Use of AutoInjectors by Civilian Emergency Medical Personnel

Use of Auto-Injectors by Civilian Emergency Medical Personnel to Treat Civilians Exposed to Nerve Agent© OEMS Approval # for 2007: 274546 (2 hours) © 1997 Lockheed Martin Energy Research Corporation

Learning Objectives Identify the antidotes to be administered in event of nerve agent exposure Identify the conditions under which antidote auto-injectors should be used Demonstrate use of antidote auto-injectors Recognize adverse reactions to use of antidotes AI 2©

IMPORTANT! Antidotes counter or relieve effects of poisons such as nerve agents. Use antidotes only when signs and symptoms of exposure are present; they will not protect if given before exposure. AI 3©

Auto-Injectors Simple, compact injection systems Permit rapid injection of required antidotes Prevent needle from being subject to crosscontamination Enable rapid and accurate administration even if care giver or patient is in protective clothing AI 4©

Auto-Injectors Auto-injectors facilitate timely treatment by providing Simple, accurate, rapid drug administration Pre-measured, controlled dose No vials, ampules or syringes to manipulate Fully automatic operation Rugged construction AI 5©

Auto-Injectors Contain pre-measured doses of nerve agent antidotes atropine and 2 -PAM chloride (2 -PAM Cl, pralidoxime chloride or PROTOPAM® CHLORIDE) FDA-approved and commercially available for use by civilian emergency medical personnel in the event of an accidental release of nerve agent or organophosphorus insecticides AI 6©

Auto-Injectors States decide if they will use them One U. S. -based manufacturer: Survival Technology Inc. (STI) AI 7©

Who Can Use Auto-Injectors Concern about administration of inappropriate and harmful doses during emergency situations • Pre-measured doses in auto-injectors safe for civilians meeting profile of military personnel – approximate ages of 18– 35 – weight at least 70 kg or 154 pounds – healthy with no compromising medical conditions Age, sex, weight and health of all other patients must first be considered when determining amount of atropine and 2 -PAM Cl to administer AI 8©

Who Can Use Auto-Injectors Only qualified civilian emergency responders with adequate training in on-site recognition and treatment of nerve agent intoxication in the event of an accidental release Majority of CSEPP States follow DOT national curriculum for Emergency Medical Technicians • Curriculum generally restricts training on use of IVs to Intermediate Emergency Medical Technicians and paramedics Review of each state’s laws and regulations needed to determine appropriate use of autoinjectors AI 9©

When To Use Auto-Injectors Use only after following events have occurred: Emergency medical personnel have donned personal protective equipment subsequent to recognizing existence of chemical agent hazard in area Some or all of symptoms of nerve agent poisoning cited are present AI 10©
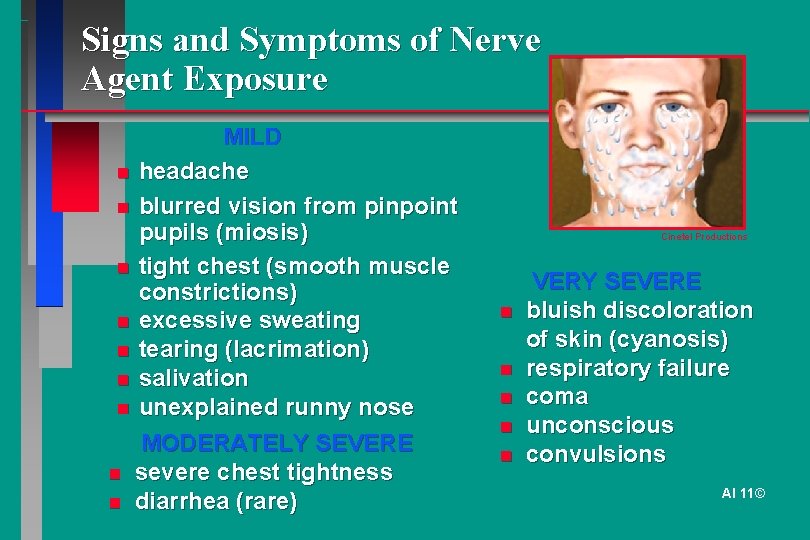
Signs and Symptoms of Nerve Agent Exposure MILD headache blurred vision from pinpoint pupils (miosis) Cinetel Productions tight chest (smooth muscle VERY SEVERE constrictions) bluish discoloration excessive sweating of skin (cyanosis) tearing (lacrimation) respiratory failure salivation unexplained runny nose coma unconscious MODERATELY SEVERE convulsions severe chest tightness AI 11© diarrhea (rare)

Treatment For Exposure To Nerve Agents Depending on severity of symptoms, immediately administer 1 atropine autoinjector, followed by 1 2 -PAM Cl auto-injector • Atropine should be given first, followed immediately by 2 -PAM Cl If nerve agent signs and symptoms are still present after 5– 10 minutes, repeat injections AI 12©

Treatment For Exposure To Nerve Agents If signs and symptoms still exist after additional 10 minutes, repeat injections for third time If signs and symptoms remain after third set of injections, do not give any more antidotes but seek immediate medical help AI 13©

