Terapia Neoadiuvante Revisione delle evidenze scientifiche Valentina Guarneri



- Slides: 27

Terapia Neoadiuvante Revisione delle evidenze scientifiche Valentina Guarneri Nonantola, 19 Novembre 2011

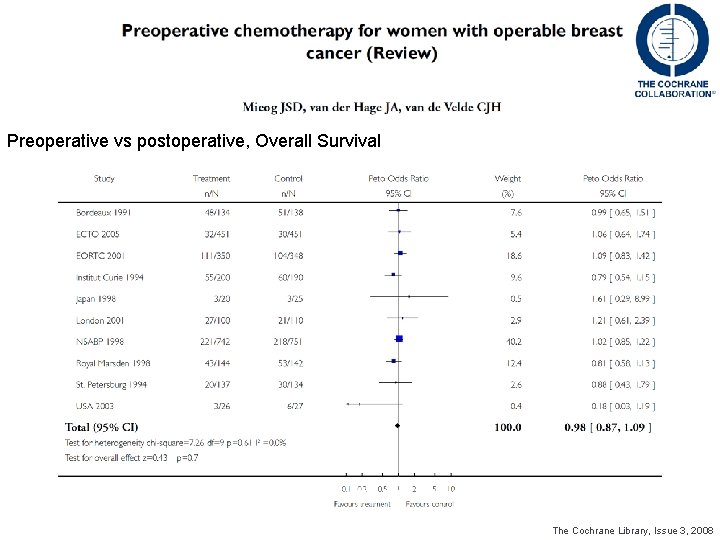
Preoperative vs postoperative, Overall Survival DDFS o OS? ? ? The Cochrane Library, Issue 3, 2008

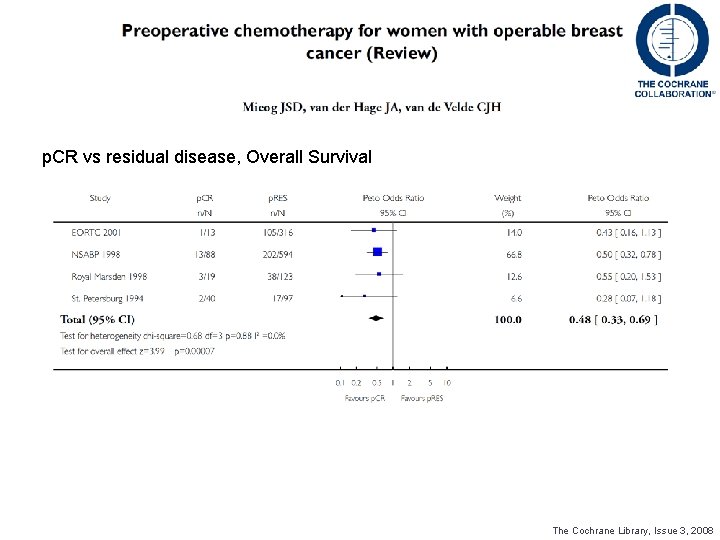
p. CR vs residual disease, Overall Survival The Cochrane Library, Issue 3, 2008

