The Stratospheric Chemistry and The Ozone Layer Stratospheric















- Slides: 36

The Stratospheric Chemistry and The Ozone Layer *Stratospheric circulation *Stratospheric photochemistry *Gas phase reactions *Ozone and Catalytic cycles Dr Tony Cox ERCA 2004 -Lecture 2


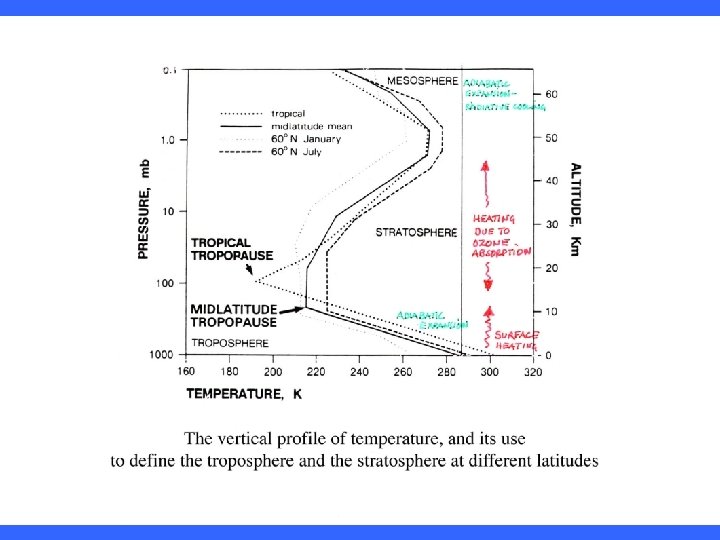
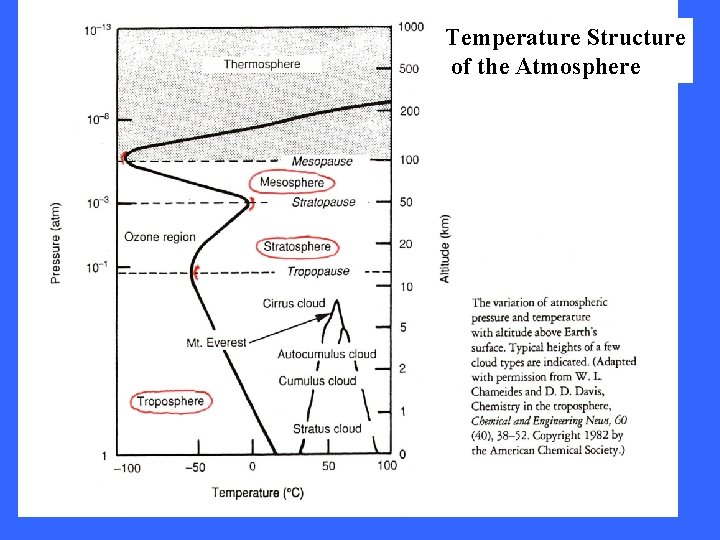
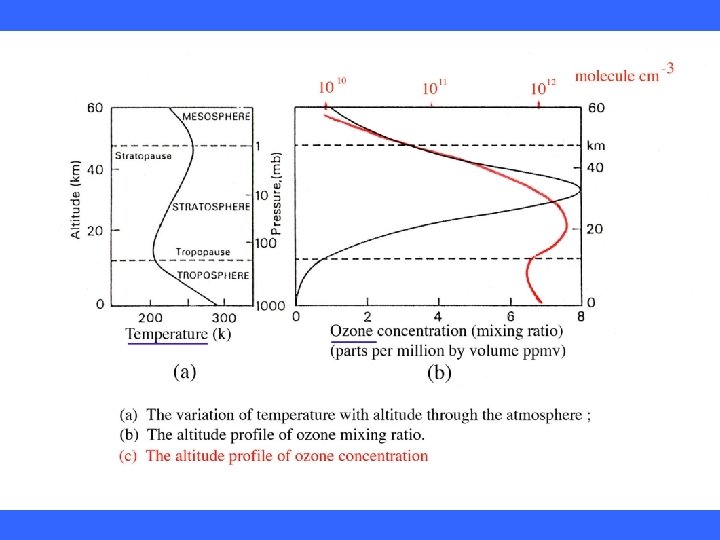
Temperature Structure of the Atmosphere