Treatment For Exposure To Nerve Agents If severe signs and symptoms are present: In case of very severe exposure, all three autoinjector kits (atropine and 2 -PAM Cl) should be administered in rapid succession; then medical help should be sought Remove secretions, maintain patient’s airway and, if necessary, use artificial ventilation 2 -PAM Cl is most effective if administered immediately after poisoning. Generally, little is accomplished if drug is given more than 6 hours after termination of exposure AI 14©

Directions for Use 1. Remove safety cap (yellow on atropine; gray on 2 -PAM Cl). Mark I kit clip holds the safety caps; may notice if using Mark I kits. Do not touch colored end of injector after removing cap; injector can and will function into fingers or hand if any pressure applied to this end of injector AI 15©

Directions for Use 2. Hold injector like a pen. Place colored end (green on atropine, black on 2 -PAM Cl) on thickest part of thigh and press hard until injector functions • Pressure automatically activates the spring, plunges the needle into the muscle and simultaneously forces fluid through it into the muscle tissues AI 16©
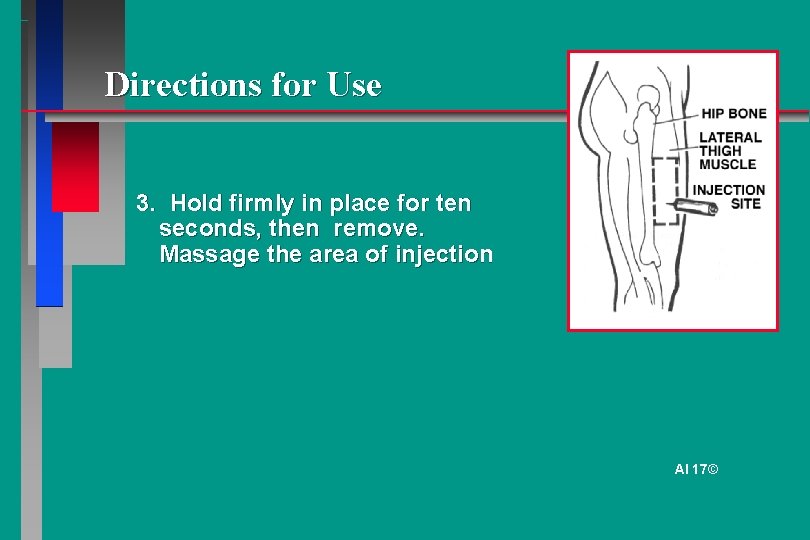
Directions for Use 3. Hold firmly in place for ten seconds, then remove. Massage the area of injection AI 17©

Directions for Use 4. After auto-injector has been activated, empty container should be disposed of properly • It cannot be refilled nor can the protruding needle be retracted • It should be disposed of in a “sharps” container per rules for handling medical wastes and possible blood-borne pathogens Note dosage on triage tag or write on chest or forehead of patient AI 18©

Directions for Use IMPORTANT: Medical personnel assisting evacuated victims of nerve agent exposure should avoid exposing themselves to crosscontamination by ensuring that they do not come in contact with patients’ clothing AI 19©

Precautions Place any patient receiving atropine under medical observation for at least 24 hours If atropine is used to treat infants, smaller amounts should be given AI 20©
Adverse Reactions 2 to 5 minutes after intramuscular injection of 2 -PAM Cl, mild to moderate pain may be experienced at site of injection AI 21©

Adverse Reactions 2 -PAM Cl in a normal person may cause: • blurred vision • double vision ( diplopia) • impaired accommodation • dizziness • headache • drowsiness • nausea • rapid heart rate (tachycardia) • increased systolic and • hyperventilation diastolic blood pressure These are relatively non-toxic responses when compared to effects of nerve agent or organophorus insecticide exposures AI 22©

Overdosage If excessive atropine is administered to a normal person: Signs of atropinization will become even more severe and patient may also develop • • • blurring of vision delirium urinary retention When signs and symptoms of atropinization develop, no more atropine should be injected until atropinization subsides AI 23©

Overdosage In case of 2 -PAM Cl: Observed in normal subjects only • • dizziness blurred vision double vision (diplopia) headache impaired accommodation nausea slightly rapid heart rate (tachycardia) Artificial respiration and other supportive therapy should be administered as needed AI 24©

Caring For Auto-Injector Supplies Auto-injectors have a five-year shelf life, making them suitable for storage in emergency stockpiles Store auto-injectors at room temperature (approximately 25°C, 77°F) Keep them from freezing May AI 25©

Caring For Auto-Injector Supplies Antidotes will freeze at temperatures below 29 F Do not carry auto-injectors in external pocket of BDO when temperature below freezing. Place in inner pocket where body heat will keep them warm Tie string to auto-injector and thread through outer layers of clothing to outside pocket or belt. Extract auto-injector by pulling the string when needed Frozen auto-injectors still usable after being thawed if they do not appear broken or cracked AI 26©

