The Dipole Moment of the Sulfuric Acid Monomer












- Slides: 23

The Dipole Moment of the Sulfuric Acid Monomer Galen Sedo, Jane Curtis, Kenneth R. Leopold Department of Chemistry, University of Minnesota

Investigating Sulfuric Acid Systems Sulfuric Acid Aerosols • A principle source of sulfate-containing atmospheric particles • High affinity for water and a high rate of nitrogen species uptake Nucleation Theory • Homogeneous and Heterogeneous particle growth • Ion-induced and ion-mediated nucleation theory • Charge-dipole interactions

Previous Dipole Moment Work H 2 SO 4 -H 2 Oa H 2 SO 4 b 3. 052(17) D 2. 725(15) D a) Brauer, C. S. ; Sedo, G. ; Leopold, K. R. Geophys. Res. Lett. 33 (2006) L 23805, doi: 10. 1029/2006 GL 028110. b) Kuczkowski, R. L. ; Suenram, R. D. ; Lovas, F. J. Journal of the American Chemical Society 1981, 103, 2561.

Previous Dipole Moment Work H 2 SO 4 -H 2 Oa 3. 052(17) D PW 91/TZPc 2. 147 D 0. 905 D (29. 7 %) H 2 SO 4 b 2. 725(15) D -0. 092 D (-3. 4 %) 2. 817 D a) Brauer, C. S. ; Sedo, G. ; Leopold, K. R. Geophys. Res. Lett. 33 (2006) L 23805, doi: 10. 1029/2006 GL 028110. b) Kuczkowski, R. L. ; Suenram, R. D. ; Lovas, F. J. Journal of the American Chemical Society 1981, 103, 2561. c) Al Natsheh, A; Nadykto, A. B. ; Mikkelsen, K. V. ; Yu, F. ; Ruuskanen, J. J. Phys. Chem. A 2004, 108, 8914.

Previous Dipole Moment Work H 2 SO 4 -H 2 Oa 3. 052(17) D PW 91/TZPc 2. 147 D MP 2/aug-cc-p. VQZa 2. 965 D 0. 905 D (29. 7 %) H 2 SO 4 b 2. 725(15) D -0. 092 D (-3. 4 %) 2. 817 D 0. 087 D (2. 9 %) 3. 147 D -0. 692 D (-25. 4 %) a) Brauer, C. S. ; Sedo, G. ; Leopold, K. R. Geophys. Res. Lett. 33 (2006) L 23805, doi: 10. 1029/2006 GL 028110. b) Kuczkowski, R. L. ; Suenram, R. D. ; Lovas, F. J. Journal of the American Chemical Society 1981, 103, 2561. c) Al Natsheh, A; Nadykto, A. B. ; Mikkelsen, K. V. ; Yu, F. ; Ruuskanen, J. J. Phys. Chem. A 2004, 108, 8914.

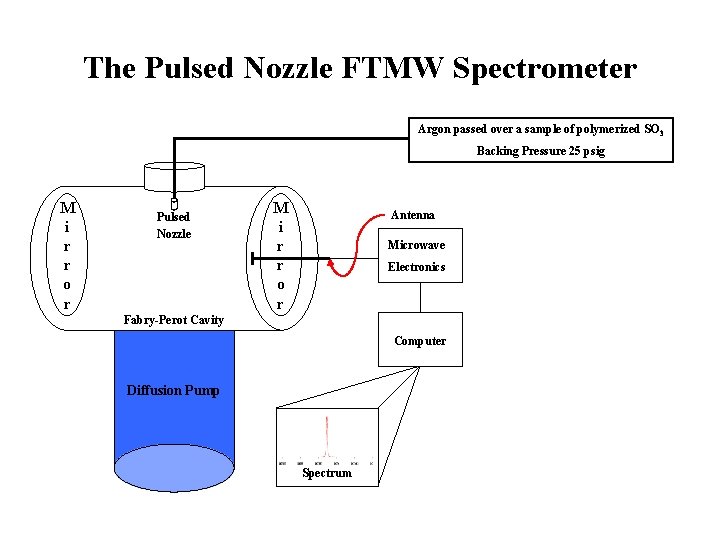
The Pulsed Nozzle FTMW Spectrometer Argon passed over a sample of polymerized SO 3 Backing Pressure 25 psig M i r r o r Pulsed Nozzle M i r r o r Antenna Microwave Electronics Fabry-Perot Cavity Computer Diffusion Pump Spectrum

