Intermolecular Interactions between Formaldehyde and Dimethyl Ether and

Intermolecular Interactions between Formaldehyde and Dimethyl Ether and between Formaldehyde and Dimethyl Sulfide in the Complex, Investigated by Fourier Transform Microwave Spectroscopy and Ab Initio Calculations Yoshiyuki Kawashima, Yoshio Tatamitani, and Yoshihiro Osamura Department of Applied Chemistry, Faculty of Engineering, Kanagawa Institute of Technology, Atsugi, Kanagawa 240 -0292, Japan Eizi Hirota The Graduate University for Advanced Studies, Hayama, Kanagawa 240 -0193, Japan
Intermolecular Interactions (1) Hydrogen bond Dipole-dipole interaction Van der Waals force strong weak (DME)2, DME-DFE, DME-TFE, and DMS-DME have three weak hydrogen bonds: C-H---Y (Y = O, S, and F) Attention is paid to the roles of these weak hydrogen bonds in the supramolecules and related biomolecules. Y. Tatamitani et. al. J. Am. Chem. Soc. 124 (2002) 2739
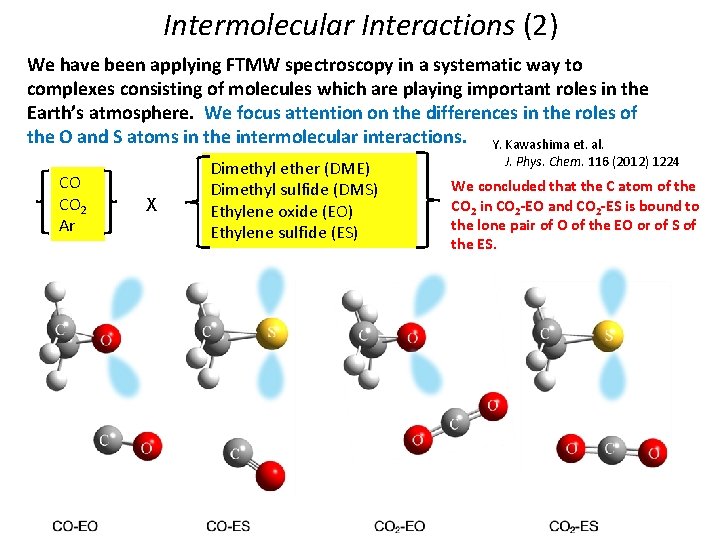
Intermolecular Interactions (2) We have been applying FTMW spectroscopy in a systematic way to complexes consisting of molecules which are playing important roles in the Earth’s atmosphere. We focus attention on the differences in the roles of the O and S atoms in the intermolecular interactions. Y. Kawashima et. al. CO CO 2 Ar X Dimethyl ether (DME) Dimethyl sulfide (DMS) Ethylene oxide (EO) Ethylene sulfide (ES) J. Phys. Chem. 116 (2012) 1224 We concluded that the C atom of the CO 2 in CO 2 -EO and CO 2 -ES is bound to the lone pair of O of the EO or of S of the ES.