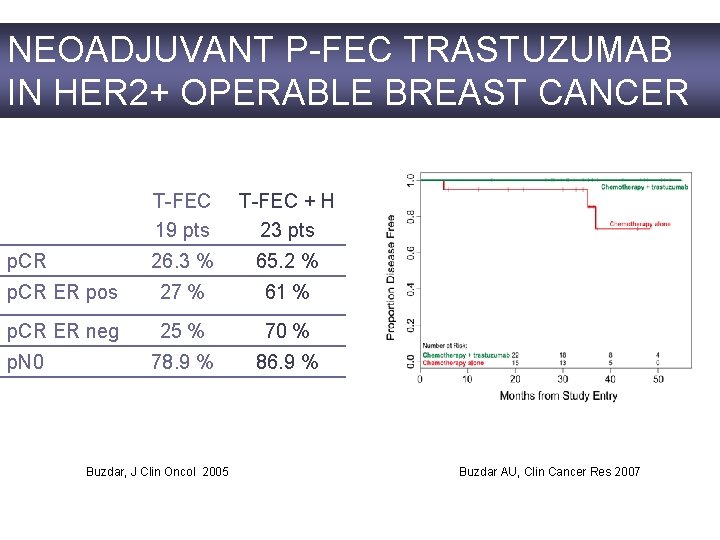
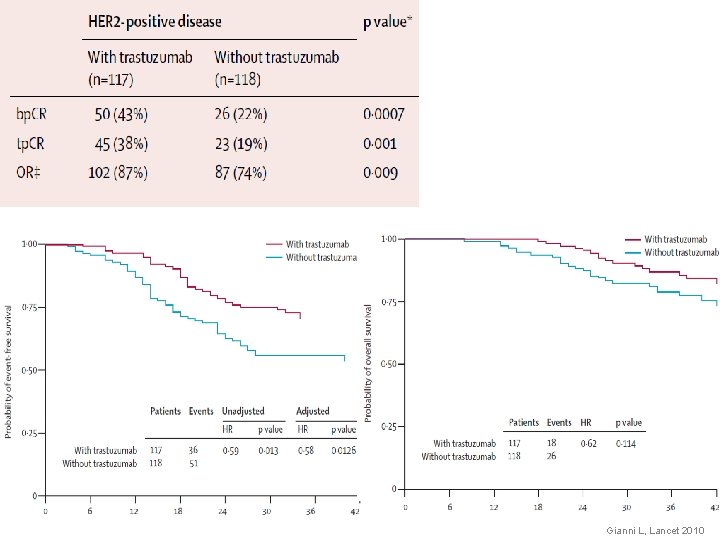
NEOADJUVANT P-FEC TRASTUZUMAB IN HER 2+ OPERABLE BREAST CANCER T-FEC 19 pts T-FEC + H 23 pts 26. 3 % 65. 2 % p. CR ER pos 27 % 61 % p. CR ER neg 25 % 70 % 78. 9 % 86. 9 % p. CR p. N 0 Buzdar, J Clin Oncol 2005 Buzdar AU, Clin Cancer Res 2007

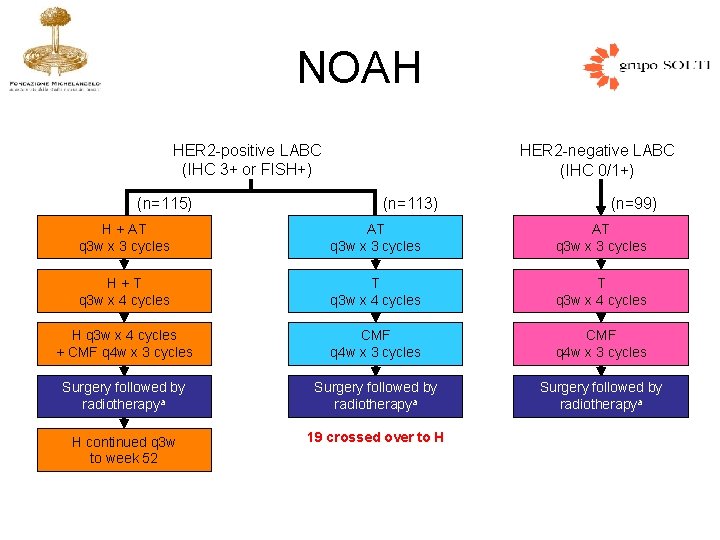
NOAH HER 2 -positive LABC (IHC 3+ or FISH+) (n=115) HER 2 -negative LABC (IHC 0/1+) (n=113) (n=99) H + AT q 3 w x 3 cycles H+T q 3 w x 4 cycles H q 3 w x 4 cycles + CMF q 4 w x 3 cycles Surgery followed by radiotherapya H continued q 3 w to week 52 19 crossed over to H