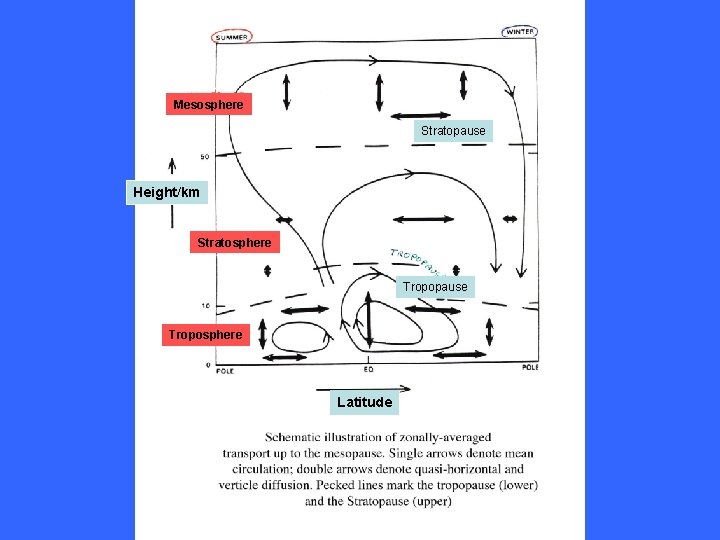
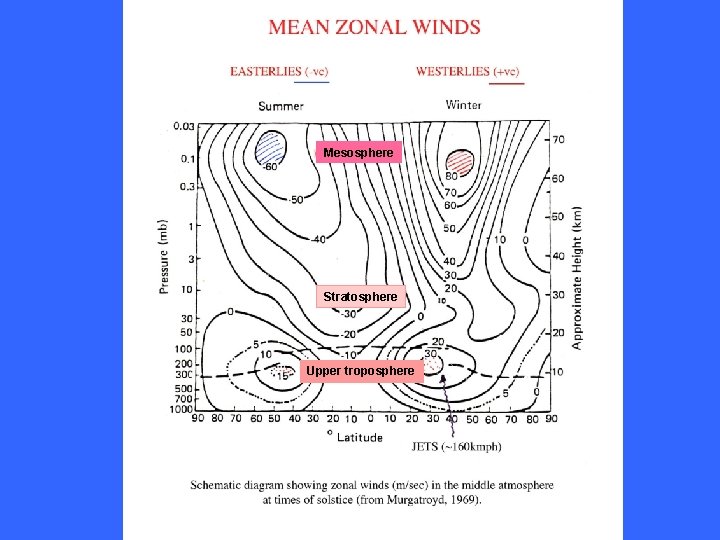
Mesosphere Stratopause Height/km Stratosphere Tropopause Troposphere Latitude

