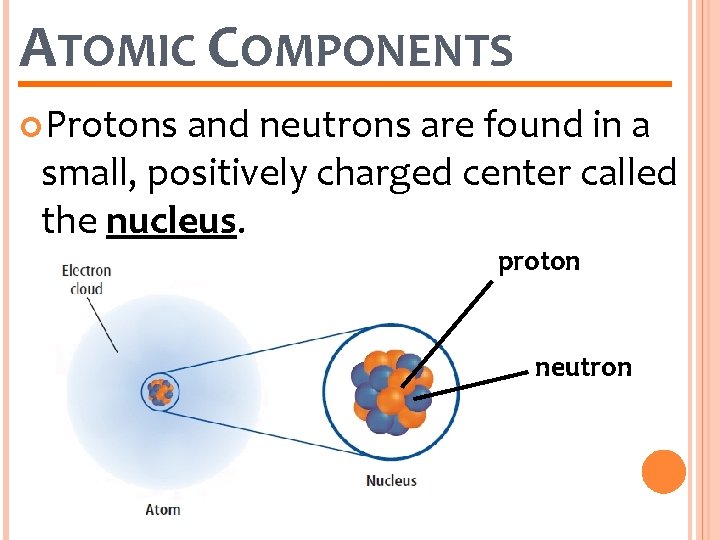
ATOMIC COMPONENTS Protons and neutrons are found in a small, positively charged center called the nucleus. proton neutron

ATOMIC COMPONENTS Protons are particles with an electrical charge of 1+ �They are made up of even smaller particles called quarks. �Different elements have different numbers of protons

ATOMIC COMPONENTS Neutrons are particles that do not have an electrical charge. �They are also made up of even smaller particles called quarks. �The number of neutrons in a single element may vary

ATOMIC COMPONENTS Electrons are particles with a charge of 1�Electrons are 1/1836 the size of a proton and can be found somewhere in the electron cloud. �The number of electrons in a single element may vary

ATOMIC MODELS Atoms are incredibly small! �It would take 24, 400 atoms stacked one on top of another to equal the thickness of a sheet of aluminum foil. We need models in order to visualize and understand the atom.

ATOMIC MODELS 400 B. C. �Democritus proposed the idea that substances were made up of tiny, indestructible particles called atoms. �Aristotle claimed that matter was uniform throughout and could be made of earth, water, air, or fire. Aristotle’s theory was accepted for the next 2, 000 years!

ATOMIC MODELS 1805: �John Dalton offered proof that atoms exist �Created an early model of the atom that showed a simple sphere

ATOMIC MODELS 1926: �The electron cloud model was developed and is still used today �Electron cloud: the area around the nucleus of an atom where its electrons are likely to be found.

ATOMIC MODELS �The electron cloud is 100, 000 times larger than the diameter of the nucleus. �Because electrons are so small and move so quickly, it is impossible to describe their exact locations.


CHECK-IN: Identify the names, charges, and locations of three kinds of particles that make up an atom.