Rotational Spectra of Adducts of Formaldehyde with Freons








- Slides: 27

Rotational Spectra of Adducts of Formaldehyde with Freons Qian Gou, 1 Gang Feng, 1 Luca Evangelisti, 1 Montserrat Vallejo-López, 2 Alberto Lesarri, 2 Walther Caminati, 1 Emilio J. Cocinero 3 1 Dipartimento di Chimica “G. Ciamician” dell’Università, Via Selmi 2, I-40126 Bologna, Italy 2 Departamento de Química Física y Química Inorganica, Facultad de Ciencias, Universidad de Valladolid, E- 47011 Valladolid, Spain 3 Departamento de Quimica Fisica, Facultad de Ciencia y Tecnologia, Universidad del Pais Vasco, E-48080 Bilbao, Spain. 2013. 06. 18. Columbus

Introduction J. Am. Chem. Soc. 1999, 121, 10098. Angew. Chem. Int. Ed. , 2006 , 45, 2438. Angew. Chem. Int. Ed. , 2011 , 50, 7871. With formaldehyde 2

I. Difluoromethane − Formaldehyde CH 2 F 2 − H 2 CO Phys. Chem. Phys. , 2013, 15, 6714. 3

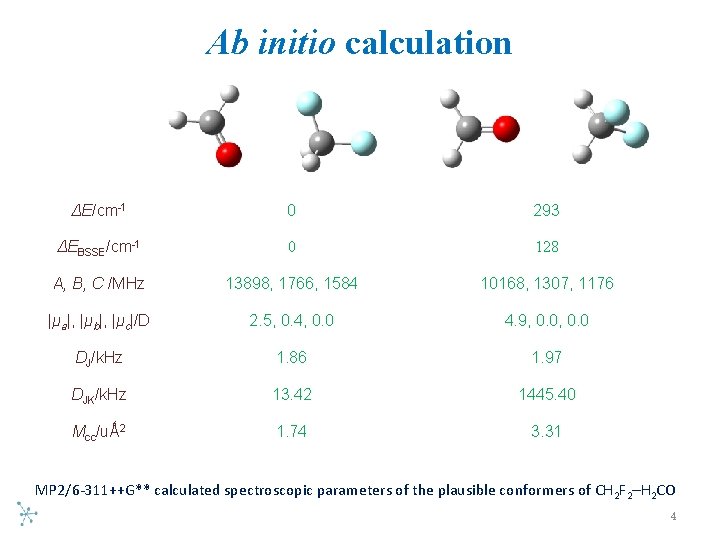
Ab initio calculation ΔE/cm-1 0 293 ΔEBSSE/cm-1 0 128 A, B, C /MHz 13898, 1766, 1584 10168, 1307, 1176 |μa|, |μb|, |μc|/D 2. 5, 0. 4, 0. 0 4. 9, 0. 0 DJ/k. Hz 1. 86 1. 97 DJK/k. Hz 13. 42 1445. 40 Mcc/uǺ 2 1. 74 3. 31 MP 2/6 311++G** calculated spectroscopic parameters of the plausible conformers of CH 2 F 2–H 2 CO 4

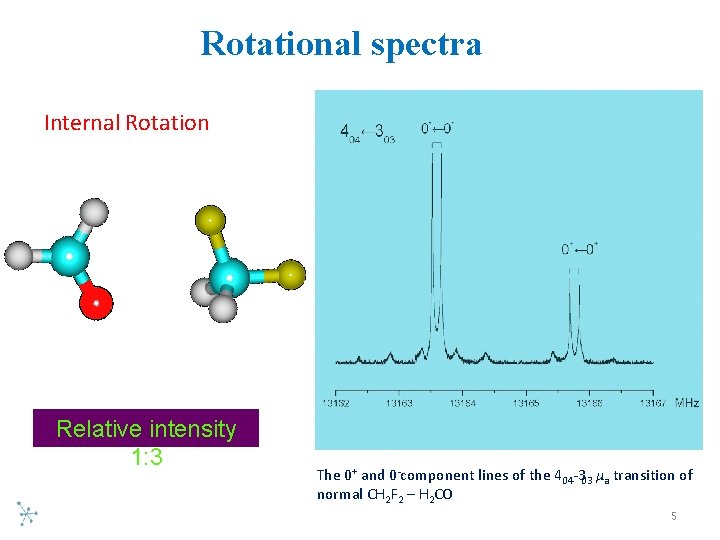
Rotational spectra Internal Rotation Relative intensity 1: 3 The 0+ and 0 component lines of the 404 303 μa transition of normal CH 2 F 2 – H 2 CO 5