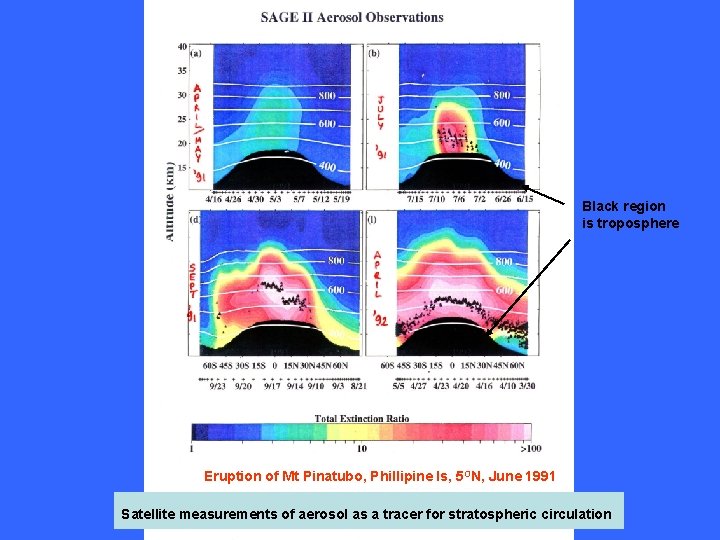
Black region is troposphere Eruption of Mt Pinatubo, Phillipine Is, 5 ON, June 1991 Satellite measurements of aerosol as a tracer for stratospheric circulation

Mesosphere Stratosphere Upper troposphere


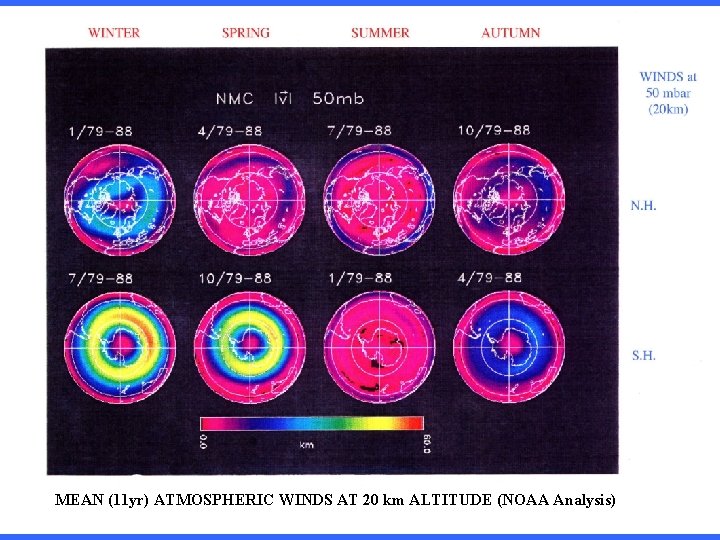
MEAN (11 yr) ATMOSPHERIC WINDS AT 20 km ALTITUDE (NOAA Analysis)

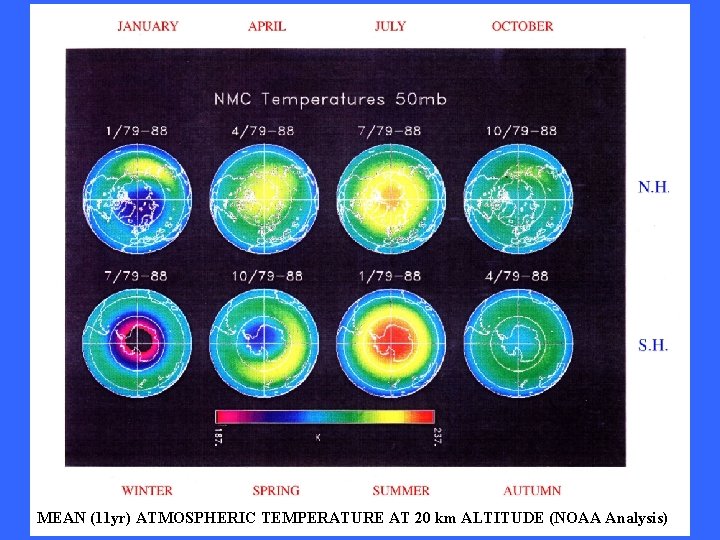
MEAN (11 yr) ATMOSPHERIC TEMPERATURE AT 20 km ALTITUDE (NOAA Analysis)

