The Increasing Use of Targeted Therapies for Leukemia












- Slides: 20

The Increasing Use of Targeted Therapies for Leukemia and Lymphoma Shira Dinner, MD Northwestern University

Educational Objectives • Define terminology used regarding leukemia • Describe the latest developments in the treatment of acute lymphoblastic leukemia (ALL) - Blinatumomab - Inotuzumab ozogamicin - Chimeric antigen receptor T cells • Engage patients and caregivers in clinical trial discussions on emerging therapies in ALL • Identify strategies fro optimal patient care - What is the best treatment for me?

Common Terminology in Leukemia • Acute: - Develops from immature cells in the bone marrow that replicate quickly and aggressively - The cells do not grow up and perform their normal function (i. e. fight infection) • Chronic: - Develops from mature cells that do not function normally - Typically progresses more slowly • Acute lymphoblastic leukemia: - An aggressive cancer that develops from a type of white blood cells in the bone marrow called lymphocytes

Common Terminology in Leukemia • Complete remission (CR) - The absence of any signs or symptoms of the disease - < 5% blast cells in the bone marrow (when looking at the cells under a microscope) and no evidence of leukemia anywhere outside of the bone marrow • Minimal residual disease (MRD) - A very low level of leukemia cells that remain in the bone marrow that cannot be seen by the microscope - Detected by looking for certain gene or chromosome mutations that may be present in the leukemia or by looking at antigens (proteins) on the surface of the leukemia cells • Refractory disease - The leukemia does not respond to treatment or does not achieve a CR • Relapsed disease - The leukemia was in CR, but has now come back

Common Terminology in Leukemia • Overall survival (OS) - The length of time from either the date of diagnosis or the start of treatment for a disease that patients diagnosed are still alive. - In a clinical trial, measuring the overall survival is one way to see how well a new treatment works. • Progression free survival - The length of time during and after the treatment of a disease that the disease remains in remission or does not get worse.

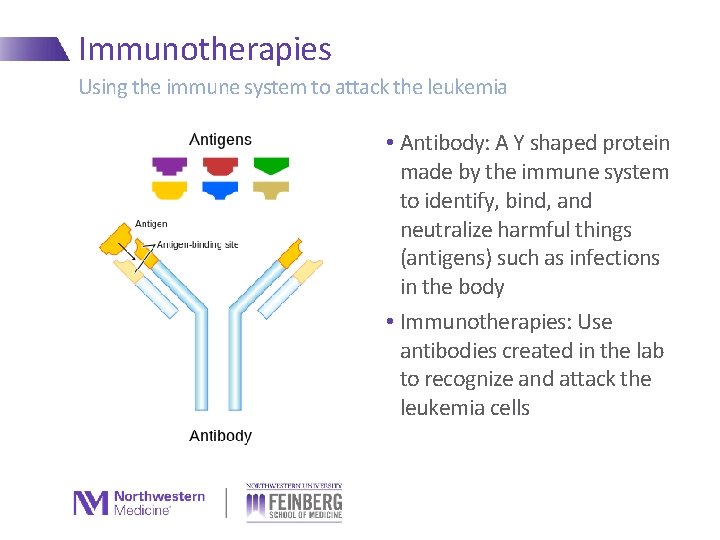
Immunotherapies Using the immune system to attack the leukemia • Antibody: A Y shaped protein made by the immune system to identify, bind, and neutralize harmful things (antigens) such as infections in the body • Immunotherapies: Use antibodies created in the lab to recognize and attack the leukemia cells

Antigens in ALL • There are many different proteins or antigens on the surface of ALL cells that we can design antibodies to target as part of immunotherapies