VAPOR EXPOSURE, MILD Mild exposure: Miosis, rhinorrhea, lacrimation VAPOR EXPOSURE TREATMENT • Mild exposure: may require decontamination only or atropine • Atropine (IM or IV) will not reverse Miosis (small pupils) or Dimness of Vision • Atropine Dosing: Starting at 1 -2 mg IM or IV • Mild Vapor exposures may only require Atropine • 2 -PAM Cl dosing 600 mg IM REMINDER: EACH MARK-1 HAS ATROPINE 2 MG AND 2 -PAM 600 MG

VAPOR EXPOSURE, MODERATE • Moderate exposure: Miosis, rhinorrhea, moderate dyspnea, GI Effects, increased secretions VAPOR EXPOSURE TREATMENT • Moderate exposure: Atropine-starting dosing 2 -6 mg IM first, then further IV/IM doses in 2 mg increments q 5 -10 min titration to secretion and ease of respiration • OR 1 to 2 MARK-ONE KITS • 2 -PAM-initial dosing of 600 IM or 500 -1000 mg slow IV in 0. 9% NS over 3060 min, if conditions allow IV access. Repeat dosing may be required (up to 3 grams, per medical control instructions. • WATCH FOR HYPERTENSION (responds to slowing or stopping infusion)

VAPOR EXPOSURE, SEVERE • Large exposure: Copious secretions, apnea, cardiovascular collapse, loss of consciousness possible, seizures, difficult to ventilate VAPOR EXPOSURE TREATMENT • Severe exposure: Ventilation and secure airway • Atropine-6 mg IM then further IV/IM doses in 2 mg increments q 5 -10 min titration to secretion and ease of ventilation. • 2 -PAM- If IV access obtainable 1000 mg over 30 mins. May need to be repeated. IM 1200 -1800 mg, if IM is only mode available • OR 3 MARK-ONE KITS • Benzodiazepine (diazepam or lorazepam) for seizure activity or post-ictal patient. DIAZEPAM 5 -10 MG IM OR SLOW IV. OR LORAZEPAM 2 -4 MG IM/IV.

LIQUID EXPOSURE: SYMPTOMS MAY BE DELAYED AS LONG AS 18 HOUR FOR MILD LIQUID EXPOSURES, EXPECIALLY FOR VX Mild exposure: Miosis, rhinorrhea, lacrimation Also may see Local Sweating, Fasciculations from small exposures Volatization is also possible, giving vapor exposure symptoms LIQUID EXPOSURE TREATMENT • Mild exposure: may require decontamination only or atropine • Atropine (IM or IV) will not reverse Miosis (small pupils) or Dimness of Vision • Atropine Dosing: Starting at 1 -2 mg IM or IV • Mild Vapor exposures may only require Atropine • 2 -PAM Cl dosing 600 mg IM

LIQUID EXPOSURE, Moderate • Moderate exposure: Up to 18 hr. after exposure. Nausea, Vomiting, Diarrhea, Localized Sweating, Muscle Fasciculations LIQUIDEXPOSURE TREATMENT • Moderate exposure: Atropine-starting dosing 2 -6 mg IM first, then further IV/IM doses in 2 mg increments q 5 -10 min titration to secretion and ease of respiration • OR 1 to 2 MARK-ONE KITS • 2 -PAM-initial dosing of 600 IM or 500 -1000 mg slow IV in 0. 9% NS over 3060 min, if conditions allow IV access. Repeat dosing may be required (up to 3 grams, per medical control instructions. • WATCH FOR HYPERTENSION (responds to slowing or stopping infusion)

• Large exposure: Copious secretions, apnea, cardiovascular collapse, loss of consciousness possible, seizures, difficult to ventilate LIQUID EXPOSURE TREATMENT • Severe exposure: Ventilation and secure airway • Atropine-6 mg IM then further IV/IM doses in 2 mg increments q 5 -10 min titration to secretion and ease of ventilation. • 2 -PAM- If IV access obtainable 1000 mg over 30 mins. May need to be repeated. IM 1200 -1800 mg, if IM is only mode available • OR 3 MARK-ONE KITS • Benzodiazepine (diazepam or lorazepam) for seizure activity or post-ictal patient. DIAZEPAM 5 -10 MG IM OR SLOW IV. OR LORAZEPAM 2 -4 MG IM/IV.

Mixing IM/IV Pralidoxime from 1 gram Vial Intramuscular solution from 1 gram vial of pralidoxime (2 -PAM). 3 ml of sterile water or normal saline for a concentration of 300 mg/ml. Intravenous: 1 gram vial of pralidoxime (2 -PAM) diluted with 20 ml of sterile water or normal saline. Add to 100 ml IV bag of normal saline. Adult dosing is 1 gram infused over at least 30 minutes in the cases of severe poisoning. More rapid dosing is associated with hypertension. For moderate poisoning 1 gram over 60 minutes for infusion. (1 -4) Very rapid infusions has been associated with reports of Respiratory Arrest. In elderly, hypertensive or those with renal insufficiency the dose is 7. 5 mg/kg IV, slow infusion if hypertension develops.
- Slides: 33