What is Np Huric Urea Sulfuric Acid urea
























- Slides: 32


What is N-p. Huric®? • Urea Sulfuric Acid (urea sulfate, MCDS). • 15/49 is a 1: 1 ratio of urea to sulfuric acid. (15‐ 0‐ 0‐ 16 S, 49% sulfuric acid) • 28/27 (28 -0 -0 -9 S, 27% sulfuric acid) Same product is p. Hairway! • 10/55 (10 -0 -0 -18 S, 55% sulfuric acid)

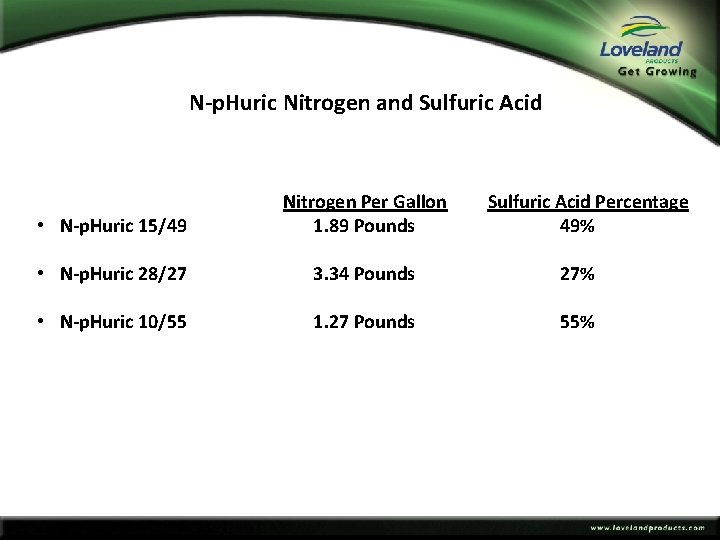
N‐p. Huric Nitrogen and Sulfuric Acid • N‐p. Huric 15/49 Nitrogen Per Gallon 1. 89 Pounds Sulfuric Acid Percentage 49% • N‐p. Huric 28/27 3. 34 Pounds 27% • N‐p. Huric 10/55 1. 27 Pounds 55%

n-p. Huric Treating Irrigation Water Sulfuric acid reaction with bicarbonate in water: ` H 2 SO 4 + 2 HCO 3⁻ → Ca⁺⁺ + SO 4²⁻ + 2 CO 2 + 2 H 2 O The addition of sulfuric acid Calcium in the Carbonate and bicarbonate molecules disassociate forming sulfate SO 4²⁻ Carbon Dioxide Gas and Water.

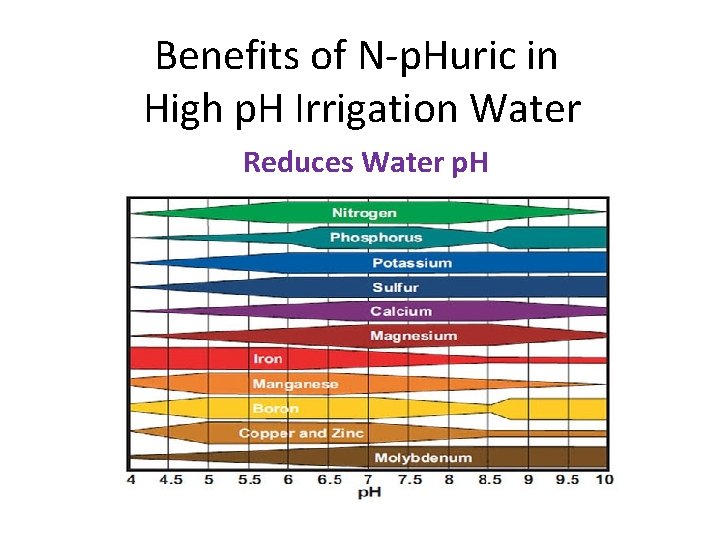
Benefits of N-p. Huric in High p. H Irrigation Water Reduces Water p. H

Benefits of N-p. Huric on High p. H Soil • Reduces Soil p. H • Solubilizes Calcium • Calcium replaces Sodium on clay particles. – Ca⁺⁺ has stronger adhesion than Na⁺ • Improves soil structure, water infiltration and drainage. • Need irrigation to flush salts below root zone. • Can also solubilize PO 4, Zn, Fe and Mn.