Potential Vorticity At ~20 km in the S. Polar Vortex

30 km Altitude 1950 16 km The QBO In equatorial regions 1978

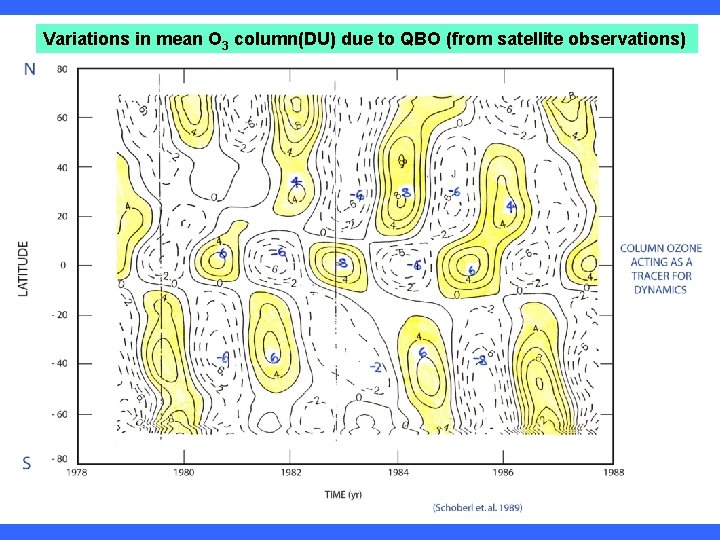
Variations in mean O 3 column(DU) due to QBO (from satellite observations)

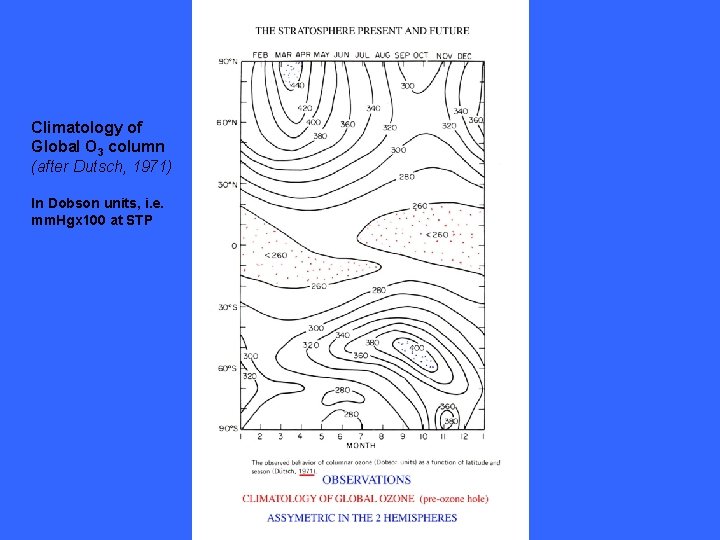
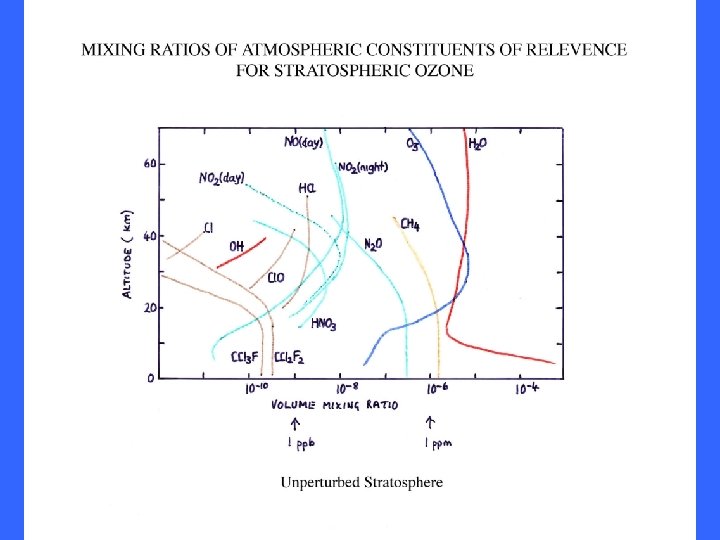
Climatology of Global O 3 column (after Dutsch, 1971) In Dobson units, i. e. mm. Hgx 100 at STP