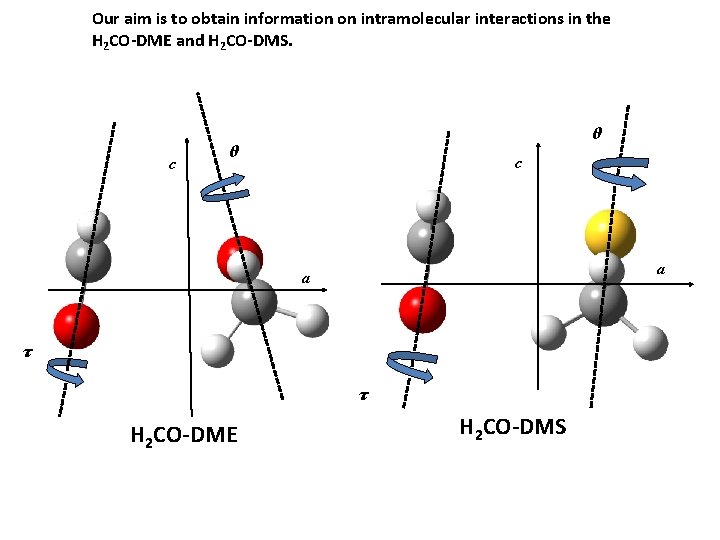
Our aim is to obtain information on intramolecular interactions in the H 2 CO-DME and H 2 CO-DMS. c θ θ c a a τ τ H 2 CO-DME H 2 CO-DMS
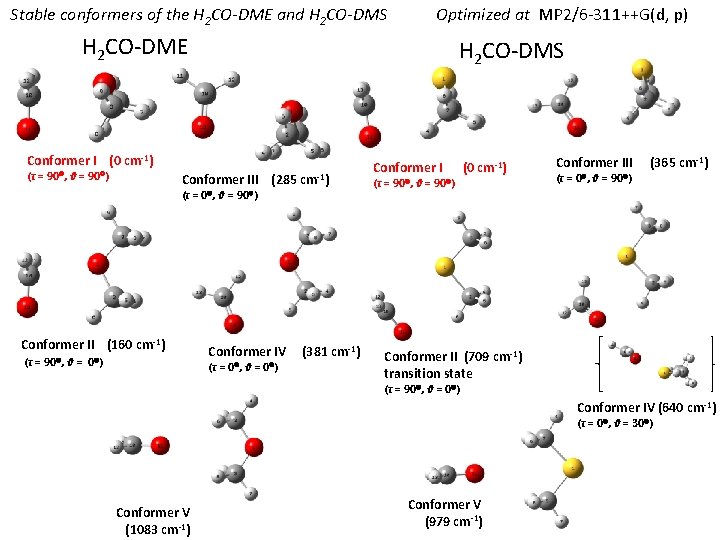
Stable conformers of the H 2 CO-DME and H 2 CO-DMS Optimized at MP 2/6 -311++G(d, p) H 2 CO-DME H 2 CO-DMS Conformer I (0 cm-1) (τ = 90 , θ = 90 ) Conformer III (285 cm-1) (τ = 0 , θ = 90 ) Conformer II (160 cm-1) (τ = 90 , θ = 0 ) Conformer IV (τ = 0 , θ = 0 ) (381 cm-1) Conformer I (0 cm-1) (τ = 90 , θ = 90 ) Conformer III (365 cm-1) (τ = 0 , θ = 90 ) Conformer II (709 cm-1) transition state (τ = 90 , θ = 0 ) Conformer IV (640 cm-1) (τ = 0 , θ = 30 ) Conformer V (1083 cm-1) Conformer V (979 cm-1)
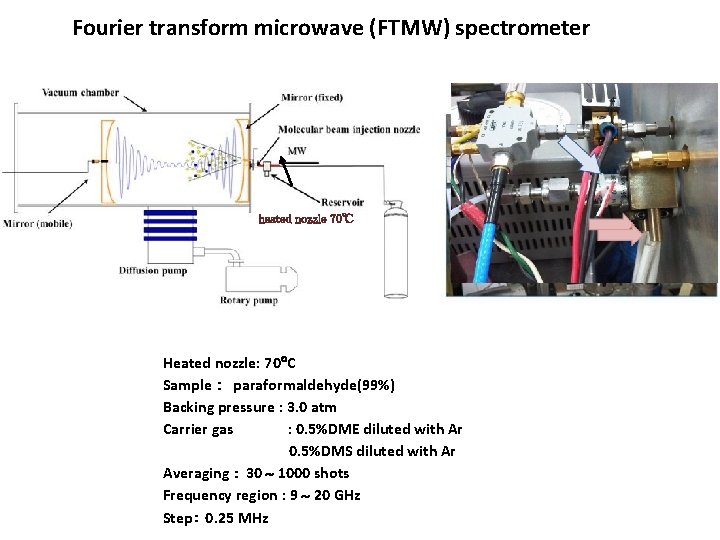
Fourier transform microwave (FTMW) spectrometer heated nozzle 70℃ buffer gas Heated nozzle: 70 C Sample : paraformaldehyde(99%) Backing pressure : 3. 0 atm Carrier gas : 0. 5%DME diluted with Ar 0. 5%DMS diluted with Ar Averaging : 30 1000 shots Frequency region : 9 20 GHz Step: 0. 25 MHz
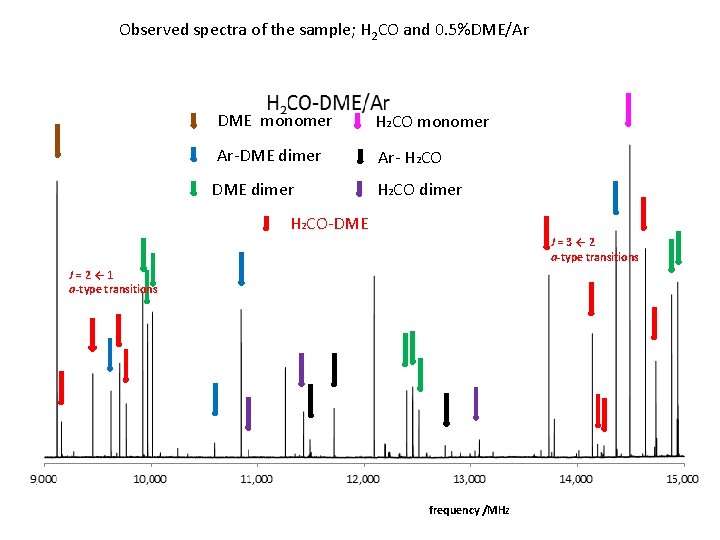