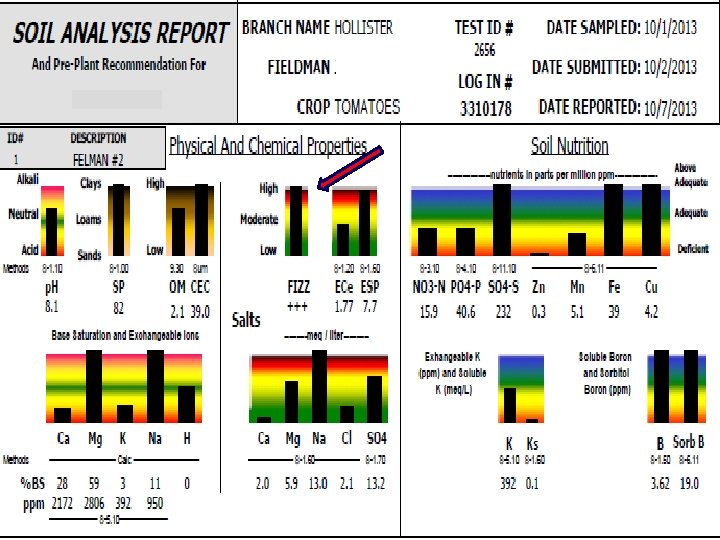
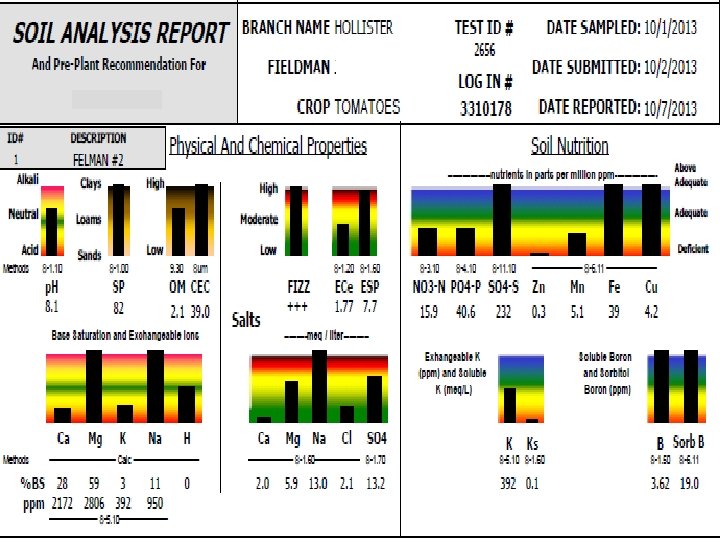
FIZZ!! • Free lime or precipitated calcium carbonate • Buffers p. H against changes • High FIZZ with normal sodium will have a soil p. H of 7. 5 -8. 3 • High FIZZ and low soluble calcium means all calcium is tied up in free lime • Need to add acid: p. HAcid, N-p. Huric, sulfur

Gypsum or Acid? What is your Fizz? Ca 2+ SO 42 - Na+ Na+ - - - Na+ + Na Ca++ - - - - Ca++ Na+ Na+






