Allogeneic Mini Transplantation Mark B Juckett M D

Allogeneic “Mini” Transplantation Mark B. Juckett M. D. June 4, 2004

Problems with BMT • Relapse – CML chronic phase – 10% – High risk AML/ALL – 50% • Toxicity – Non-relapse mortality of 10 – 40% – Graft vs. Host disease (GVHD) of 40 – 60% • Cost
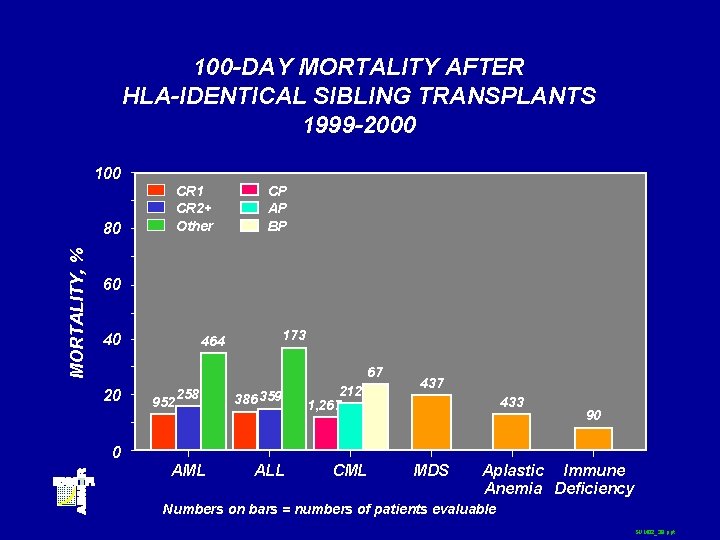
100 -DAY MORTALITY AFTER HLA-IDENTICAL SIBLING TRANSPLANTS 1999 -2000 100 CR 1 CR 2+ Other MORTALITY, % 80 CP AP BP 60 40 173 464 67 20 0 952 258 AML 386 359 ALL 212 1, 267 CML 437 433 MDS 90 Aplastic Immune Anemia Deficiency Numbers on bars = numbers of patients evaluable SUM 02_39. ppt

What is GVHD? • An cell mediated reaction of donor origin against recipient tissues • It requires: – a donor graft with immunologically competent cells – a recipient unable to mount immune response – recipient expresses tissue antigens that are not present in the donor.

Pathogenesis of GVHD Present self Ags to Donor React to Recipient Ags Recipient APC Recipient Donor T cells Donor

Why Does allogeneic BMT Work? • “Roundup” theory – eradicate all hematopoeitic tissue

Why Does allogeneic BMT Work? • Rescue patient with healthy stem cells • Graft vs. Host reactions a nuisance