Observed spectra of the sample; H 2 CO and 0. 5%DME/Ar DME monomer H 2 CO monomer Ar-DME dimer Ar- H 2 CO DME dimer H 2 CO-DME J=3← 2 a-type transitions J=2← 1 a-type transitions frequency /MHz
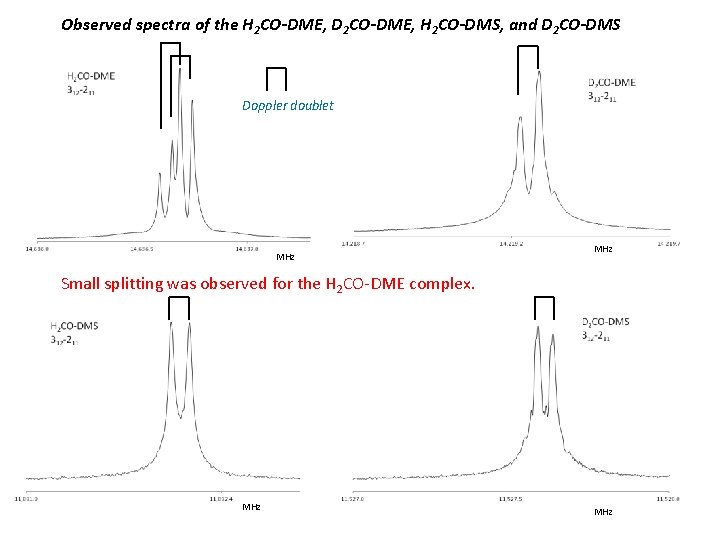
Observed spectra of the H 2 CO-DME, D 2 CO-DME, H 2 CO-DMS, and D 2 CO-DMS Doppler doublet MHz Small splitting was observed for the H 2 CO-DME complex. MHz
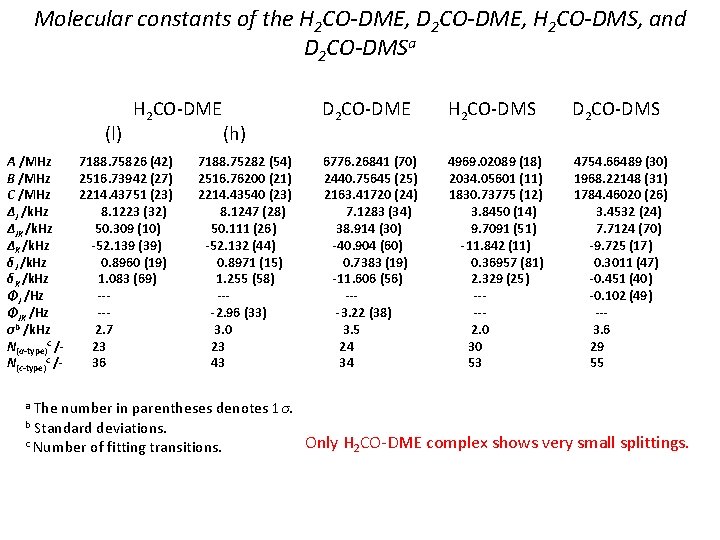
Molecular constants of the H 2 CO-DME, D 2 CO-DME, H 2 CO-DMS, and D 2 CO-DMSa (l) A /MHz B /MHz C /MHz ΔJ /k. Hz ΔJK /k. Hz ΔK /k. Hz δJ /k. Hz δK /k. Hz ΦJ /Hz ΦJK /Hz σb /k. Hz N(a-type)c /N(c-type)c /- H 2 CO-DME 7188. 75826 (42) 2516. 73942 (27) 2214. 43751 (23) 8. 1223 (32) 50. 309 (10) -52. 139 (39) 0. 8960 (19) 1. 083 (69) ----2. 7 23 36 (h) 7188. 75282 (54) 2516. 76200 (21) 2214. 43540 (23) 8. 1247 (28) 50. 111 (26) -52. 132 (44) 0. 8971 (15) 1. 255 (58) ---2. 96 (33) 3. 0 23 43 D 2 CO-DME H 2 CO-DMS D 2 CO-DMS 6776. 26841 (70) 2440. 75645 (25) 2163. 41720 (24) 7. 1283 (34) 38. 914 (30) -40. 904 (60) 0. 7383 (19) -11. 606 (56) ---3. 22 (38) 3. 5 24 34 4969. 