Chapter 6 Electromagnetic Radiation wavelength Visible light Amplitude
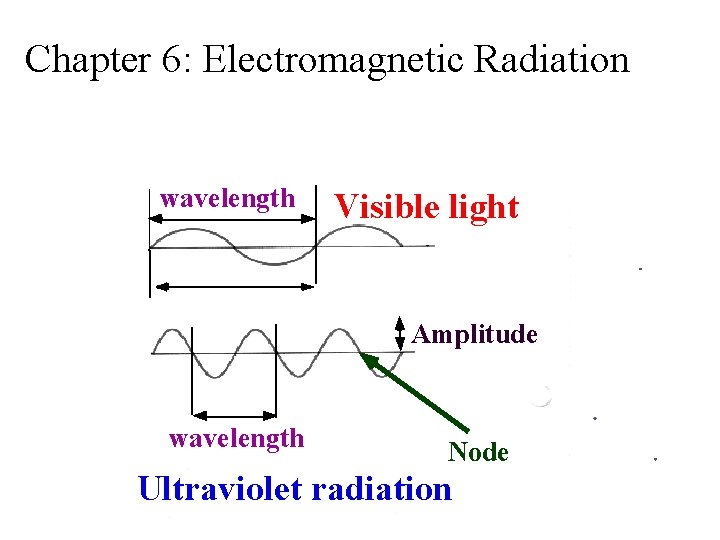
Chapter 6: Electromagnetic Radiation wavelength Visible light Amplitude wavelength Node Ultraviolet radiation
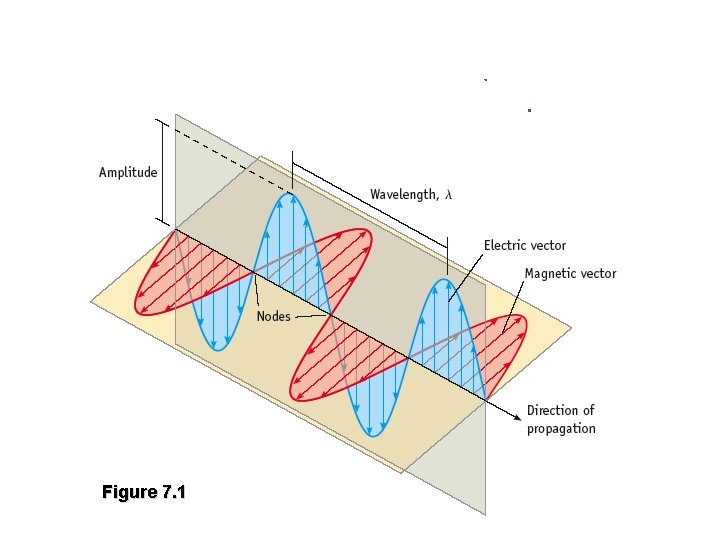
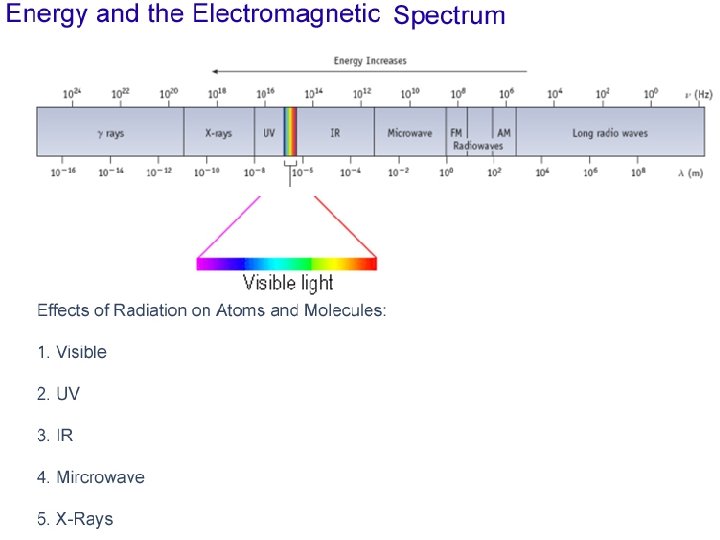
Figure 7. 1
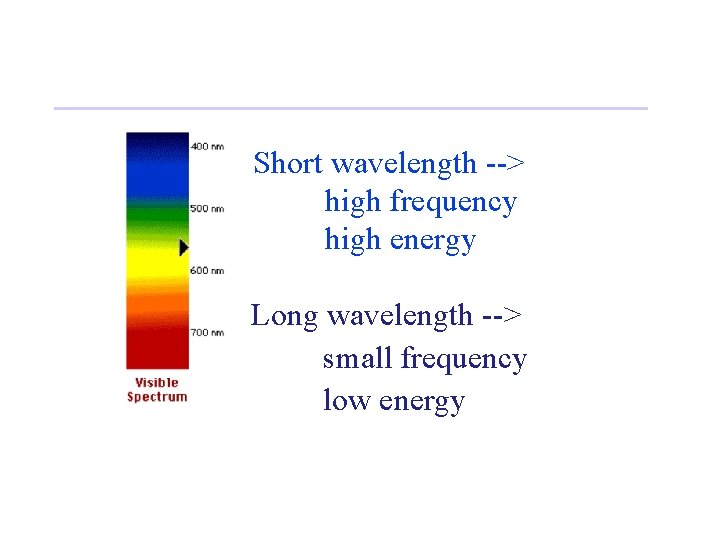
Short wavelength --> high frequency high energy Long wavelength --> small frequency low energy
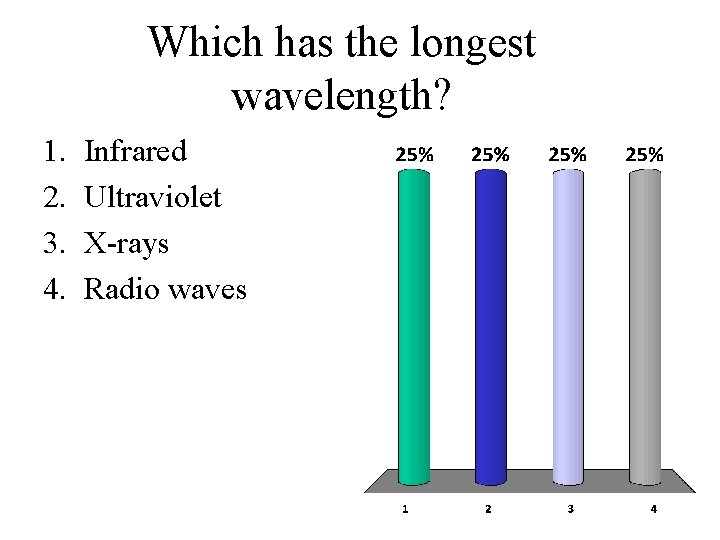
Which has the longest wavelength? 1. 2. 3. 4. Infrared Ultraviolet X-rays Radio waves