Gianni L, Lancet 2010

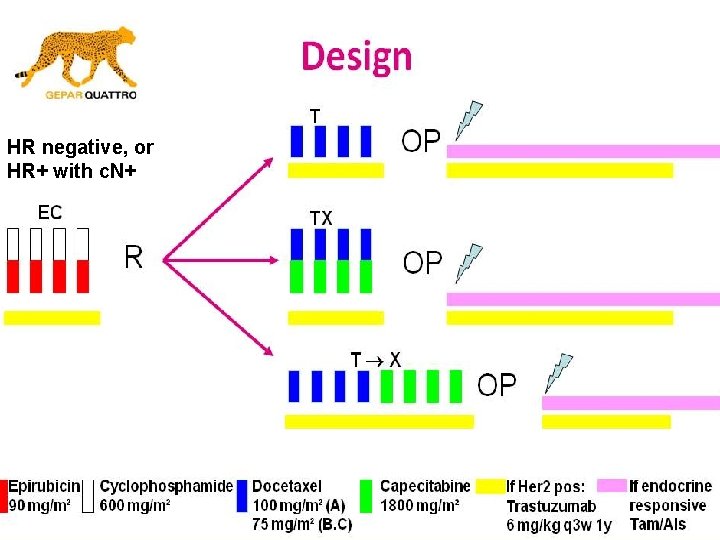
HR negative, or HR+ with c. N+

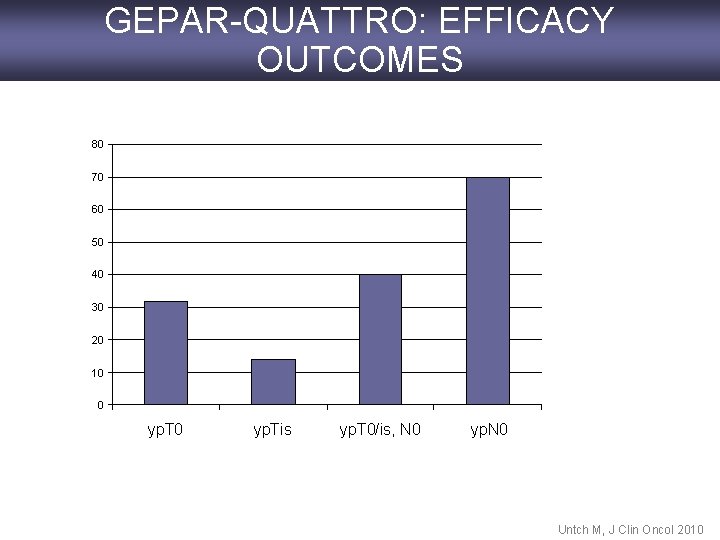
GEPAR-QUATTRO: EFFICACY OUTCOMES 80 70 60 50 40 30 20 10 0 yp. Tis yp. T 0/is, N 0 yp. N 0 Untch M, J Clin Oncol 2010

