Physics at the end of XIX Century and





- Slides: 8

Physics at the end of XIX Century and Major Discoveries of XX Century Thompson’s experiment (discovery of electron) Emission and absorption of light Spectra: • Continues spectra • Line spectra Three problems: • “Ultraviolet catastrophe” • Photoelectric effect • Michelson experiment 1

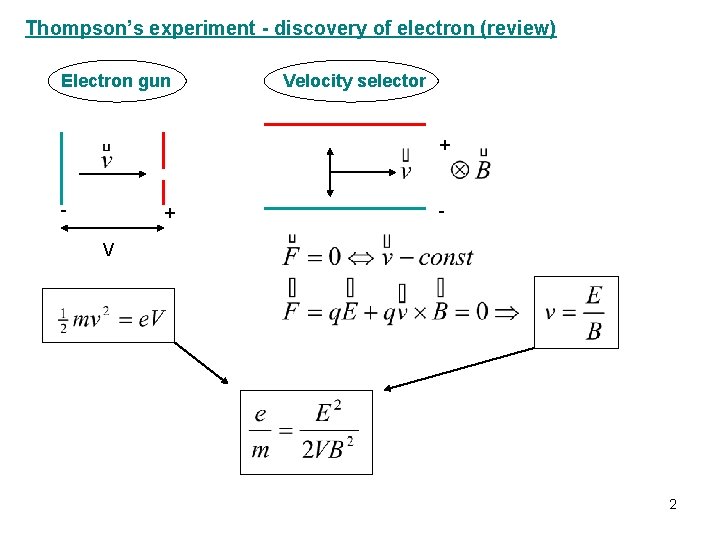
Thompson’s experiment - discovery of electron (review) Electron gun Velocity selector + - V 2

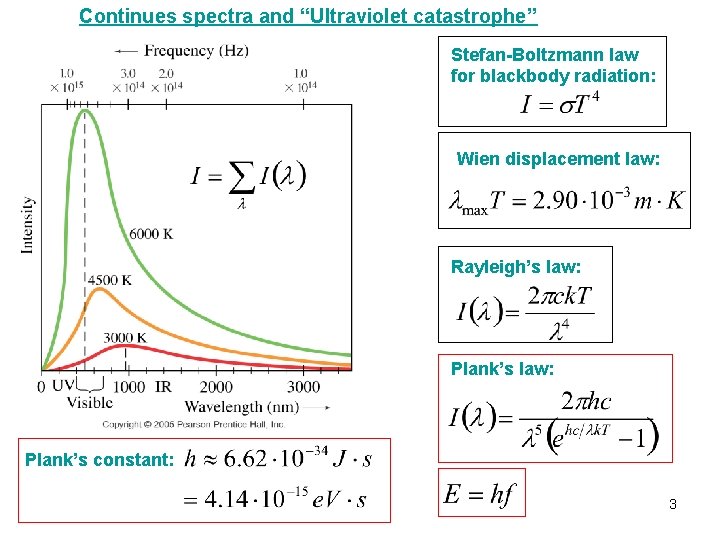
Continues spectra and “Ultraviolet catastrophe” Stefan-Boltzmann law for blackbody radiation: Wien displacement law: Rayleigh’s law: Plank’s constant: 3

Example 1: What are the wavelength and the frequency corresponding to the most intense light emitted by a giant star of surface temperature 5000 K? Example 2: What are the wavelength and the frequency of the most intense radiation from an object with temperature 100°C? 4

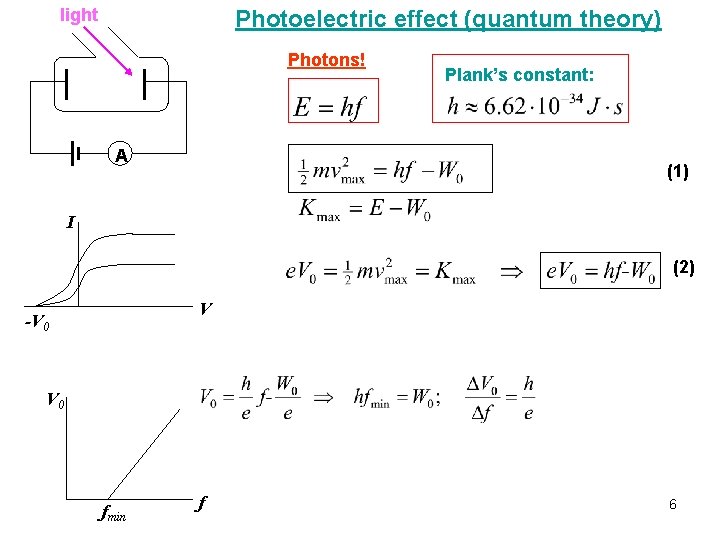
Photoelectric effect Experiment: If light strikes a metal, electrons are emitted. • the effect does not occur if the frequency of the light is too low • the kinetic energy of the electrons increases with frequency light Classical theory can not explain these results. If light is a wave, classical theory predicts: • Frequency would not matter • Number of electrons and their energy should increase with intensity A Quantum theory: Einstein suggested that, given the success of Planck’s theory, light must be emitted and absorbed in small energy packets, “photons” with energy: If light is particles, theory predicts: • Increasing intensity increases number of electrons but not energy • Above a minimum energy required to break atomic bond, kinetic energy of electrons will increase linearly with frequency • There is a cutoff frequency below which no electrons will be emitted, regardless of intensity 5

light Photoelectric effect (quantum theory) Photons! A Plank’s constant: (1) I (2) V -V 0 fmin f 6

Example: The work function for a certain sample is 2. 3 e. V. What is the stopping potential for electrons ejected from the sample by 7. 0*1014 Hz electromagnetic radiation? Example: The work function for sodium, cesium, copper, and iron are 2. 3, 2. 1, 4. 7, and 4. 5 e. V respectively. Which of these metals will not emit electrons when visible light shines on it? Copper, and iron will not emit electrons 7

Example: Rank the following radiations according to their associated photon energies, greatest first: (a) yellow light from a sodium vapor lamp (b) a gamma ray emitted by a radioactive (c) a radio wave emitted the antenna of a commercial radio station (d) a microwave beam emitted by airport traffic control radar Example: At what rate are photons emitted by a 100 W sodium vapor lamp if we can assume the emission is entirely at a wave-length of 590 nm? 8