Ultraviolet Photodissociation Dynamics of the 3 Cyclohexenyl Radical




- Slides: 15

Ultraviolet Photodissociation Dynamics of the 3 -Cyclohexenyl Radical Michael Lucas, Yanlin Liu, Jasmine Minor, Raquel Bryant, Jingsong Zhang Department of Chemistry University of California, Riverside 69 th International Symposium on Molecular Spectroscopy 6/17/2014

Cyclohexyenl Radical Cycloalkanes are important component of conventional fuels Cyclohexane model cycloalkane �Major producer of benzene Previous Research: cyclohexyl, phenyl What effect does the double bond have on the photochemistry?

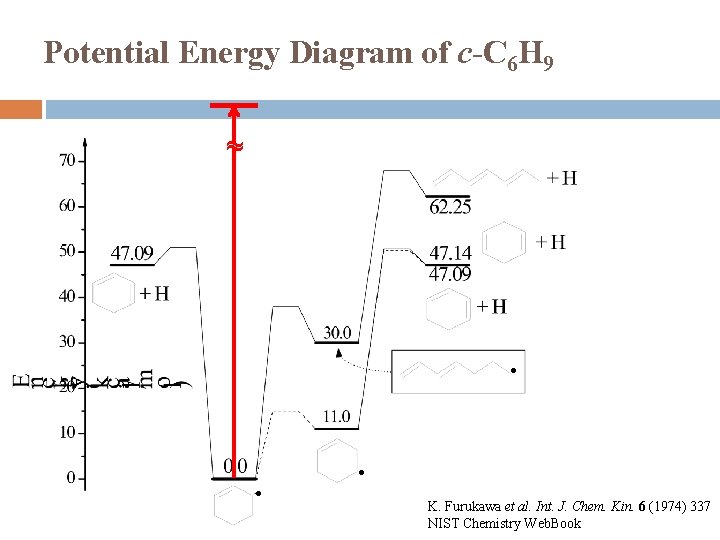
Potential Energy Diagram of c-C 6 H 9 ~ ● ● ● K. Furukawa et al. Int. J. Chem. Kin. 6 (1974) 337 NIST Chemistry Web. Book

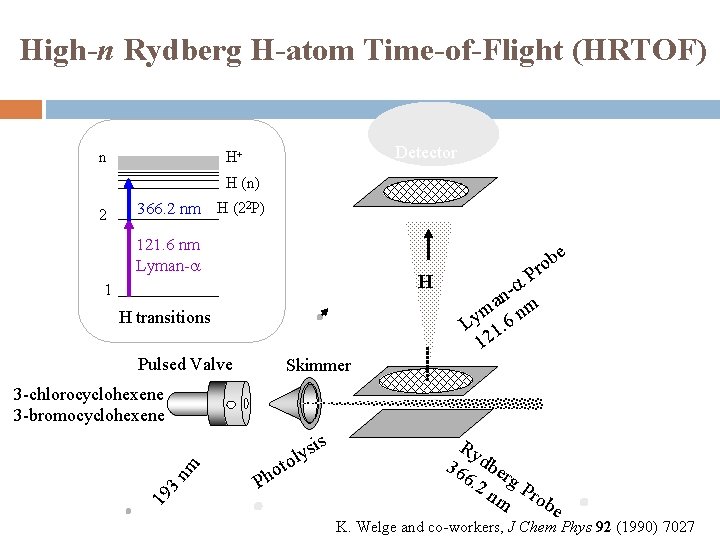
High-n Rydberg H-atom Time-of-Flight (HRTOF) n Detector H+ H (n) 2 366. 2 nm H (22 P) 121. 6 nm Lyman-a e H 1 H transitions Pulsed Valve ob r P -a n ma nm y L 1. 6 12 Skimmer 19 3 nm 3 -chlorocyclohexene 3 -bromocyclohexene o Ph to s i s ly Ry 36 dber 6. 2 g nm Prob e K. Welge and co-workers, J Chem Phys 92 (1990) 7027