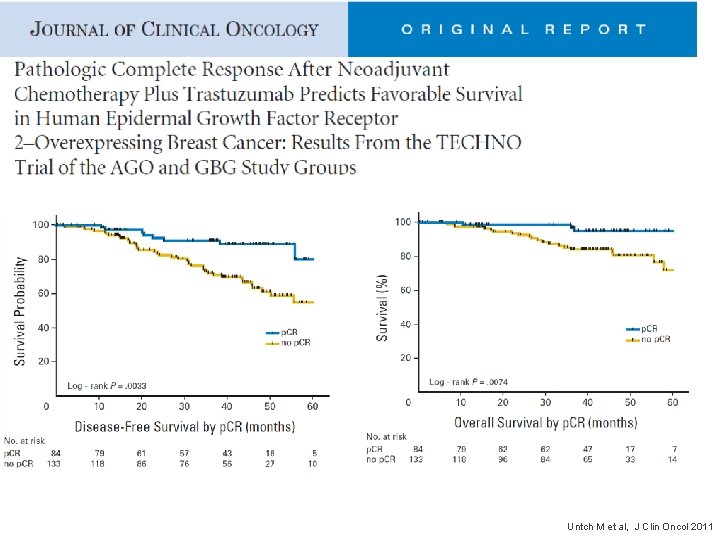
Untch M et al, J Clin Oncol 2011





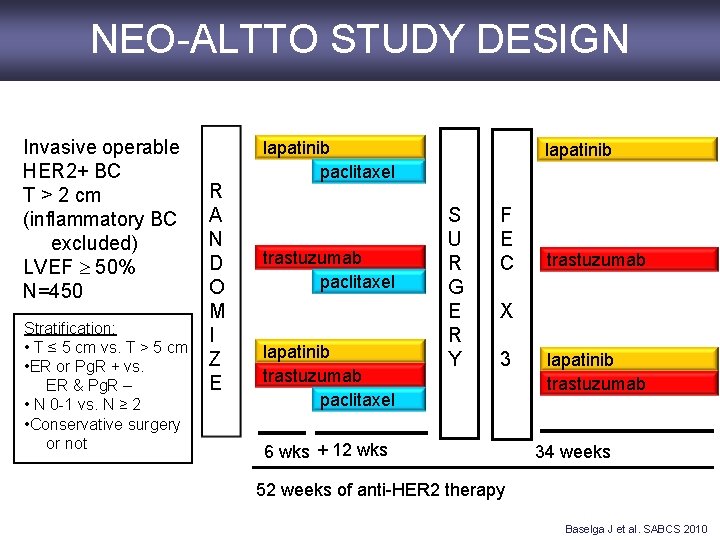
NEO-ALTTO STUDY DESIGN Invasive operable HER 2+ BC T > 2 cm (inflammatory BC excluded) LVEF 50% N=450 Stratification: • T ≤ 5 cm vs. T > 5 cm • ER or Pg. R + vs. ER & Pg. R – • N 0 -1 vs. N ≥ 2 • Conservative surgery or not R A N D O M I Z E lapatinib paclitaxel trastuzumab paclitaxel lapatinib S U R G E R Y F E C trastuzumab X 3 6 wks + 12 wks lapatinib trastuzumab 34 weeks 52 weeks of anti-HER 2 therapy Baselga J et al. SABCS 2010

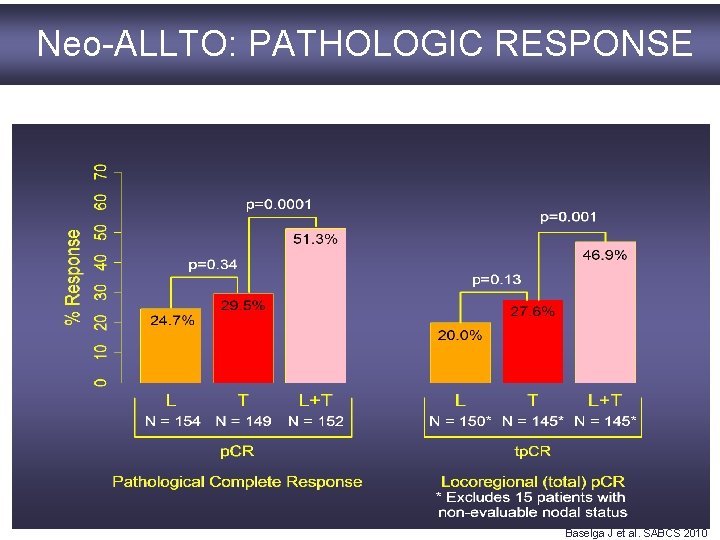
Neo-ALLTO: PATHOLOGIC RESPONSE L: lapatinib; T: trastuzumab; L+T: lapatinib plus trastuzumab p. CR pathologic complete response Baselga J et al. SABCS 2010

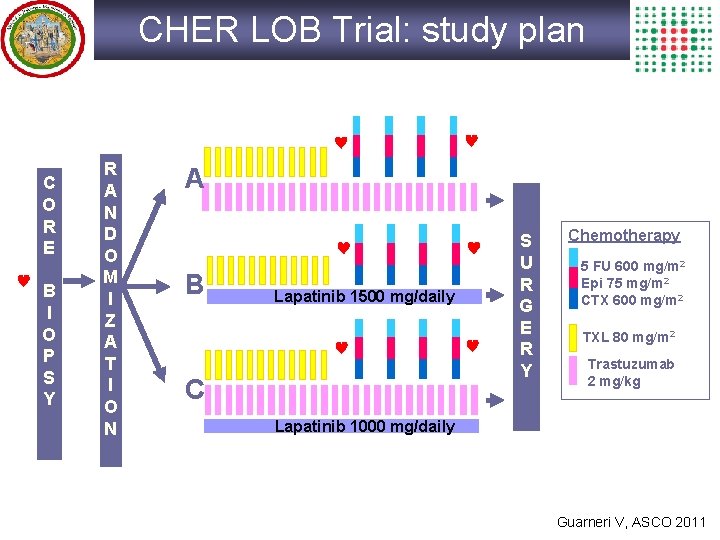
CHER LOB Trial: study plan C O R E B I O P S Y R A N D O M I Z A T I O N A B Lapatinib 1500 mg/daily C S U R G E R Y Chemotherapy 5 FU 600 mg/m 2 Epi 75 mg/m 2 CTX 600 mg/m 2 TXL 80 mg/m 2 Trastuzumab 2 mg/kg Lapatinib 1000 mg/daily Guarneri V, ASCO 2011