Past Approaches used to Improve Outcome • Intensify regimen (More Roundup) • Better matching (twin donor best? ) • Improve immune suppression – i. e. “GVHD prophylaxis” • Remove immune cells capable of GVHD – “T cell depletion” started at UW
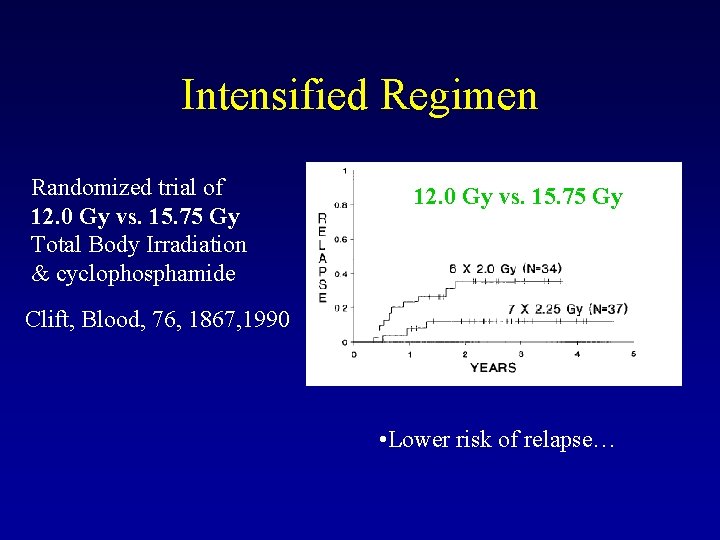
Intensified Regimen Randomized trial of 12. 0 Gy vs. 15. 75 Gy Total Body Irradiation & cyclophosphamide 12. 0 Gy vs. 15. 75 Gy Clift, Blood, 76, 1867, 1990 • Lower risk of relapse…
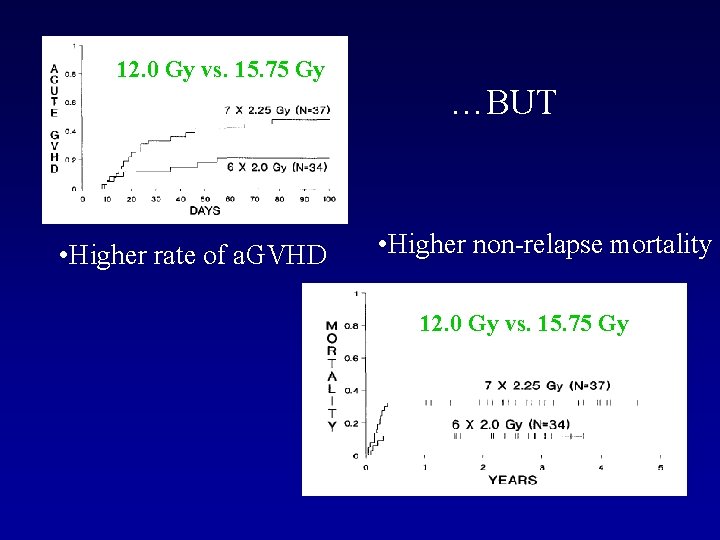
12. 0 Gy vs. 15. 75 Gy • Higher rate of a. GVHD …BUT • Higher non-relapse mortality 12. 0 Gy vs. 15. 75 Gy

GVHD Prophylaxis - How much? Aggressive Prophylaxis Minimal Prophylaxis • LESS GVHD • MORE infection • MORE relapse • MORE GVHD • LESS infection • LESS relapse SURVIVAL