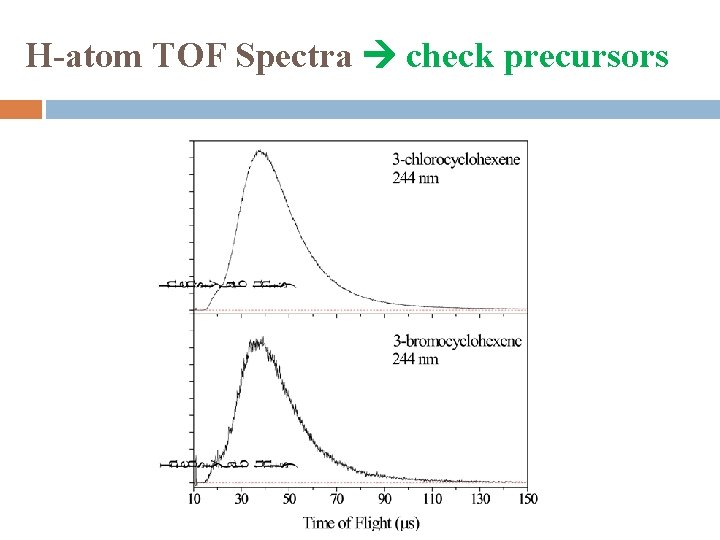
H-atom TOF Spectra check precursors

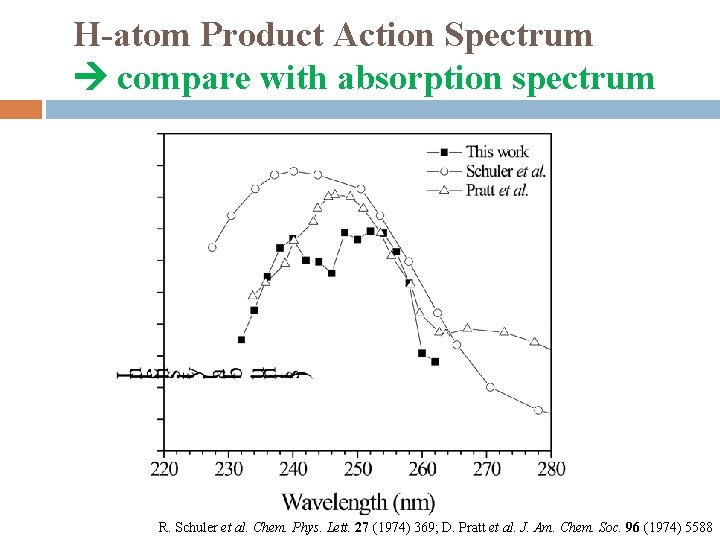
H-atom Product Action Spectrum compare with absorption spectrum R. Schuler et al. Chem. Phys. Lett. 27 (1974) 369; D. Pratt et al. J. Am. Chem. Soc. 96 (1974) 5588

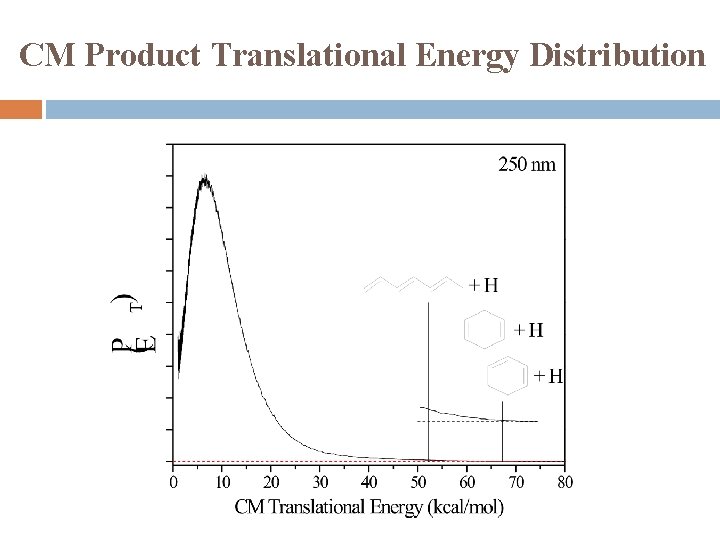
CM Product Translational Energy Distribution