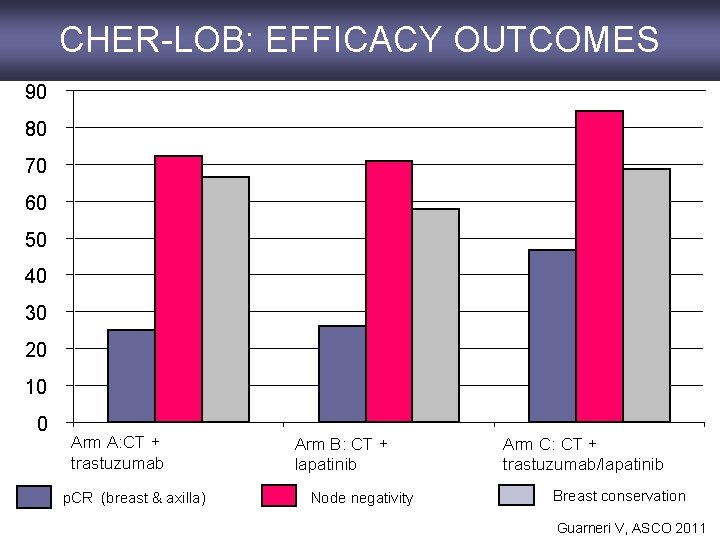
CHER-LOB: EFFICACY OUTCOMES 90 80 70 60 50 40 30 20 10 0 Arm A: CT + trastuzumab p. CR (breast & axilla) Arm B: CT + lapatinib Node negativity Arm C: CT + trastuzumab/lapatinib Breast conservation Guarneri V, ASCO 2011

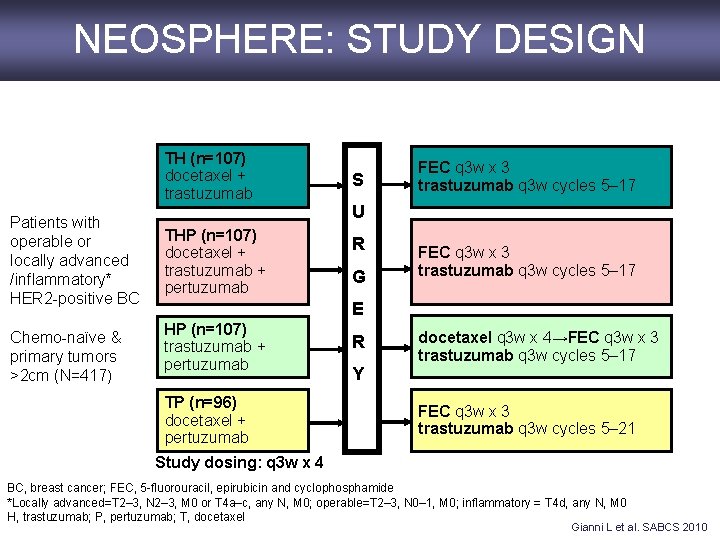
NEOSPHERE: STUDY DESIGN TH (n=107) docetaxel + trastuzumab Patients with operable or locally advanced /inflammatory* HER 2 -positive BC Chemo-naïve & primary tumors >2 cm (N=417) THP (n=107) docetaxel + trastuzumab + pertuzumab S FEC q 3 w x 3 trastuzumab q 3 w cycles 5– 17 U R G FEC q 3 w x 3 trastuzumab q 3 w cycles 5– 17 E HP (n=107) trastuzumab + pertuzumab TP (n=96) docetaxel + pertuzumab R Y docetaxel q 3 w x 4→FEC q 3 w x 3 trastuzumab q 3 w cycles 5– 17 FEC q 3 w x 3 trastuzumab q 3 w cycles 5– 21 Study dosing: q 3 w x 4 BC, breast cancer; FEC, 5 -fluorouracil, epirubicin and cyclophosphamide *Locally advanced=T 2– 3, N 2– 3, M 0 or T 4 a–c, any N, M 0; operable=T 2– 3, N 0– 1, M 0; inflammatory = T 4 d, any N, M 0 H, trastuzumab; P, pertuzumab; T, docetaxel Gianni L et al. SABCS 2010