02089 (18) 4754. 66489 (30) 2034. 05601 (11) 1968. 22148 (31) 1830. 73775 (12) 1784. 46020 (26) 3. 8450 (14) 3. 4532 (24) 9. 7091 (51) 7. 7124 (70) -11. 842 (11) -9. 725 (17) 0. 36957 (81) 0. 3011 (47) 2. 329 (25) -0. 451 (40) ---0. 102 (49) ----2. 0 3. 6 30 29 53 55 The number in parentheses denotes 1σ. Standard deviations. c Number of fitting transitions. Only H 2 CO-DME complex shows very small splittings. a b
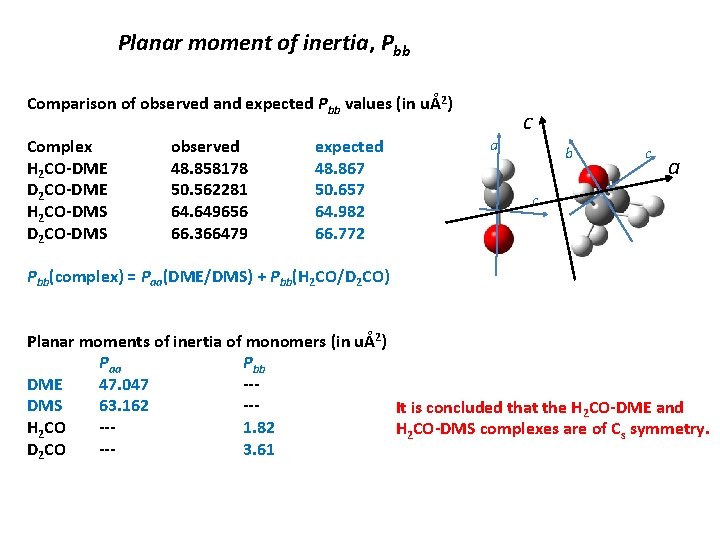
Planar moment of inertia, Pbb Comparison of observed and expected Pbb values (in uÅ2) Complex H 2 CO-DME D 2 CO-DME H 2 CO-DMS D 2 CO-DMS observed 48. 858178 50. 562281 64. 649656 66. 366479 expected 48. 867 50. 657 64. 982 66. 772 a c b c a c Pbb(complex) = Paa(DME/DMS) + Pbb(H 2 CO/D 2 CO) Planar moments of inertia of monomers (in uÅ2) Paa Pbb DME 47. 047 --DMS 63. 162 --It is concluded that the H 2 CO-DME and H 2 CO --1. 82 H 2 CO-DMS complexes are of Cs symmetry. D 2 CO --3. 61
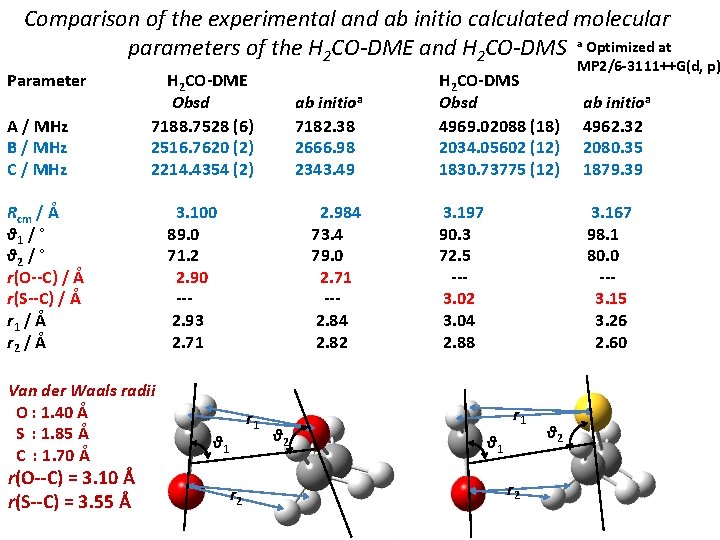