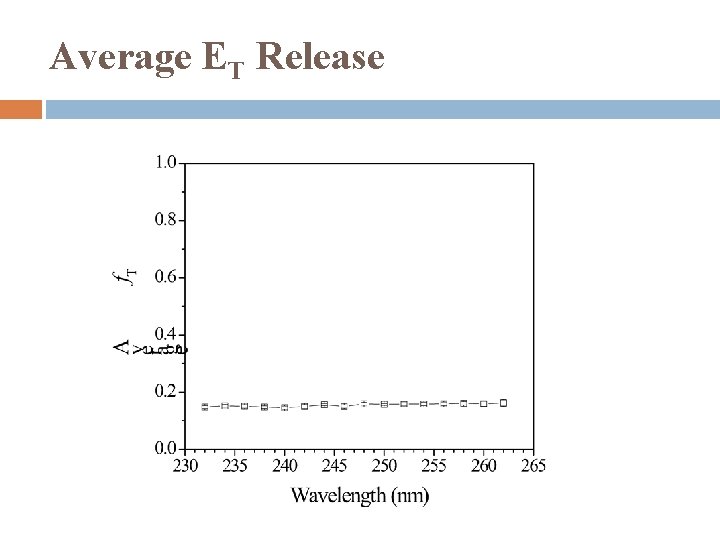
Average ET Release

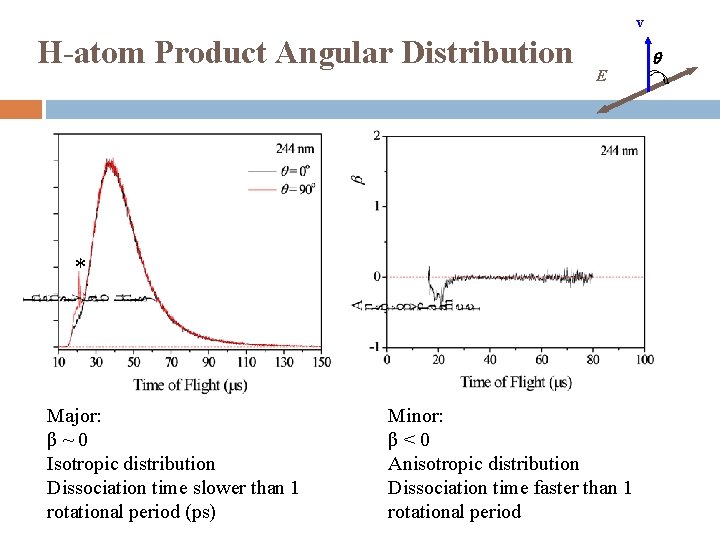
v H-atom Product Angular Distribution q E * Major: β~0 Isotropic distribution Dissociation time slower than 1 rotational period (ps) Minor: β<0 Anisotropic distribution Dissociation time faster than 1 rotational period

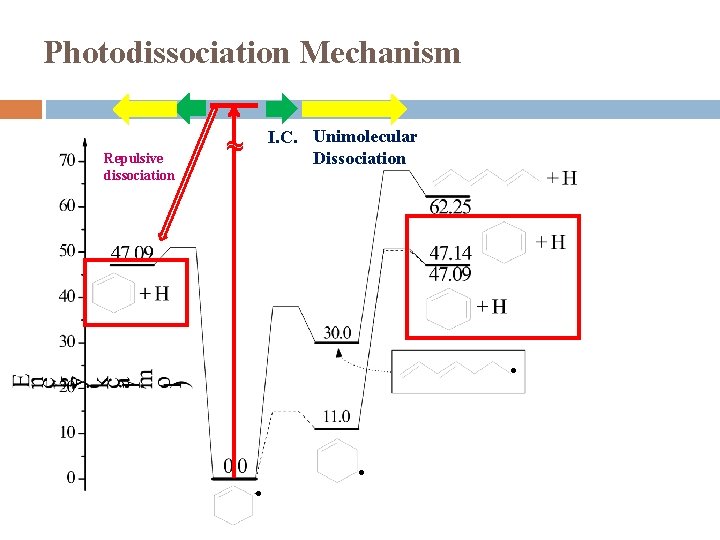
Photodissociation Mechanism Repulsive dissociation I. C. Unimolecular Dissociation ~ ● ● ●