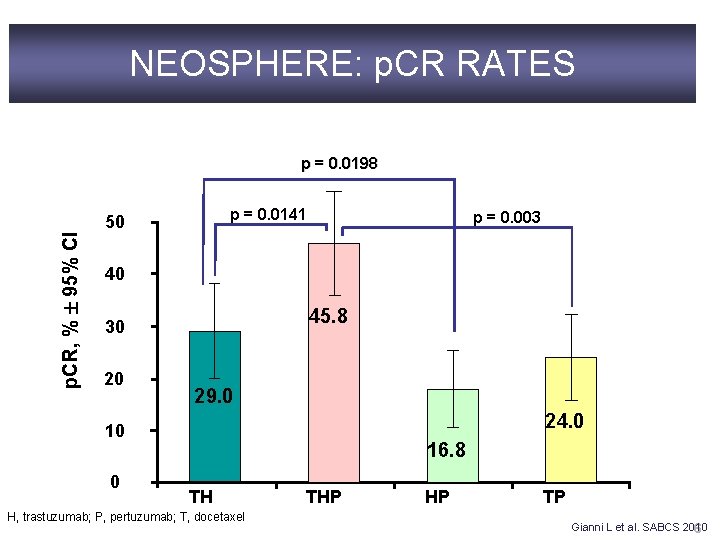
NEOSPHERE: p. CR RATES p. CR, % 95% CI p = 0. 0198 p = 0. 0141 50 p = 0. 003 40 45. 8 30 20 29. 0 24. 0 10 0 16. 8 TH H, trastuzumab; P, pertuzumab; T, docetaxel THP HP TP Gianni L et al. SABCS 2010 6

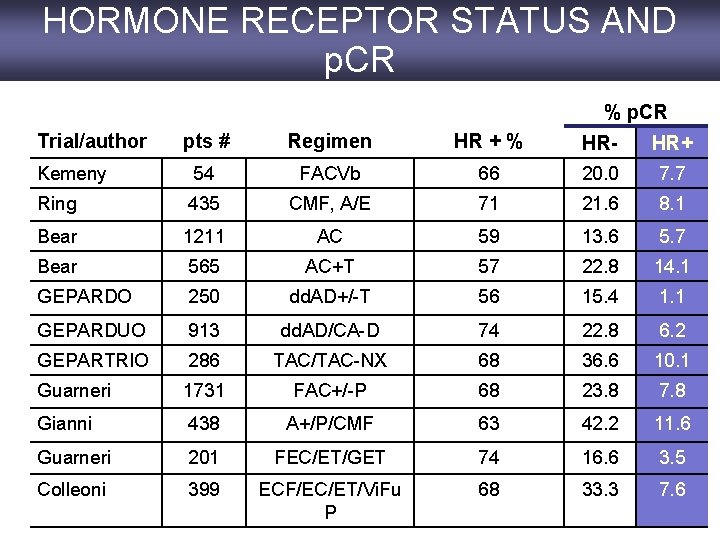
HORMONE RECEPTOR STATUS AND p. CR % p. CR Trial/author pts # Regimen HR + % HR- HR+ Kemeny 54 FACVb 66 20. 0 7. 7 Ring 435 CMF, A/E 71 21. 6 8. 1 Bear 1211 AC 59 13. 6 5. 7 Bear 565 AC+T 57 22. 8 14. 1 GEPARDO 250 dd. AD+/-T 56 15. 4 1. 1 GEPARDUO 913 dd. AD/CA-D 74 22. 8 6. 2 GEPARTRIO 286 TAC/TAC-NX 68 36. 6 10. 1 Guarneri 1731 FAC+/-P 68 23. 8 7. 8 Gianni 438 A+/P/CMF 63 42. 2 11. 6 Guarneri 201 FEC/ET/GET 74 16. 6 3. 5 Colleoni 399 ECF/EC/ET/Vi. Fu P 68 33. 3 7. 6