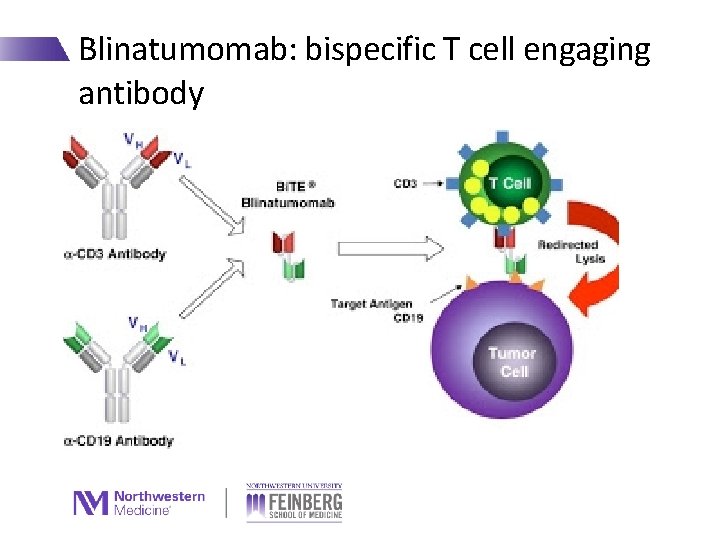
Blinatumomab: bispecific T cell engaging antibody

Phase 2 Blinatumomab in Relapsed/Refractory ALL Pts (%) CR/CRi during first 2 cycles, n (%) CR CRi Allogeneic SCT after CR/CRi Median Overall Survival 81/189 (43%) 63/189 (33%) 18/189 (10%) 32 of 81 (40%) 6. 1 months 80% of patients had received at least 1 prior chemotherapy to treat relapsed or refractory ALL Topp et al. Lancet Oncology 2015; 16(1): 57 -66.

Blinatumomab: Side effects • • Most Common side effects − Fever − Headache − Neutropenic fever 113 (60%) 65 (34%) 53 (28%) Neurologic side effects occurred in 98 (52%) patients, all reversible • This could include confusion or delirium to rarely stroke or seizure symptoms − Most were mild and resolved spontaneously 74 (76%) − Most occurred during Cycle 1 85 (87%) • 29 pts discontinued for neuro side effects Topp et al. Lancet Oncology 2015; 16(1): 57 -66. Topp et al. Lancet Oncology Jan; 16(1): 57 -66.

Inotuzumab Ozogamicin (IO): antibody-drug conjugate

Phase 3 IO vs Standard of Care (SOC) chemotherapy in relapsed/refractory ALL Baseline characteristic INO (n=109) SOC (n=109) CR 1 duration ≥ 12 mo 43% 35% Age < 55 yo 61% 63% Salvage 1 67% 63% Safety The most common serious side effects were low blood counts Serious liver or gallbladder side effects occurred in 9% INO vs 3% SOC Veno-occlusive liver disease (VOD) 14% INO vs 1% SOC VOD: blockage in small veins in the liver that can cause fluid Retention and increased liver function tests (esp bilirubin) INO: 5 VOD events during treatment, 10 post SCT (2 fatal) De. Angelo et al. EHA 2015. Abstract 2073