Non-selective T cell depletion Relative Risk Grade 2 -4 AGVHD 0. 50 Graft Rejection 3. 37 Relapse (5 yr) 1. 87 Transplant Mortality 1. 60 Champlin, Blood, 95, 3996, 2000
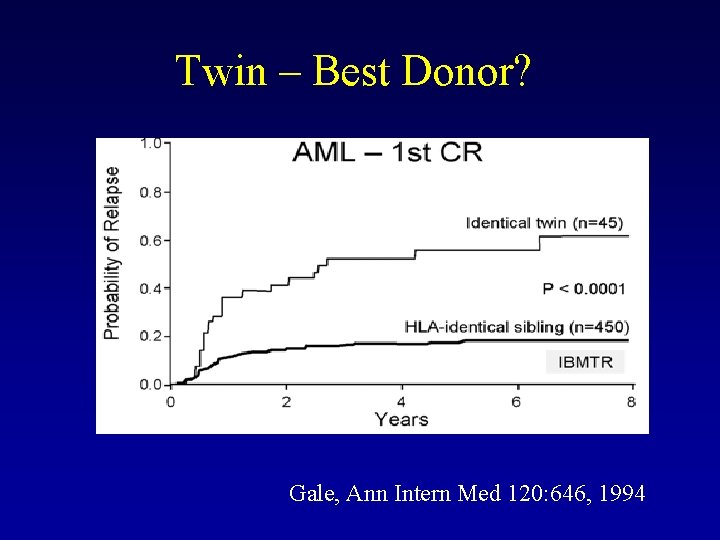
Twin – Best Donor? Gale, Ann Intern Med 120: 646, 1994
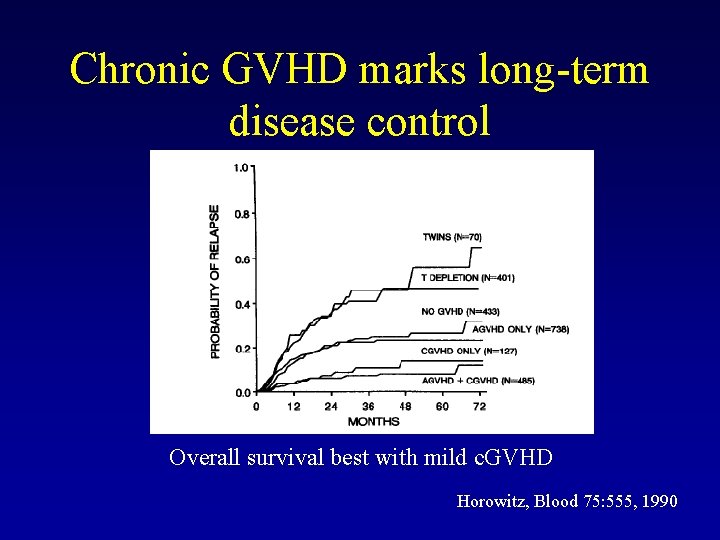
Chronic GVHD marks long-term disease control Overall survival best with mild c. GVHD Horowitz, Blood 75: 555, 1990
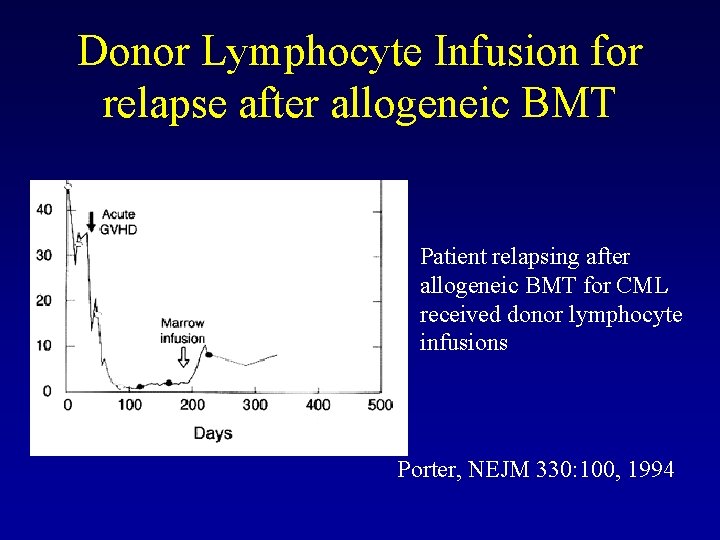
Donor Lymphocyte Infusion for relapse after allogeneic BMT Patient relapsing after allogeneic BMT for CML received donor lymphocyte infusions Porter, NEJM 330: 100, 1994
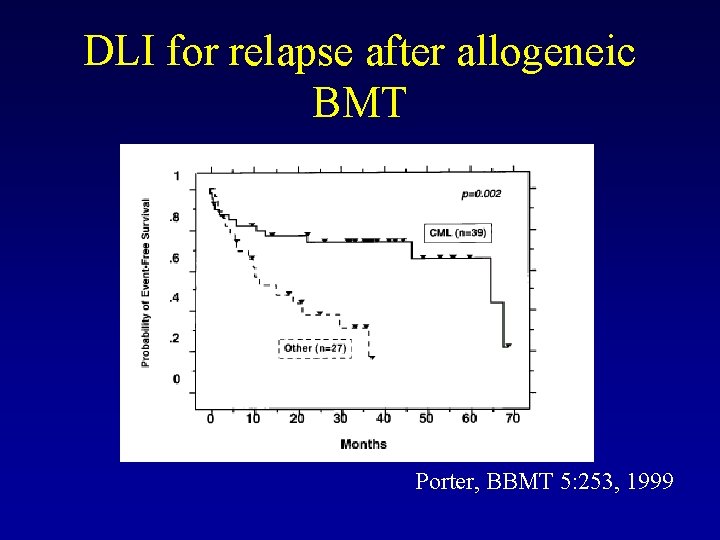
DLI for relapse after allogeneic BMT Porter, BBMT 5: 253, 1999