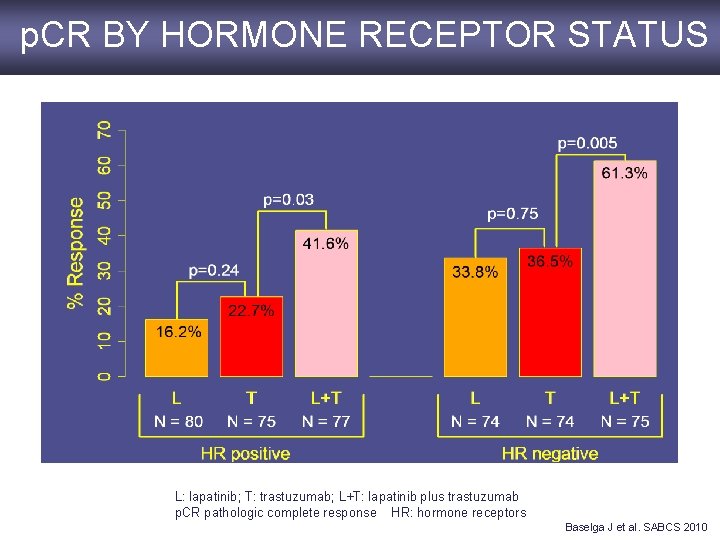
p. CR BY HORMONE RECEPTOR STATUS L: lapatinib; T: trastuzumab; L+T: lapatinib plus trastuzumab p. CR pathologic complete response HR: hormone receptors Baselga J et al. SABCS 2010

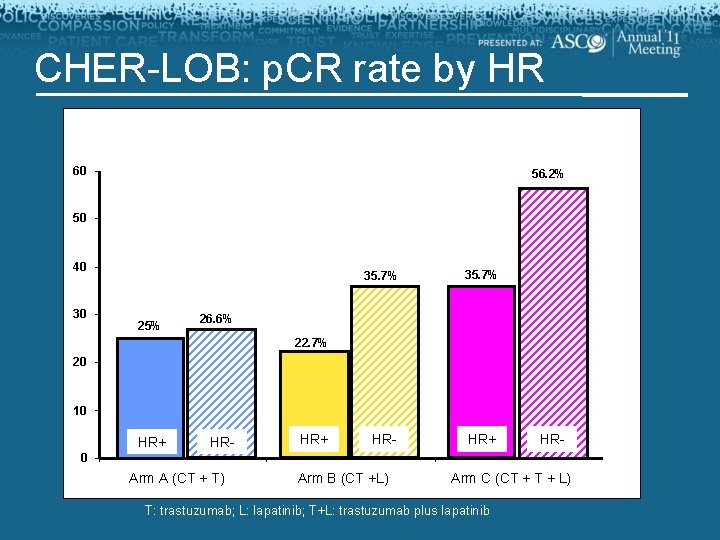
CHER-LOB: p. CR rate by HR 60 56. 2% 50 40 30 25% 35. 7% HR- HR+ 26. 6% 22. 7% 20 10 HR+ HR- 0 Arm A (CT + T) Arm B (CT +L) Arm C (CT + L) T: trastuzumab; L: lapatinib; T+L: trastuzumab plus lapatinib