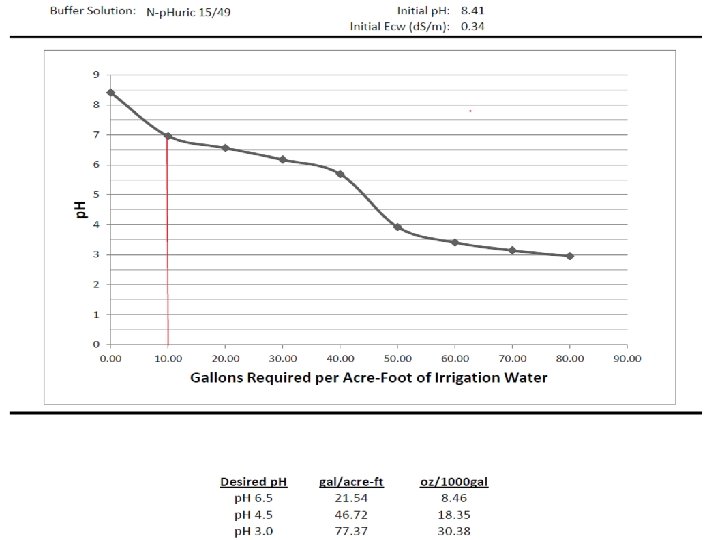
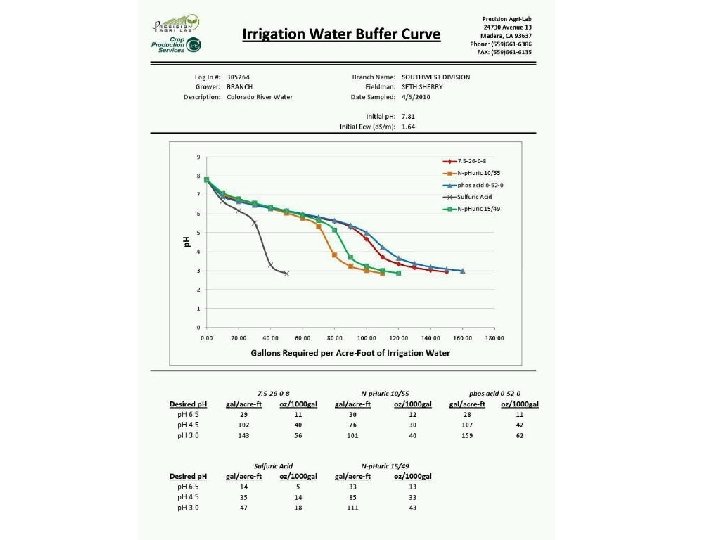
2656 Soil with Free Lime 3053 Water p. H Buffer/Titration (Specify Acid)



Handling N-p. Huric® • Concentrated N-p. Huric is much less corrosive to human flesh when compared to sulfuric acid. • Eye-protection is a must. • Avoid mist inhalation. • Non-flammable. • Non-volatile.

Storing and handling N-p. Huric® • Polyethylene, polypropylene and 316 L Stainless are approved materials for storage and concentrated application. May Be Used: Ceramic E. P. D. M Kynar Polyethylene Polypropylene P. V. C and C. P. V. C Rayton 316 L Stainless Steel Teflon Viton Zalak Do Not Use: Aluminum Nylon Polyester FRP Neoprene Brass Buna-N Cast Iron Celcon Delrin Epoxy Fiberglass Galvanized Steel Hypalon Mild Steel Natural Rubber




Sulfuric Acid - Dangers • Low p. H denatures proteins in skin (chemical burns) • Exothermic reaction with water in skin causes thermal burns • Hygroscopic nature of acid dewaters your skin hence skin turns “Black” like Carbon • Oxidizing property of Acid damages tissue and causes chemical burns

Nitrogen Volatility • MCDS (Moncarbamide Dihydrogen Sulfate) 15/49 (15 -0 -0 -16, 49% sulfuric acid) No Volatility • MCDS (Moncarbamide Dihydrogen Sulfate) 10/55 (10 -0 -0 -18, 55% sulfuric acid) No Volatility • DCDS (Dicarbamide Dihydrogen Sulfate) 28/27 (28 -0 -0 -9, 27% sulfuric acid) Low Volatility