Comparison of the experimental and ab initio calculated molecular parameters of the H 2 CO-DME and H 2 CO-DMS a Optimized at Parameter A / MHz B / MHz C / MHz H 2 CO-DME Obsd 7188. 7528 (6) 2516. 7620 (2) 2214. 4354 (2) Rcm / Å θ 1 / ° θ 2 / ° r(O--C) / Å r(S--C) / Å r 1 / Å r 2 / Å Van der Waals radii O : 1. 40 Å S : 1. 85 Å C : 1. 70 Å r(O--C) = 3. 10 Å r(S--C) = 3. 55 Å ab initioa 7182. 38 2666. 98 2343. 49 3. 100 89. 0 71. 2 2. 90 --2. 93 2. 71 2. 984 73. 4 79. 0 2. 71 --2. 84 2. 82 r 1 θ 1 r 2 θ 2 H 2 CO-DMS Obsd 4969. 02088 (18) 2034. 05602 (12) 1830. 73775 (12) 3. 197 90. 3 72. 5 --3. 02 3. 04 2. 88 MP 2/6 -3111++G(d, p) ab initioa 4962. 32 2080. 35 1879. 39 3. 167 98. 1 80. 0 --3. 15 3. 26 2. 60 r 1 θ 1 r 2 θ 2
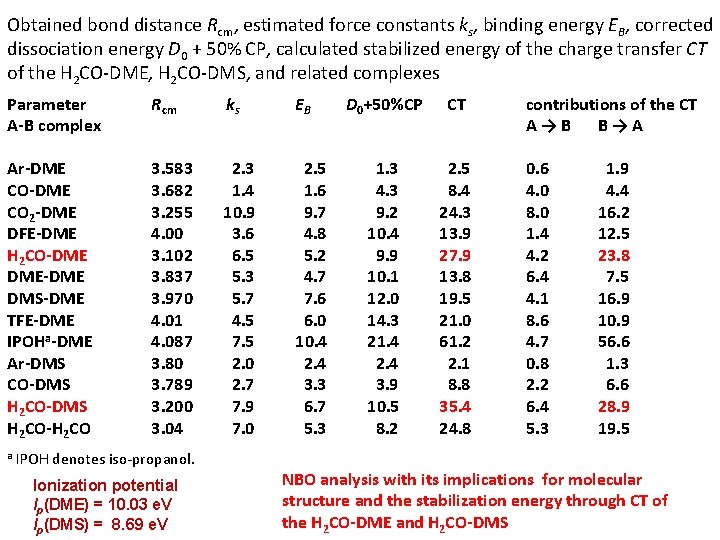
Obtained bond distance Rcm, estimated force constants ks, binding energy EB, corrected dissociation energy D 0 + 50% CP, calculated stabilized energy of the charge transfer CT of the H 2 CO-DME, H 2 CO-DMS, and related complexes Parameter A-B complex Rcm ks EB Ar-DME CO 2 -DME DFE-DME H 2 CO-DME DME-DME DMS-DME TFE-DME IPOHa-DME Ar-DMS CO-DMS H 2 CO-H 2 CO 3. 583 3. 682 3. 255 4. 00 3. 102 3. 837 3. 970 4. 01 4. 087 3. 80 3. 789 3. 200 3. 