Learning Points • Preparative regimen provides short-term disease control – not cure. • Preparative regimen toxicity increases risk of acute GVHD (“cytokine storm”) • A “graft vs. disease” response exists – Varies with respect to disease • Long term disease control related to immunological effects from the donor – Correlates with chronic GVHD

New Paradigm • • Hematopoeitic stem cell transplantation succeeds when a chronic “allo”immune process is created that is specific to the disease/diseased tissue. The “preparative regimen” is necessary to provide: – Sufficient immune suppression for donor engraftment And – Short-term disease control sufficient to allow the autoimmune process to develop.

Strategies for Improvement • Reduce the intensity of the preparative regimen – Use agents specific to the disease & immunosuppressive • Speed neutrophil engraftment – Peripheral blood stem cell collection • Improve lymphoid immune reconstitution – Donor lymphocyte infusion

Spectrum of Preparative Regimens Immunosuppresion Cy/12 Gy TBI Bu/F/ATG 2 Gy TBI/Flu Bu/Cy MF FC 2 Gy TBI Flag Myelosuppression Human LD 50 = 4 Gy Myeloablative dose = 8 Gy
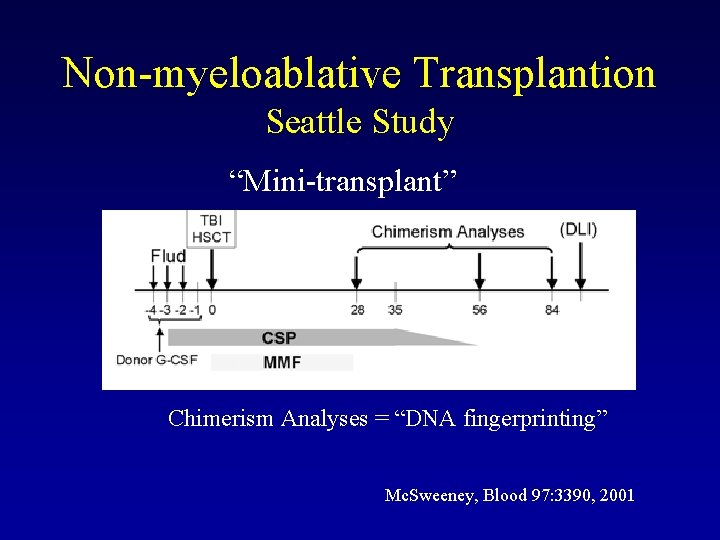
Non-myeloablative Transplantion Seattle Study “Mini-transplant” Chimerism Analyses = “DNA fingerprinting” Mc. Sweeney, Blood 97: 3390, 2001