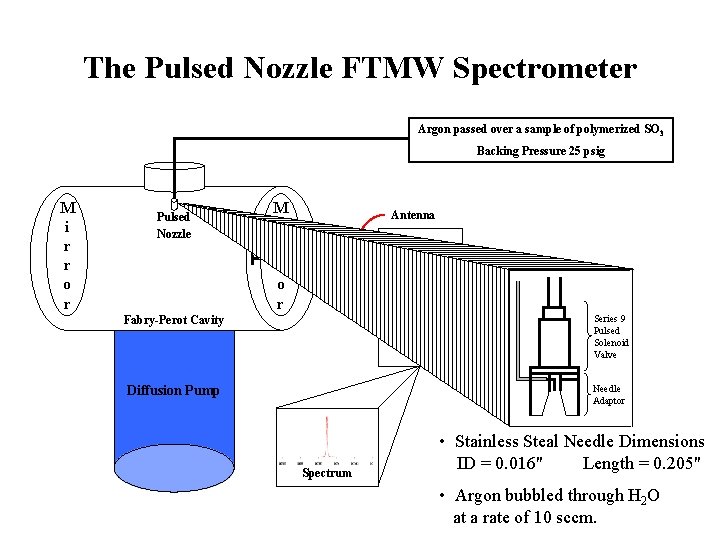
The Pulsed Nozzle FTMW Spectrometer Argon passed over a sample of polymerized SO 3 Backing Pressure 25 psig M i r r o r Pulsed Nozzle M i r r o r Antenna Microwave Electronics Fabry-Perot Cavity Computer Diffusion Pump Series 9 Pulsed Solenoid Valve Needle Adaptor Spectrum • Stainless Steal Needle Dimensions ID = 0. 016" Length = 0. 205" • Argon bubbled through H 2 O at a rate of 10 sccm.

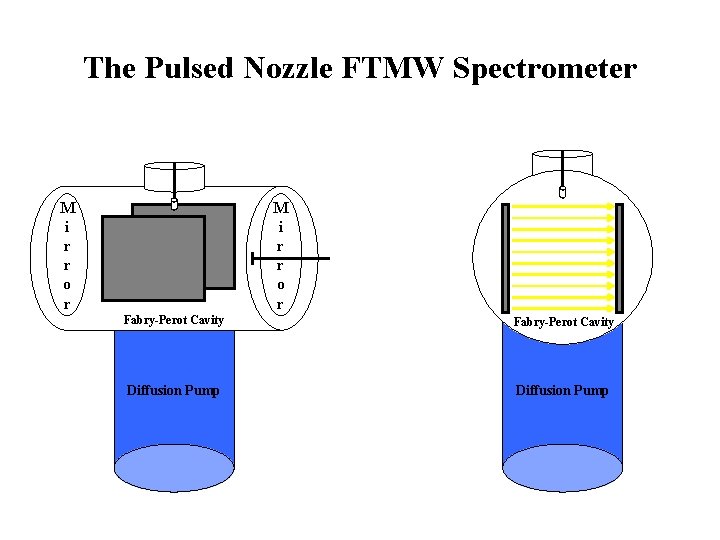
The Pulsed Nozzle FTMW Spectrometer M i r r o r Fabry-Perot Cavity Diffusion Pump

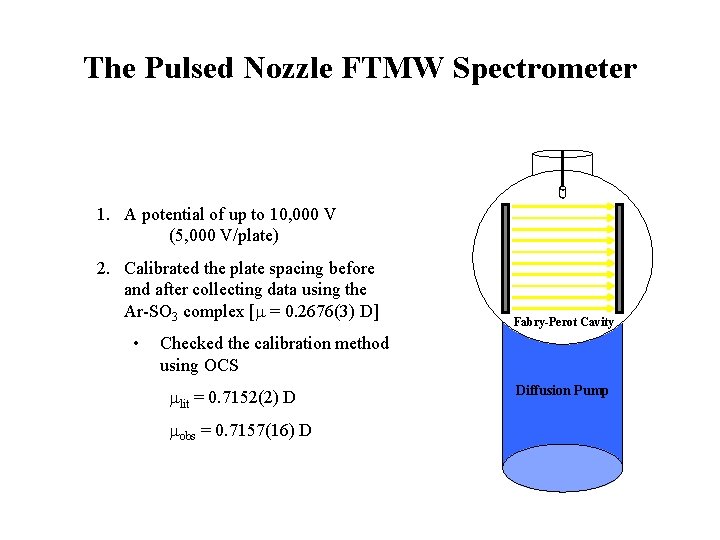
The Pulsed Nozzle FTMW Spectrometer 1. A potential of up to 10, 000 V (5, 000 V/plate) 2. Calibrated the plate spacing before and after collecting data using the Ar-SO 3 complex [m = 0. 2676(3) D] • Fabry-Perot Cavity Checked the calibration method using OCS mlit = 0. 7152(2) D mobs = 0. 7157(16) D Diffusion Pump