Chemical Kinetics I

Chemical Kinetics II

Chemical Kinetics III

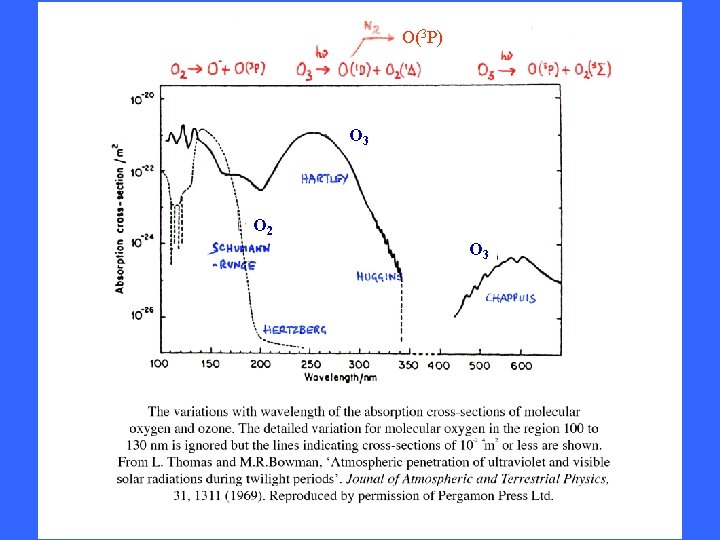
O(3 P) O 3 O 2 O 3

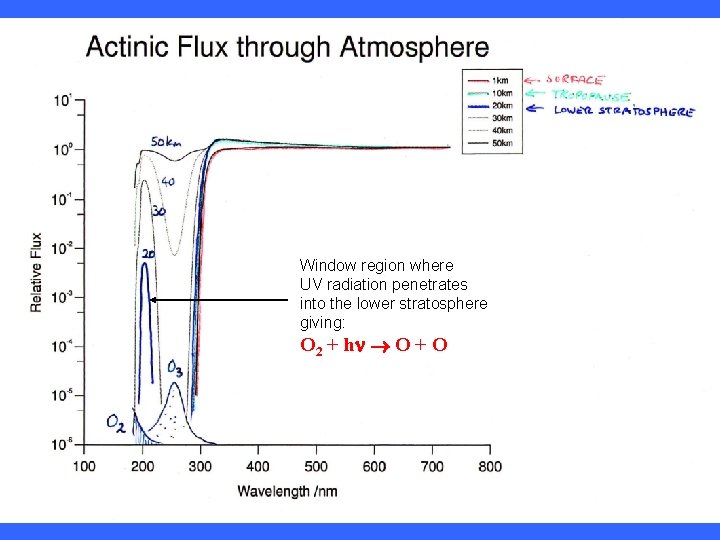
Window region where UV radiation penetrates into the lower stratosphere giving: O 2 + hn O + O

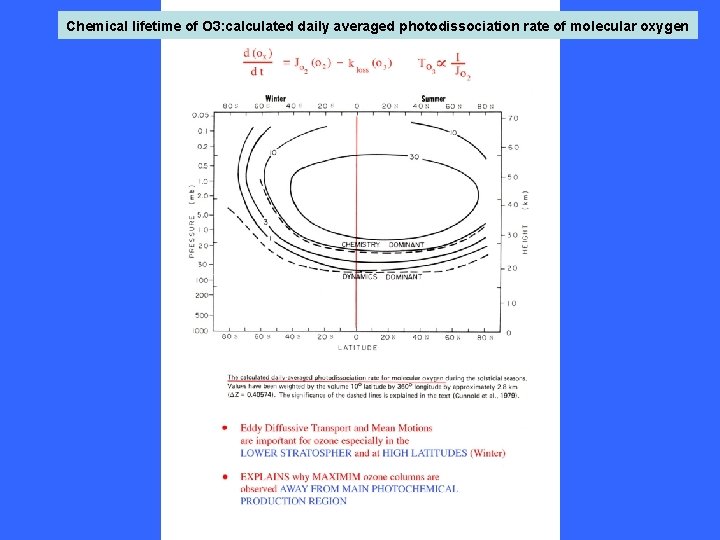
Chemical lifetime of O 3: calculated daily averaged photodissociation rate of molecular oxygen