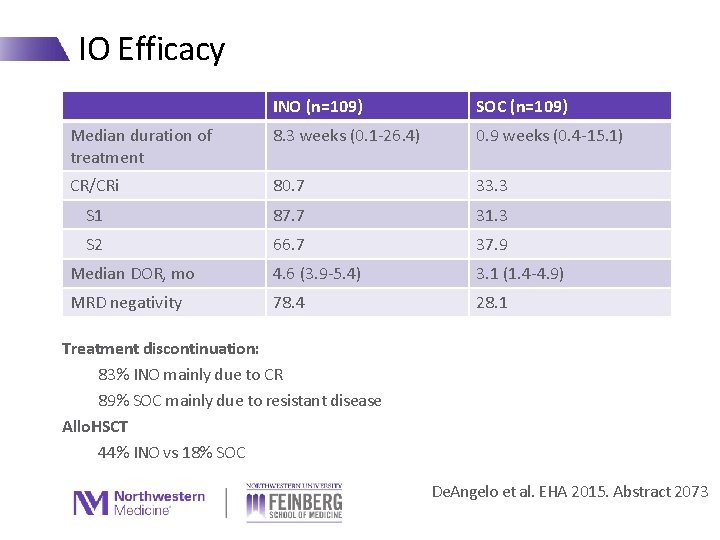
IO Efficacy INO (n=109) SOC (n=109) Median duration of treatment 8. 3 weeks (0. 1 -26. 4) 0. 9 weeks (0. 4 -15. 1) CR/CRi 80. 7 33. 3 S 1 87. 7 31. 3 S 2 66. 7 37. 9 Median DOR, mo 4. 6 (3. 9 -5. 4) 3. 1 (1. 4 -4. 9) MRD negativity 78. 4 28. 1 Treatment discontinuation: 83% INO mainly due to CR 89% SOC mainly due to resistant disease Allo. HSCT 44% INO vs 18% SOC De. Angelo et al. EHA 2015. Abstract 2073

Chimeric Antigen Receptor T Cells

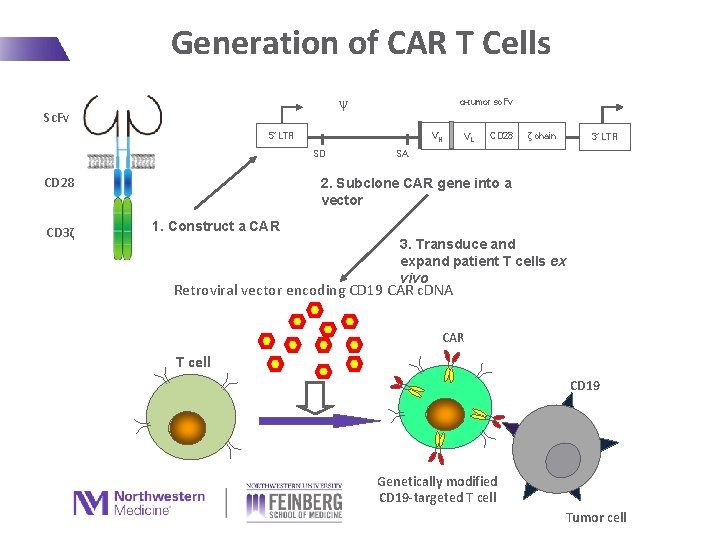
Generation of CAR T Cells ψ Sc. Fv α-tumor sc. Fv 5’ LTR VH SD CD 28 CD 3ζ VL CD 28 ζ chain 3’ LTR SA 2. Subclone CAR gene into a vector 1. Construct a CAR 3. Transduce and expand patient T cells ex vivo Retroviral vector encoding CD 19 CAR c. DNA CAR T cell CD 19 Genetically modified CD 19 -targeted T cell Tumor cell

Common Side Effects • Cytokine release syndrome (CRS) - Fever - Hypotension - Respiratory insufficiency • Neurological changes or symptoms - Delirium/disorientation Global encephalopathy (tremors, seizure-like jerks) Aphasia Seizure • Can occur within 1 -14 days of T cell infusion - Patients must be monitored in the hospital • More severe symptoms in patients with higher leukemia disease burden