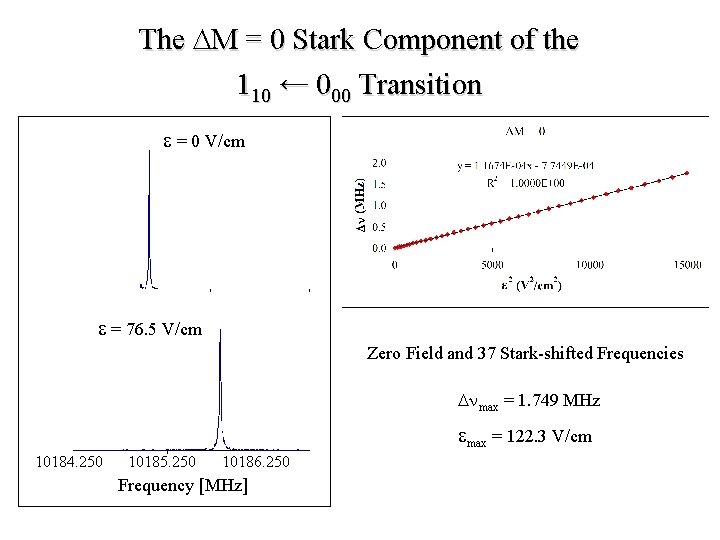
The DM = 0 Stark Component of the 110 ← 000 Transition e = 0 V/cm e = 76. 5 V/cm Zero Field and 37 Stark-shifted Frequencies Dnmax = 1. 749 MHz emax = 122. 3 V/cm 10184. 250 10185. 250 10186. 250 Frequency [MHz]

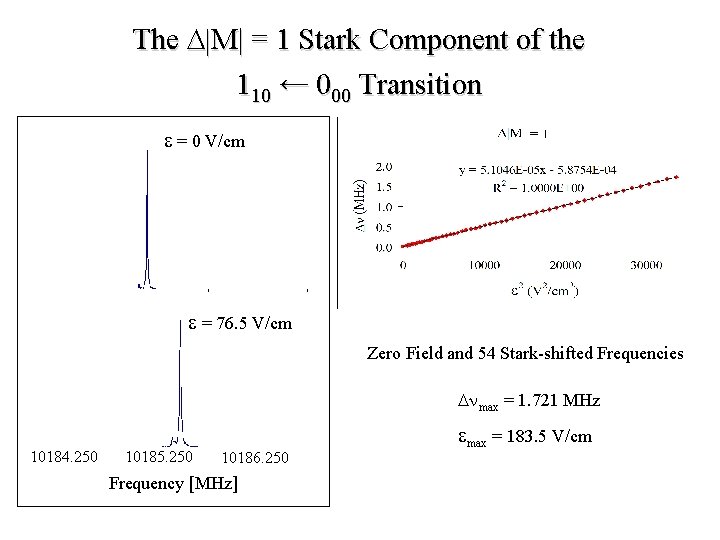
The D|M| = 1 Stark Component of the 110 ← 000 Transition e = 0 V/cm e = 76. 5 V/cm Zero Field and 54 Stark-shifted Frequencies Dnmax = 1. 721 MHz 10184. 250 10185. 250 10186. 250 Frequency [MHz] emax = 183. 5 V/cm

Sulfuric Acid Molecular Constantsa a) Kuczkowski, R. L. ; Suenram, R. D. ; Lovas, F. J. “Microwave Spectrum, Structure, and Dipole Moment of Sulfuric Acid. ” Journal of the American Chemical Society 1981, 103, 2561 -2566.

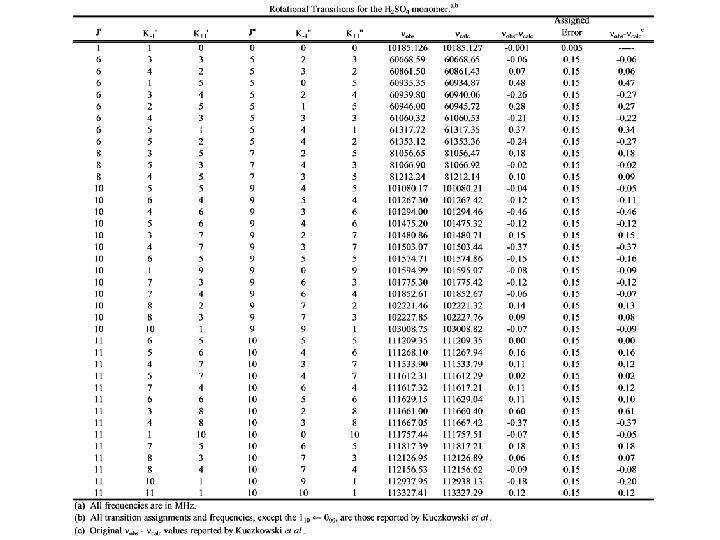
Sulfuric Acid Transition Frequenciesa a) Kuczkowski, R. L. ; Suenram, R. D. ; Lovas, F. J. “Microwave Spectrum, Structure, and Dipole Moment of Sulfuric Acid. ” Journal of the American Chemical Society 1981, 103, 2561 -2566.