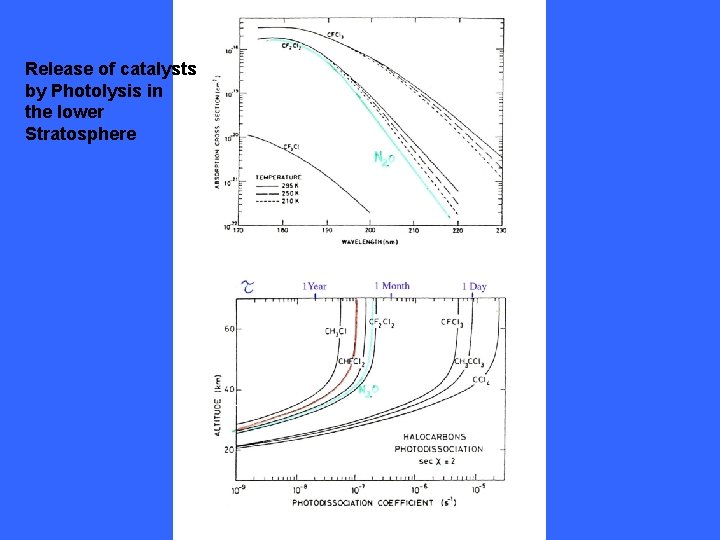
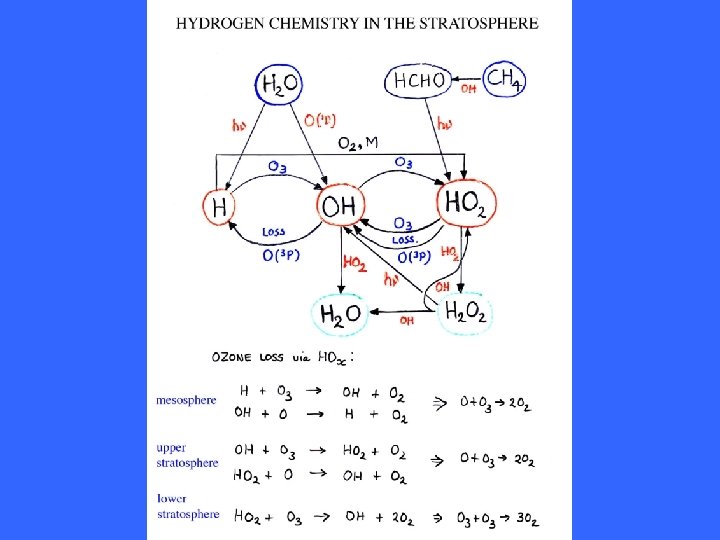
Release of catalysts by Photolysis in the lower Stratosphere

O 2 + hn(l<243 nm) O + O oxygen photolysis O + O 2 + M O 3 + M ozone formation O + O 3 O 2 + O 2 ozone destruction O 3 + hn(l<310 nm) O(1 D) + O 2 O 3 + hn(l<1180 nm) O + O 2 oxygen photolysis NO 2 + hn(l<243 nm) NO + O 2 + M O 3 + M Net X + O 3 XO + O 2 XO + O X + O 2 O + O 3 O 2 + O 2 [X = H, OH, NO, Cl, Br, etc] NO 2 photolysis ozone formation ozone destruction