Rotational spectra Spectroscopic parameters for complex of CH 2 F 2–H 2 CO including 13 C isotopologues CH 2 F 2 -HCOH(0+) CH 2 F 2 -HCOH(0 -) 13 CH 2 F 2 -HCOH(0 A/MHz 13725. 398(3) 13719. 351(3) 13675(2) 13671(2) B/MHz 1737. 258(1) 1736. 720(1) 1731. 0654(8) 1698. 7766(8) C/MHz 1559. 247(1) 1559. 218(1) 1554. 1606(8) 1528. 0736(8) DJ/k. Hz 2. 33(1) 2. 29(2) 2. 22(2) DJK/k. Hz 21. 25(5) 20. 78(5) [20. 78] d 1/k. Hz -0. 24(1) -0. 23(1) [-0. 23] d 2 k. Hz 0. 029(6) 0. 022(6) [0. 022] /k. Hz 2. 59 2. 13 4. 94 2. 98 N 23 23 9 9 -) CH 2 F 2 -H 13 COH(0 -) It’s not possible to determine the vibrational energy spacing (∆E) between the two tunneling states 6

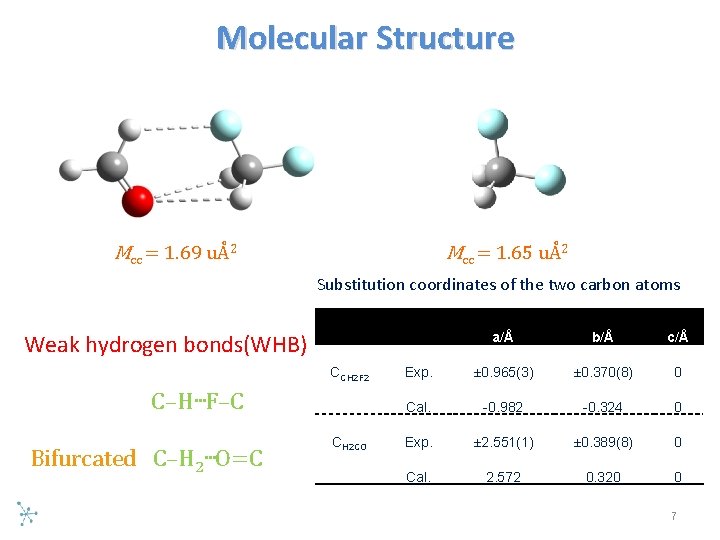
Molecular Structure Mcc = 1. 69 uÅ2 Mcc = 1. 65 uÅ2 Substitution coordinates of the two carbon atoms Weak hydrogen bonds(WHB) CCH 2 F 2 C–H…F–C Bifurcated C–H 2 …O=C CH 2 CO a/Å b/Å c/Å Exp. ± 0. 965(3) ± 0. 370(8) 0 Cal. -0. 982 -0. 324 0 Exp. ± 2. 551(1) ± 0. 389(8) 0 Cal. 2. 572 0. 320 0 7

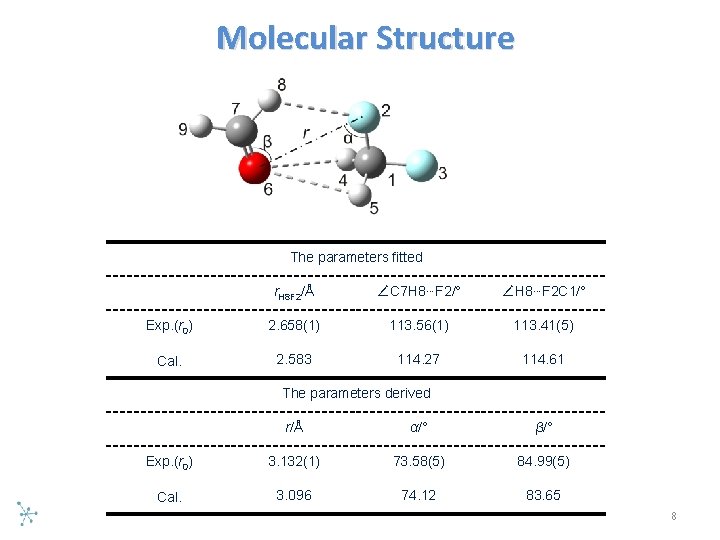
Molecular Structure The parameters fitted r. H 8 F 2/Å ∠C 7 H 8…F 2/° ∠H 8…F 2 C 1/° Exp. (r 0) 2. 658(1) 113. 56(1) 113. 41(5) Cal. 2. 583 114. 27 114. 61 The parameters derived r/Å α/° β/° Exp. (r 0) 3. 132(1) 73. 58(5) 84. 99(5) Cal. 3. 096 74. 12 83. 65 8