Patients – Seattle Study • • MM MDS CLL CML AML NHL HD Other 41 26 19 17 17 19 12 5 • Eligibility – Age greater than 50 • Or – Ineligible for Conventional BMT • Aspergillis infection • Liver/cardiac/pulm disease • Previous BMT Mc. Sweeney, Blood 97: 3390, 2001
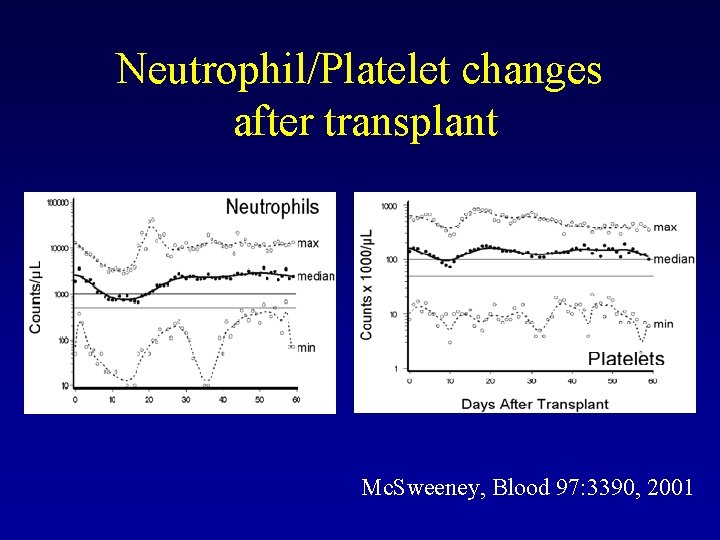
Neutrophil/Platelet changes after transplant Mc. Sweeney, Blood 97: 3390, 2001
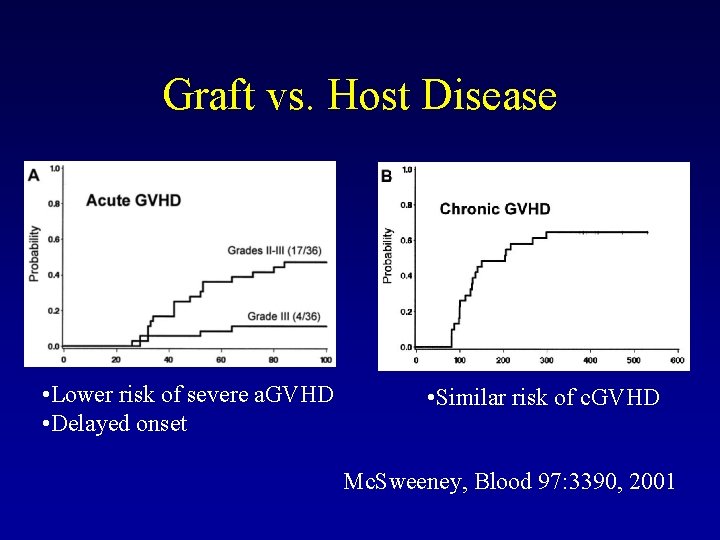
Graft vs. Host Disease • Lower risk of severe a. GVHD • Delayed onset • Similar risk of c. GVHD Mc. Sweeney, Blood 97: 3390, 2001
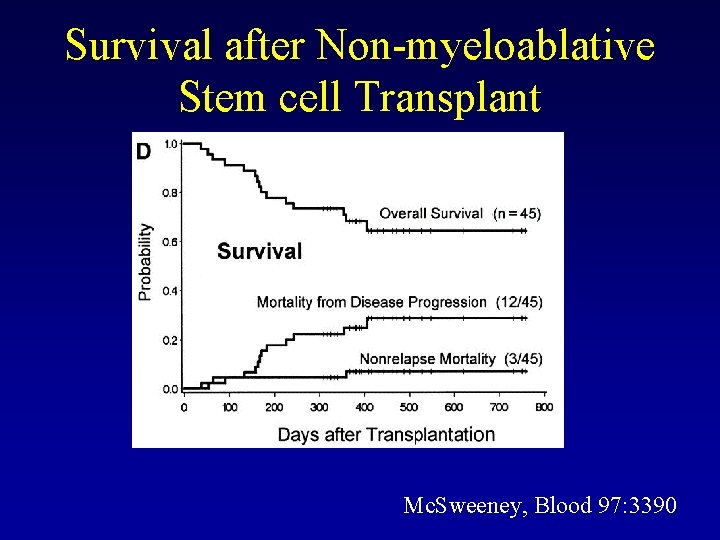
Survival after Non-myeloablative Stem cell Transplant Mc. Sweeney, Blood 97: 3390