Rank the following in order of increasing frequency: microwaves radiowaves X-rays blue light red light UV light IR light

• Waves have a frequency • Use the Greek letter “nu”, , for frequency, and units are “cycles per sec” = c • All radiation: • • c = velocity of light = 3. 00 x 108 m/sec • Long wavelength --> small frequency • Short wavelength --> high frequency

What is the wavelength of WONY? What is the wavelength of cell phone radiation? Frequency = 850 MHz What is the wavelength of a microwave oven? Frequency = 2. 45 GHz

Quantization of Energy Light acts as if it consists of particles called PHOTONS, with discrete energy. Energy of radiation is proportional to frequency E = h • h = Planck’s constant = 6. 6262 x 10 -34 J • s

E = h • Relationships:
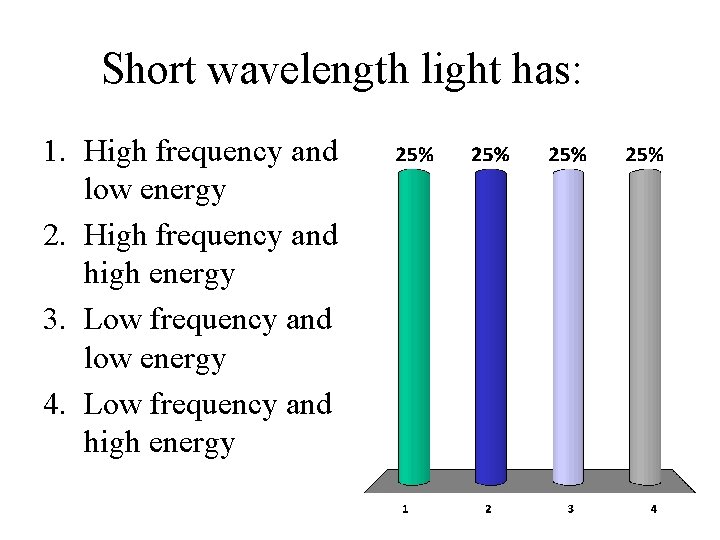
Short wavelength light has: 1. High frequency and low energy 2. High frequency and high energy 3. Low frequency and low energy 4. Low frequency and high energy

Rank the following in order of increasing photon energy: microwaves radiowaves X-rays blue light red light UV light IR light

E = h • What is the energy of a WONY photon?