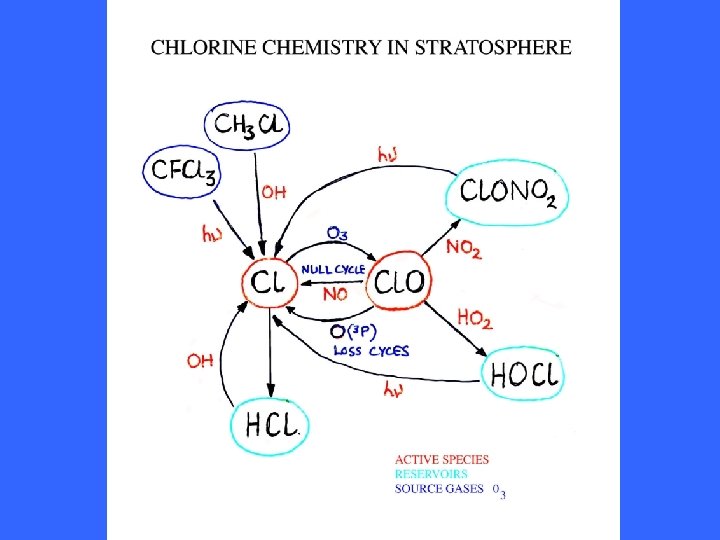
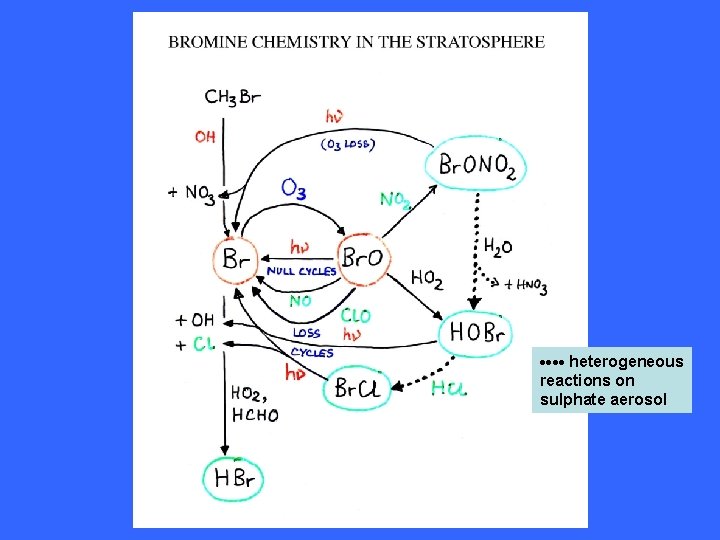
Interconversion of Cl & Cl. O Formation and release from reservoirs of Cl & Cl. O

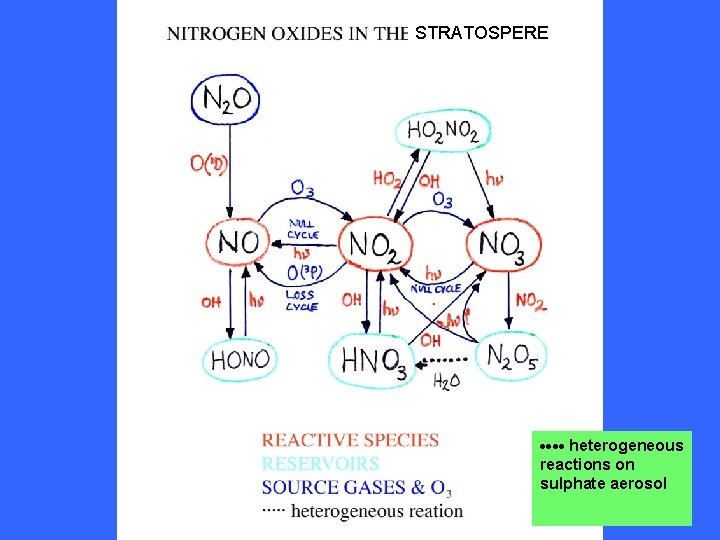
STRATOSPERE heterogeneous reactions on sulphate aerosol