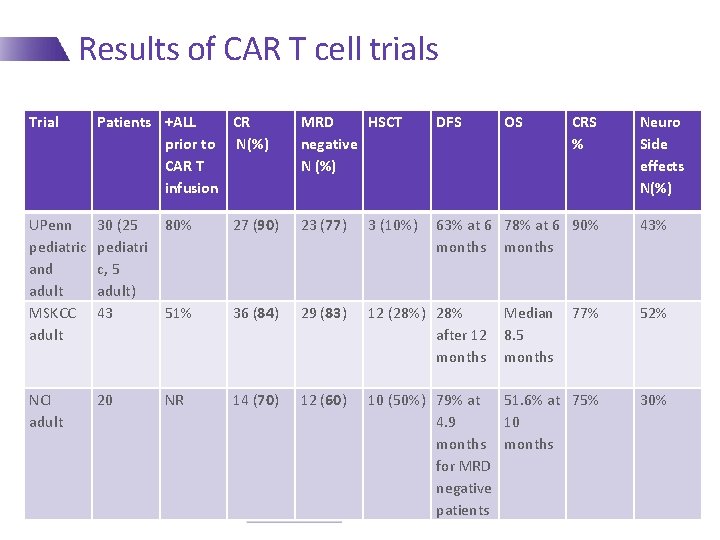
Results of CAR T cell trials Trial Patients +ALL CR prior to N(%) CAR T infusion MRD HSCT negative N (%) DFS OS UPenn pediatric and adult MSKCC adult 30 (25 pediatri c, 5 adult) 43 80% 27 (90) 23 (77) 3 (10%) 63% at 6 78% at 6 90% months 51% 36 (84) 29 (83) 12 (28%) 28% after 12 months 77% 52% NCI adult 20 NR 14 (70) 12 (60) 10 (50%) 79% at 51. 6% at 75% 4. 9 10 months for MRD negative patients 30% Median 8. 5 months CRS % Neuro Side effects N(%) 43%

Remaining questions with CAR T cells • How long do CAR T cells last in the body? • Do you need an allogeneic stem cell transplant after achieving remission with CAR T cells? • Could patients be retreated with CAR T cells?

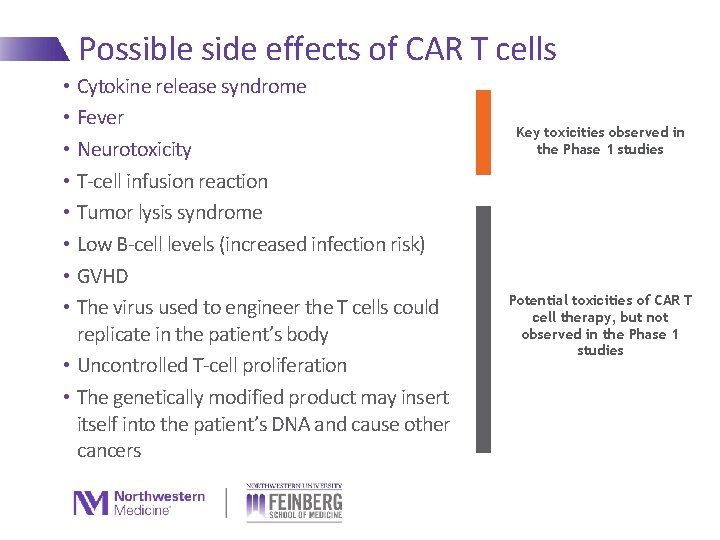
Possible side effects of CAR T cells • Cytokine release syndrome • Fever • Neurotoxicity • T-cell infusion reaction • Tumor lysis syndrome • Low B-cell levels (increased infection risk) • GVHD • The virus used to engineer the T cells could replicate in the patient’s body • Uncontrolled T-cell proliferation • The genetically modified product may insert itself into the patient’s DNA and cause other cancers Key toxicities observed in the Phase 1 studies Potential toxicities of CAR T cell therapy, but not observed in the Phase 1 studies 19

Summary • New therapies for leukemia and lymphoma are taking advantage of copying or utilizing the immune system to attack the cancer • Several new immunotherapies are available for ALL - No clinical trials have compared blinatumomab vs IO vs CAR T cells - We do not yet know which is the “best” treatment or what order we should give the treatments in - It is important to consider side effects when selecting a treatment with your doctor • Blinatumomab: neurologic side effects, cytokine release syndrome • IO: liver side effects • CAR T cells: CRS, neurologic side effects • Talk to your doctor about a clinical trial