Energy of Radiation What is the frequency of UV light with a wavelength of 230 nm? What is the energy of 1 photon of UV light with wavelength = 230 nm?
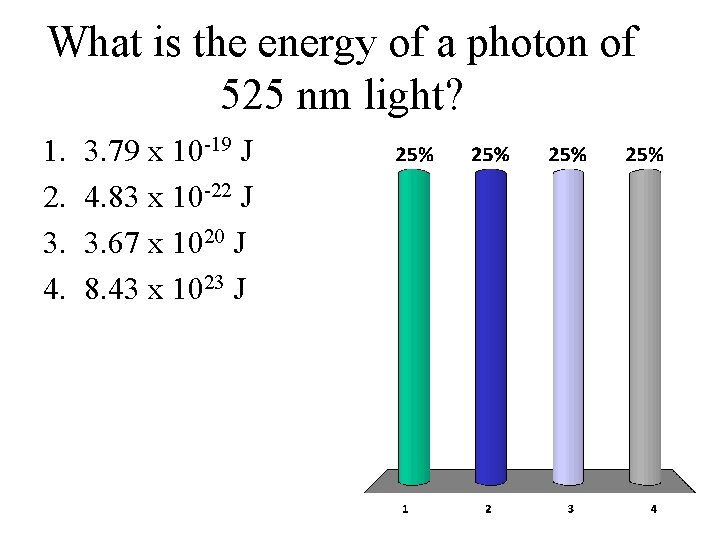
What is the energy of a photon of 525 nm light? 1. 2. 3. 4. 3. 79 x 10 -19 J 4. 83 x 10 -22 J 3. 67 x 1020 J 8. 43 x 1023 J

What is the energy of a mole of 230 nm photons? Can this light break C-C bonds with an energy of 346 k. J/mol?

Does 1200 nm light have enough energy to break C-C bonds?

Where does light come from? • Excited solids emit a continuous spectrum of light • Excited gas-phase atoms emit only specific wavelengths of light (“lines”)
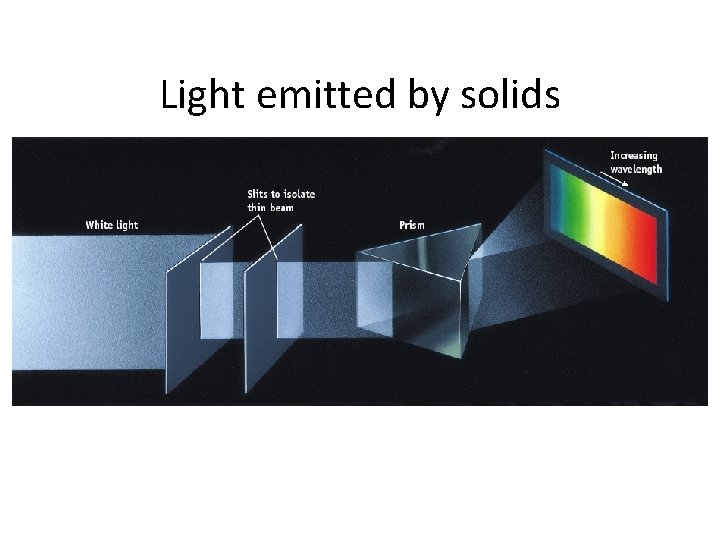
Light emitted by solids
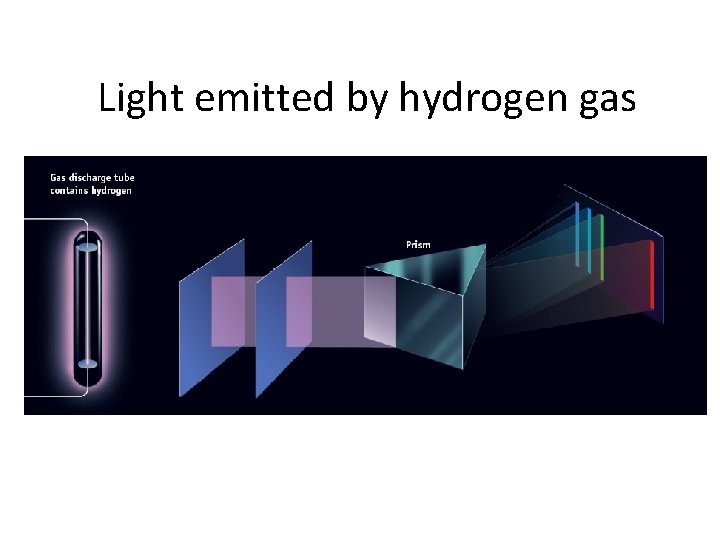
Light emitted by hydrogen gas
- Slides: 21