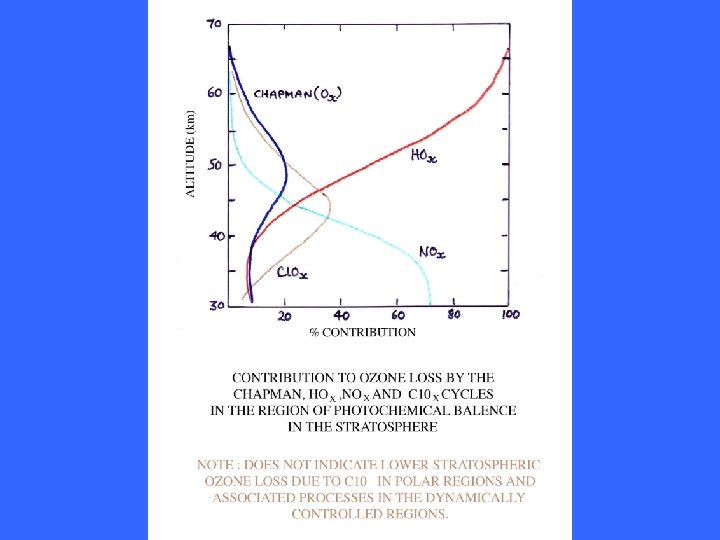
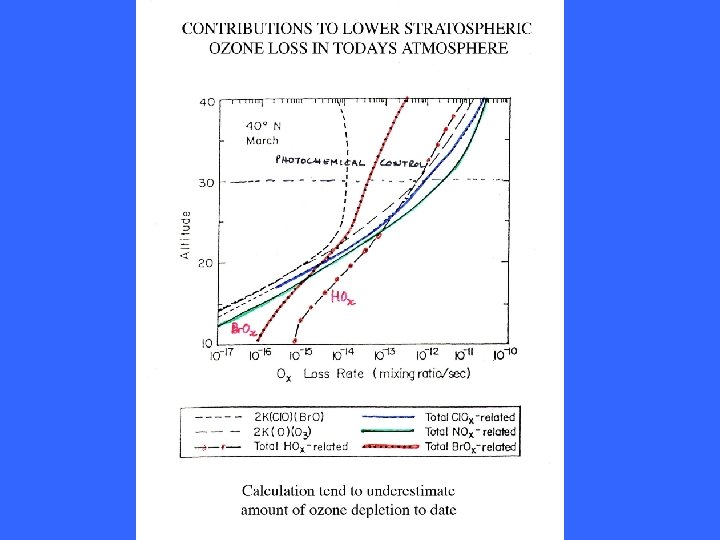
OZONE DEPLETION CYCLES IN THE LOWER STRATOSPHERE Concentration of O atoms fall off in lower stratosphere as photolysis of O 2 and O 3 reduces. Catalytic destruction through cycles involving participation of atomic O is less important. Other cycles not involving O(3 P) can occur.

OZONE DEPLETION CYCLES INVOLVING HALOGENS THE LOWER STRATOSPHERE Involving Cl. Ox/NOx coupling Cl. O + NO 2 = Cl. ONO 2 + hn = Cl + NO 3 + hn = NO + O 2 NO + O 3 = NO 2 + O 2 Cl + O 3 = Cl. O + O 2 net O 3 + O 3 = 3 O 2 Involving NOx/Halogen (X = Br or I) coupling XO + HO 2 = HOX + O 2 HOX + hn = X + HO OH + O 3 = HO 2 + O 2 X + O 3 = XO + O 2 net O 3 + O 3 = 3 O 2

heterogeneous reactions on sulphate aerosol