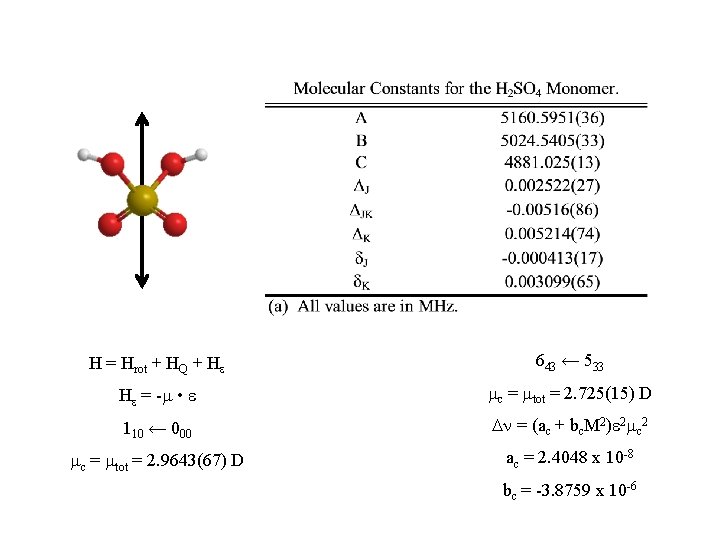
H = Hrot + HQ + He 643 ← 533 He = -m • e mc = mtot = 2. 725(15) D 110 ← 000 Dn = (ac + bc. M 2)e 2 mc 2 mc = mtot = 2. 9643(67) D ac = 2. 4048 x 10 -8 bc = -3. 8759 x 10 -6

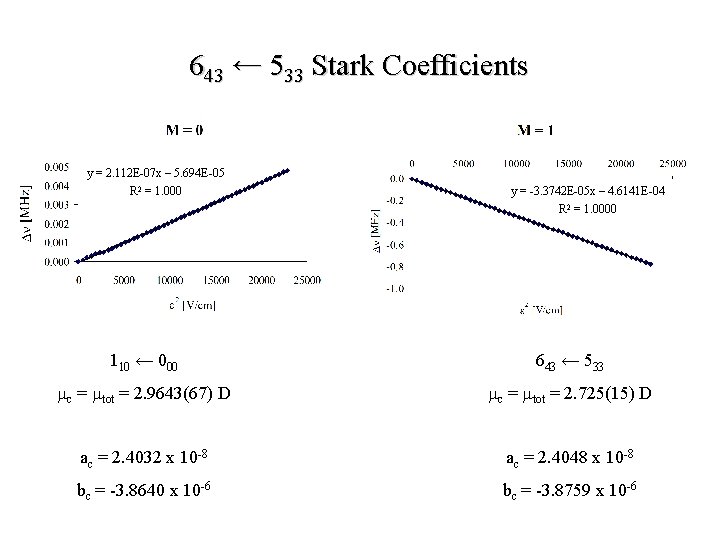
643 ← 533 Stark Coefficients y = 2. 112 E-07 x – 5. 694 E-05 R 2 = 1. 000 y = -3. 3742 E-05 x – 4. 6141 E-04 R 2 = 1. 0000 110 ← 000 643 ← 533 mc = mtot = 2. 9643(67) D mc = mtot = 2. 725(15) D ac = 2. 4032 x 10 -8 ac = 2. 4048 x 10 -8 bc = -3. 8640 x 10 -6 bc = -3. 8759 x 10 -6

Error Analysis Three Primary Sources of Experimental Error 1. Least Squares Analysis mls = 2. 96434 D Dls = 0. 00023 D

Error Analysis Three Primary Sources of Experimental Error 1. Least Squares Analysis mls = 2. 96434 D Dls = 0. 00023 D 2. Plate Spacing dplate = 32. 702 cm Dplate = 0. 064 cm