04 2. 3 1. 4 10. 9 3. 6 6. 5 5. 3 5. 7 4. 5 7. 5 2. 0 2. 7 7. 9 7. 0 2. 5 1. 6 9. 7 4. 8 5. 2 4. 7 7. 6 6. 0 10. 4 2. 4 3. 3 6. 7 5. 3 a IPOH denotes iso-propanol. Ionization potential Ip(DME) = 10. 03 e. V Ip(DMS) = 8. 69 e. V D 0+50%CP CT 1. 3 4. 3 9. 2 10. 4 9. 9 10. 1 12. 0 14. 3 21. 4 2. 4 3. 9 10. 5 8. 2 2. 5 8. 4 24. 3 13. 9 27. 9 13. 8 19. 5 21. 0 61. 2 2. 1 8. 8 35. 4 24. 8 contributions of the CT A→B B→A 0. 6 4. 0 8. 0 1. 4 4. 2 6. 4 4. 1 8. 6 4. 7 0. 8 2. 2 6. 4 5. 3 1. 9 4. 4 16. 2 12. 5 23. 8 7. 5 16. 9 10. 9 56. 6 1. 3 6. 6 28. 9 19. 5 NBO analysis with its implications for molecular structure and the stabilization energy through CT of the H 2 CO-DME and H 2 CO-DMS
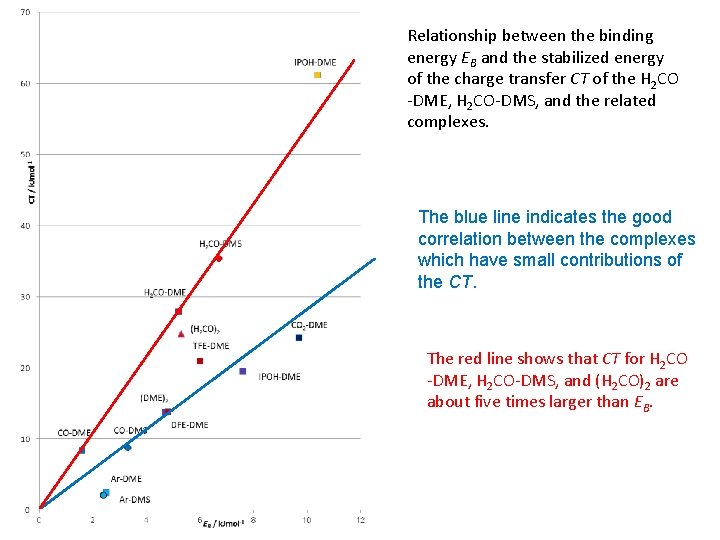
Relationship between the binding energy EB and the stabilized energy of the charge transfer CT of the H 2 CO -DME, H 2 CO-DMS, and the related complexes. The blue line indicates the good correlation between the complexes which have small contributions of the CT. The red line shows that CT for H 2 CO -DME, H 2 CO-DMS, and (H 2 CO)2 are about five times larger than EB.