Summary UV photodissociation dynamics of cyclohexenyl was studied in 232 -262 nm for the first time Observed: cyclohexenyl → cyclohexadiene + H Modest translational energy release, f. T ~ 0. 15 Two components � Major: Isotropic distribution, β ~ 0 Dissociation mechanism: internal conversion from excited electronic state followed by unimolecular dissociation on ground electronic state � Minor: Anisotropic distribution, β < 0 Dissociation mechanism: direct dissociation from excite state or repulsive part of ground state

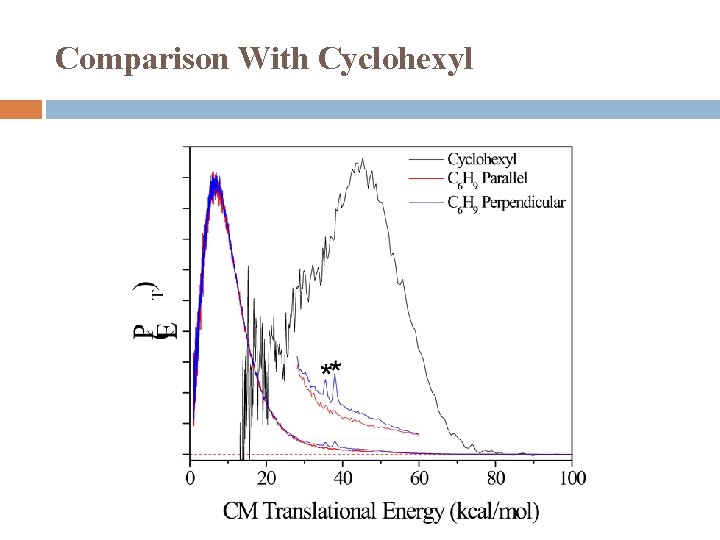
Comparison With Cyclohexyl

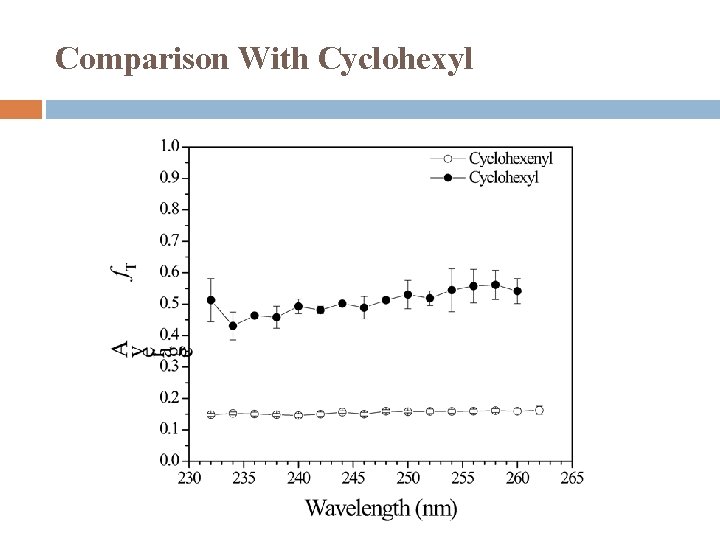
Comparison With Cyclohexyl

Comparison With Cyclohexyl Cyclohexenyl Moderate translational energy release Two component Major � Isotropic distribution � Statistical distribution � Hot radical dissociation mechanism Minor � Anisotropic distribution, β < 0 � Repulsive dissociation mechanism Cyclohexyl Large translational energy release Anisotropic distribution, β > 0 Non-statistical distribution Dissociation mechanism: direct dissociation from the excited state and/or on the repulsive part of the ground state (possibly via conical intersection).

Acknowledgements Prof. Jingsong Zhang Yanlin Liu Jasmine Minor Raquel Bryant Zhang Group