• High levels of bicarbonates pull calcium out of solution, reducing the presence of calcium on soil exchange sites. • • Bicarbonate levels in irrigation water are: – 0‐ 100 ppm (low) – 100‐ 180 ppm (moderate – 180‐ 600 ppm (severe)

Examples of uses • Water Treatment • Drip Grade Fertilizer – PO 4 through drip • Soil Applied Fertilizer – Sidedress, Pop-ups, Starters • Onion Foliar Fertilizer • Stubble digestion • Defoliant

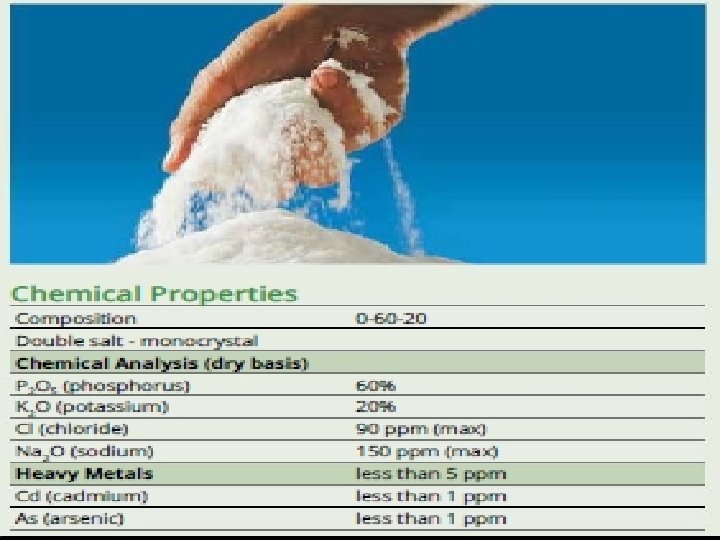
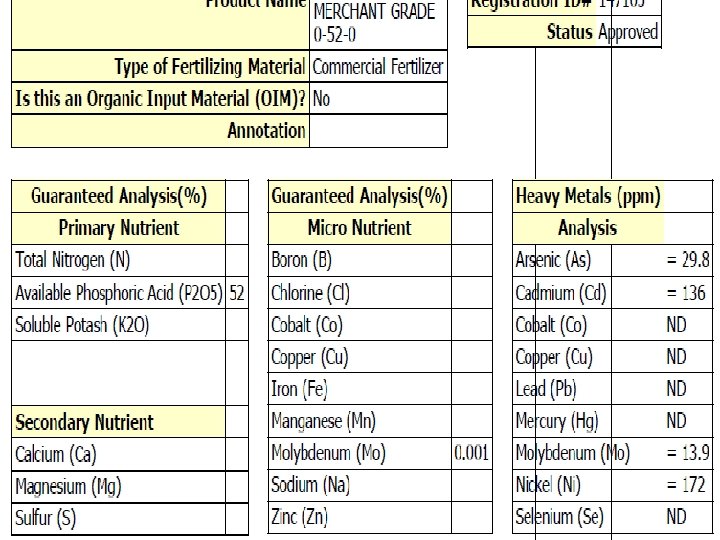
Acid Fertilizer Options Products: Sulfuric Acid N-p. Huric Pe. Kacid Phosphoric Acid Compatible Blends: 7. 5 – 26 – 0 3 – 40 – 0 10 – 20 – 0 5– 5– 5 2 – 10 – 6 6 - 16 - 0 7‐ 7‐ 0 5 – 25 – 5 5 – 26 – 2

More blend options • Zn, Fe, Mn Sulfates up to 2% total elemental analysis. • Zn, Fe, Mn Polysaccharides up to 2% total elemental analysis. • 15/49 is compatible with Watermaxx® 2




Components of an injection system A good, durable p. H meter costs approx. $100. Useful for most applications.

Alkaline Soils Need Acid Fertilizer Blends WATER AND SOIL ANALYSIS CUSTOM LIQUID FERTILIZER BLENDS NEED BASED CUSTOM FORMULATIONS