Grade 3 -5 toxicity by day 100 Organ system Nonmyeloablative Myeloablative Cardiovascular 47% 74% Gastrointenstinal 12% 76% Hepatic 37% 57% Infection 56% 84% Pulmonary 6% 21% Diaconescu, Blood, 102: 261 a, 2003
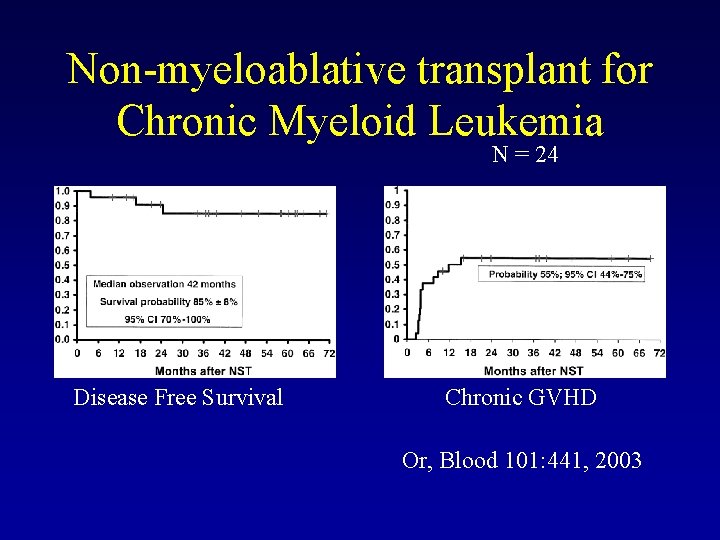
Non-myeloablative transplant for Chronic Myeloid Leukemia N = 24 Disease Free Survival Chronic GVHD Or, Blood 101: 441, 2003
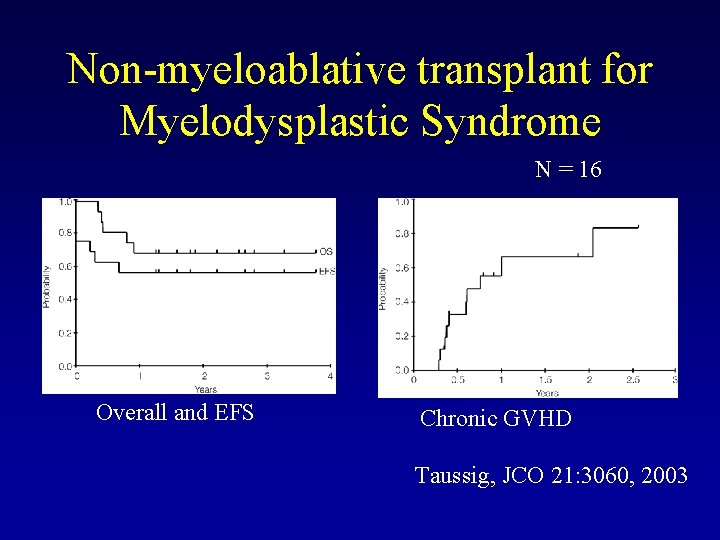
Non-myeloablative transplant for Myelodysplastic Syndrome N = 16 Overall and EFS Chronic GVHD Taussig, JCO 21: 3060, 2003
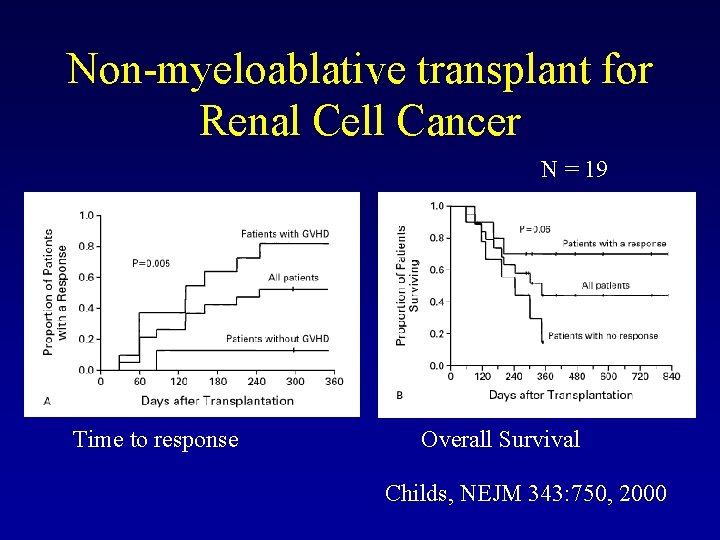
Non-myeloablative transplant for Renal Cell Cancer N = 19 Time to response Overall Survival Childs, NEJM 343: 750, 2000
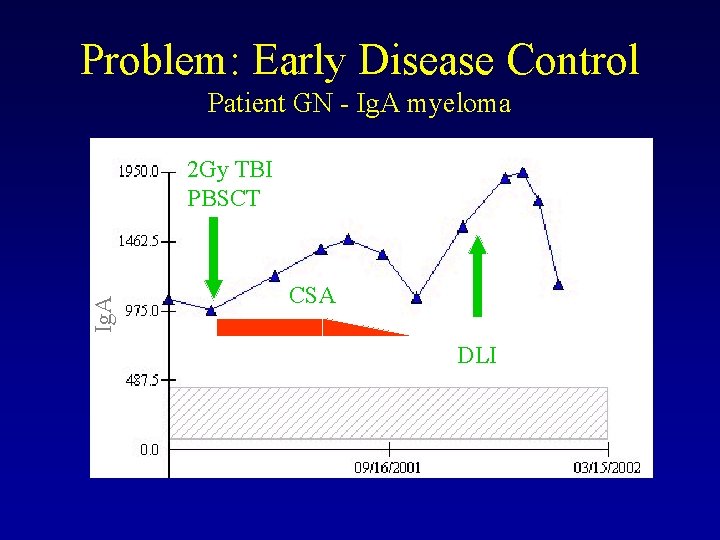
Problem: Early Disease Control Patient GN - Ig. A myeloma Ig. A 2 Gy TBI PBSCT CSA DLI