Error Analysis Three Primary Sources of Experimental Error 1. Least Squares Analysis mls = 2. 96434 D Dls = 0. 00023 D 2. Plate Spacing dplate = 32. 702 cm Dplate = 0. 064 cm 3. Calibration Standard m. Ar-SO 3 = 0. 2676 D DAr-SO 3 = 0. 0003 D

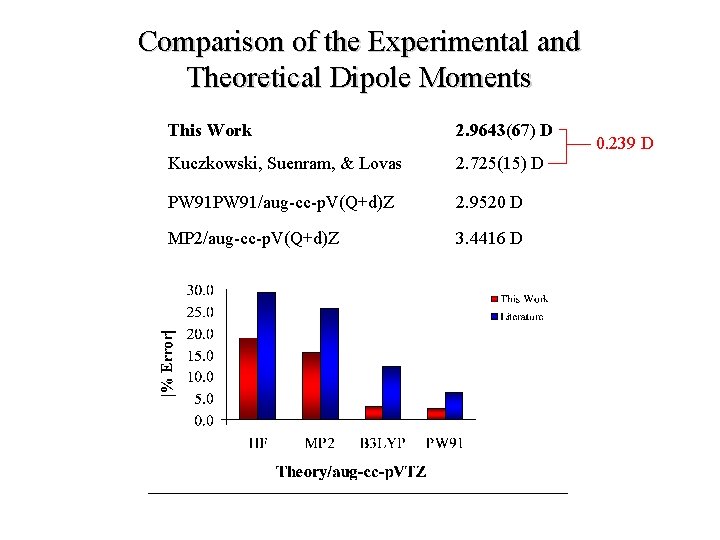
Comparison of the Experimental and Theoretical Dipole Moments This Work 2. 9643(67) D Kuczkowski, Suenram, & Lovas 2. 725(15) D PW 91/aug-cc-p. V(Q+d)Z 2. 9520 D MP 2/aug-cc-p. V(Q+d)Z 3. 4416 D 0. 239 D

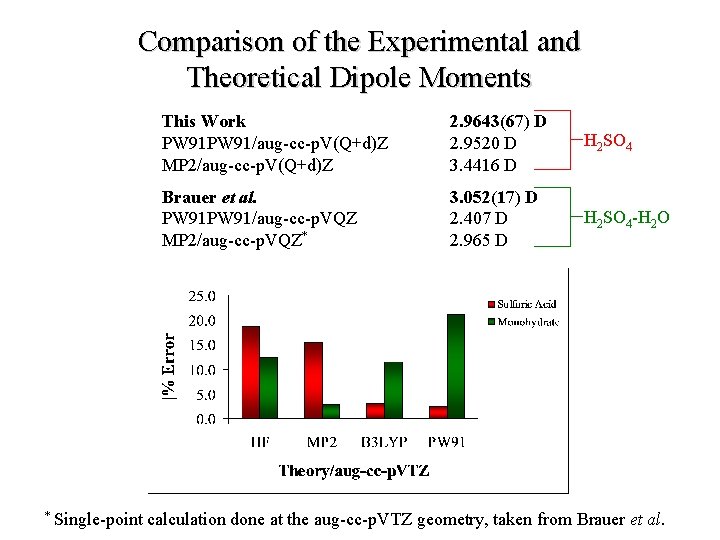
Comparison of the Experimental and Theoretical Dipole Moments * Single-point This Work PW 91/aug-cc-p. V(Q+d)Z MP 2/aug-cc-p. V(Q+d)Z 2. 9643(67) D 2. 9520 D 3. 4416 D H 2 SO 4 Brauer et al. PW 91/aug-cc-p. VQZ MP 2/aug-cc-p. VQZ* 3. 052(17) D 2. 407 D 2. 965 D H 2 SO 4 -H 2 O calculation done at the aug-cc-p. VTZ geometry, taken from Brauer et al.

Conclusions 1. The dipole moment of the sulfuric acid monomer has been refined using Fourier transform microwave spectroscopy. • 91 Stark-shifted frequencies were collect for the 110 ← 000 rotational transition. • The newly measured value, 2. 9643(67) D, represents an increase of approximately 0. 24 D over the previously published value. 2. The dipole moments of the sulfuric acid monomer and mono-hydrate were calculated using both ab initio and Density Functional Theory. • The calculated dipole moments show convergence with increasing basis set size. • The agreement between the measured experimental values and those of theory various drastically with the method employed.

Acknowledgements • Dr. Kenneth Leopold • Jane Curtis • Dr. Carolyn Brauer Funding • National Science Foundation (NSF) • Petroleum Research Fund (PRF) • Minnesota Supercomputing Institute (MSI)