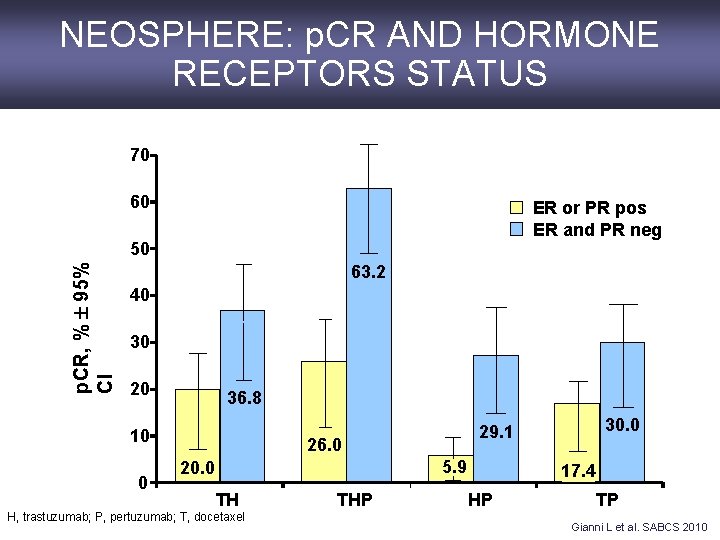
NEOSPHERE: p. CR AND HORMONE RECEPTORS STATUS 70 60 ER or PR pos ER and PR neg p. CR, % 95% CI 50 63. 2 40 30 20 36. 8 10 0 26. 0 5. 9 20. 0 TH H, trastuzumab; P, pertuzumab; T, docetaxel THP 30. 0 29. 1 17. 4 HP TP Gianni L et al. SABCS 2010

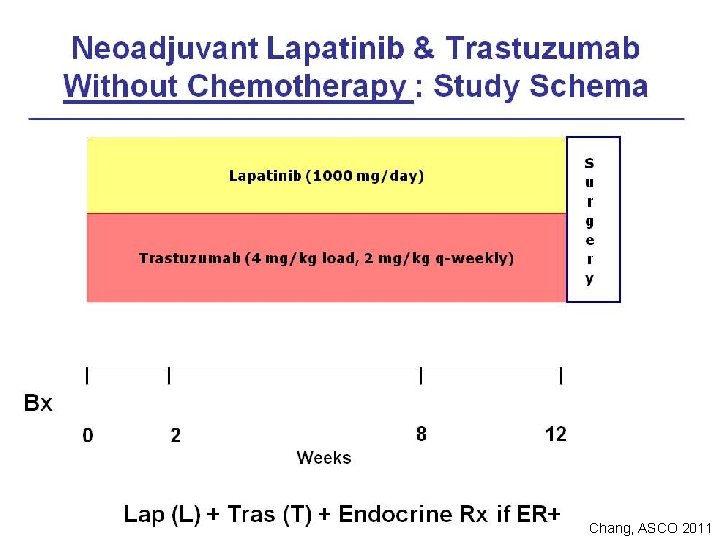
Chang, ASCO 2011

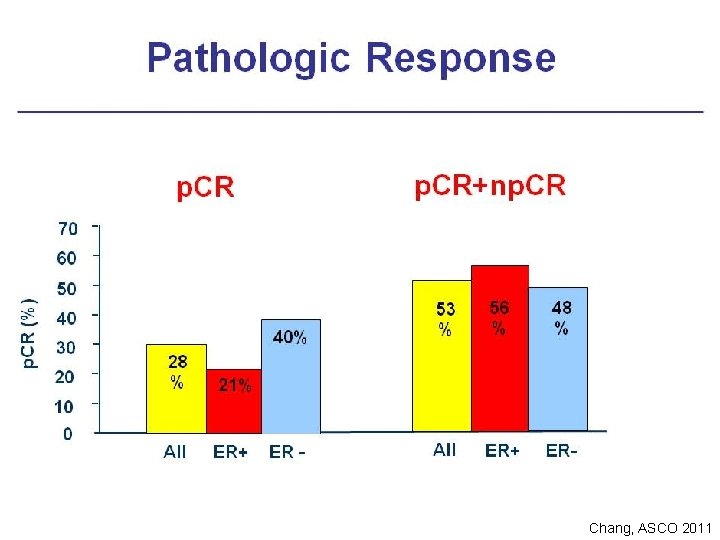
Chang, ASCO 2011

PST IN HER 2+ OPERABLE BREAST CANCER: KEY FINDINGS • • • Patient selection is mandatory for the integration of novel agents in cancer treatment Chemotherapy + trastuzumab is the gold standard Double-HER 2 blockade increases the p. CR rate Endocrine pathway is still important even in presence of HER 2 co-expression A dual anti-HER 2 blockade + endocrine therapy is promising The preoperative setting is ideal to test new combinations through the “window of opportunity model”