Findings from NST trials • Early toxicity reduced – Heme toxicity much shortened • Outpatient management feasible • Engraftment successful – with fludarabine added to regimen • Risk of a. GVHD reduced and delayed • Risk of c. GVHD unchanged but delayed • Early disease progression common

Disease Sensitivity to “Graft vs. Malignancy” • Sensitive – – CML Follicular lymphoma Mantle cell lymphoma CLL • Insensitive – ALL – High-grade NHL • Intermediate – – – AML Diffuse large NHL Multiple myeloma Hodgkin disease Renal cell Breast cancer

Strategies to Improve NST • Treat to remission prior to transplant • Use disease specific chemotherapy in regimen • Incorporate monoclonal antibodies • Infuse engineered lymphocytes • Use Auto followed by Allo strategy – Allow recovery/healing prior to allo transplant

“Auto/Allo” strategy for Myeloma Auto PBSCT Allogeneic PBSCT Recovery 60 – 90 days High dose Melphalan Immune suppression 2 Gy TBI BMT CTN 0102

“Auto/Allo” - Results • • • 54 patients (median age 52) Overall 1 -year survival 78% at 18 months Event Free Survival 2 -year 55% Day-200 mortality 7% GVHD – Acute 39% – Chronic 46% • Response Rate 81% (CR 52%, PR 29%) Maloney, Blood 98: 1822 a

Problem: Need for phase III trials! • Blood and Marrow Transplant Clinical Trials Network (BMT CTN) – NCI sponsored cooperative trials group – Composed of 14 Core Transplant Centers – Goal to complete high-quality clinical trials in BMT

BMT CTN Protocol 0102 Myeloma

BMT CTN Protocol 0202 Follicular Lymphoma

Conclusions • Allogeneic transplantation works due to a “Graft vs. Malignancy” immune response. • NST approaches have improved the safety of transplantation. • NST allows transplantation of patients not eligible for standard approaches. • Phase III studies are need to determine place in therapy.
- Slides: 39