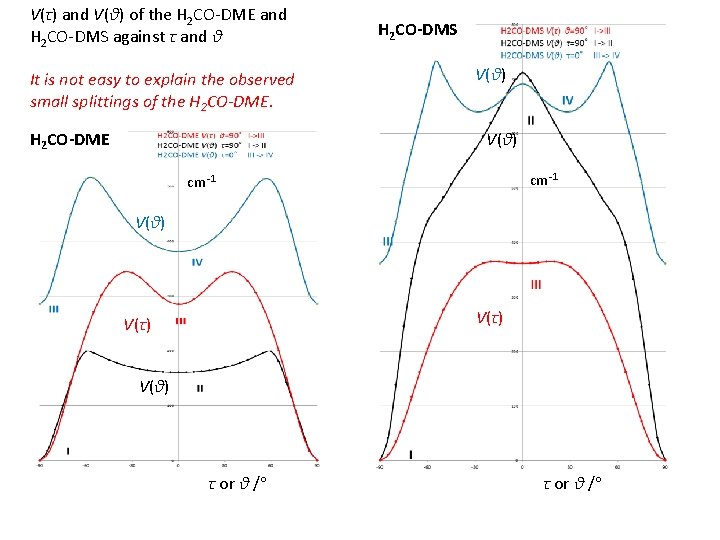
V(τ) and V(θ) of the H 2 CO-DME and H 2 CO-DMS against τ and θ It is not easy to explain the observed small splittings of the H 2 CO-DME H 2 CO-DMS V(θ) cm-1 V(θ) V(τ) V(θ) τ or θ /
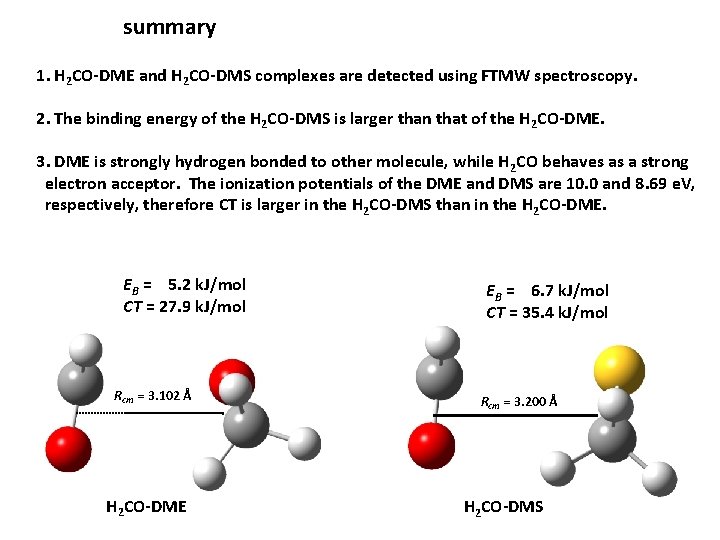
summary 1. H 2 CO-DME and H 2 CO-DMS complexes are detected using FTMW spectroscopy. 2. The binding energy of the H 2 CO-DMS is larger than that of the H 2 CO-DME. 3. DME is strongly hydrogen bonded to other molecule, while H 2 CO behaves as a strong electron acceptor. The ionization potentials of the DME and DMS are 10. 0 and 8. 69 e. V, respectively, therefore CT is larger in the H 2 CO-DMS than in the H 2 CO-DME. EB = 5. 2 k. J/mol CT = 27. 9 k. J/mol Rcm = 3. 102 Å H 2 CO-DME EB = 6. 7 k. J/mol CT = 35. 4 k. J/mol Rcm = 3. 200 Å H 2 CO-DMS
- Slides: 15