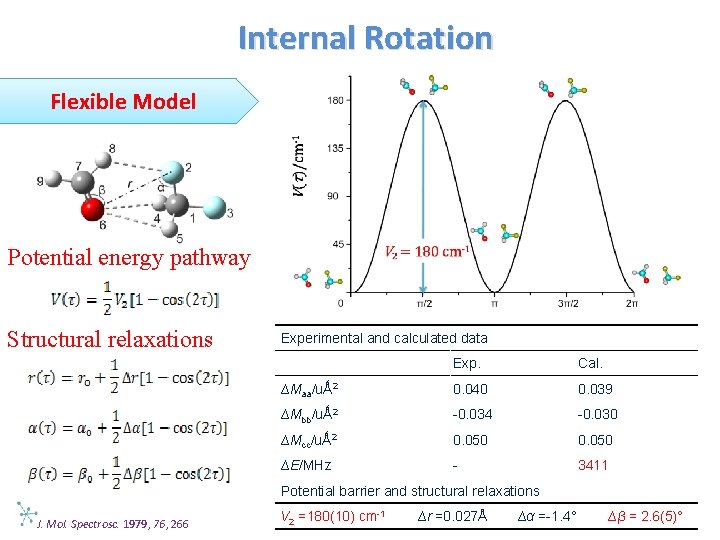
Internal Rotation Flexible Model Potential energy pathway Structural relaxations Experimental and calculated data Exp. Cal. ∆Maa/uǺ 2 0. 040 0. 039 ∆Mbb/uǺ 2 -0. 034 -0. 030 ∆Mcc/uǺ 2 0. 050 ∆E/MHz - 3411 Potential barrier and structural relaxations J. Mol. Spectrosc. 1979, 76, 266 V 2 =180(10) cm-1 ∆r =0. 027Å ∆α =-1. 4° ∆β = 2. 6(5)°

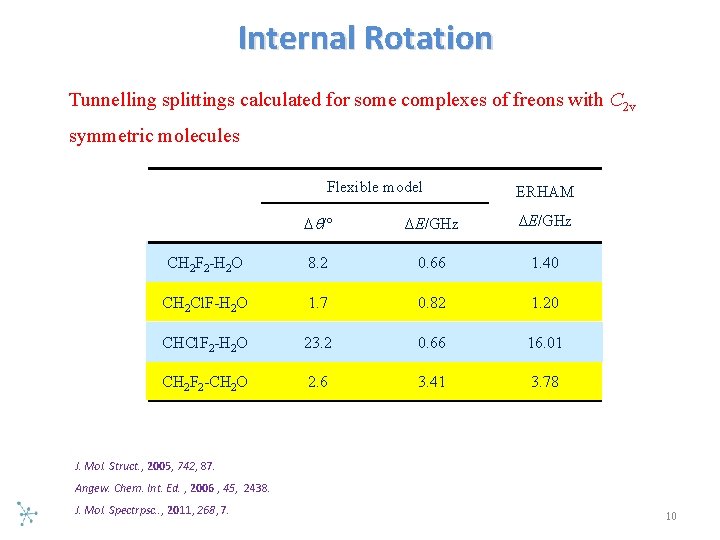
Internal Rotation Tunnelling splittings calculated for some complexes of freons with C 2 v symmetric molecules Flexible model ERHAM /° ΔE/GHz CH 2 F 2 -H 2 O 8. 2 0. 66 1. 40 CH 2 Cl. F-H 2 O 1. 7 0. 82 1. 20 CHCl. F 2 -H 2 O 23. 2 0. 66 16. 01 CH 2 F 2 -CH 2 O 2. 6 3. 41 3. 78 J. Mol. Struct. , 2005, 742, 87. Angew. Chem. Int. Ed. , 2006 , 45, 2438. J. Mol. Spectrpsc. . , 2011, 268, 7. 10

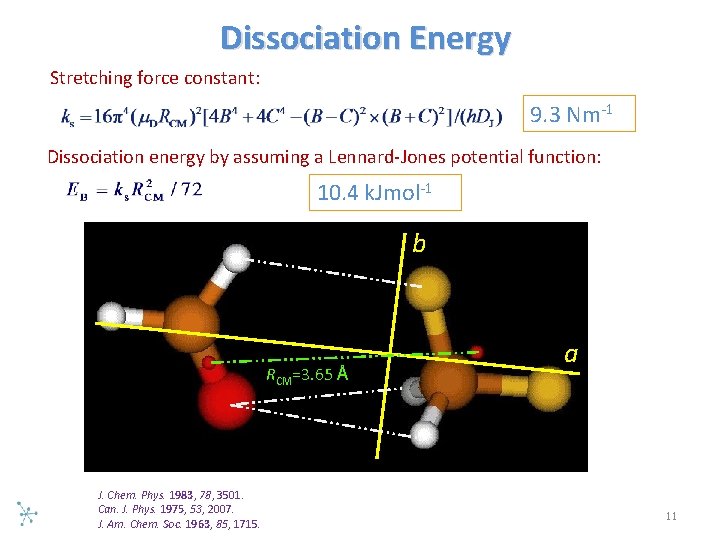
Dissociation Energy Stretching force constant: 9. 3 Nm 1 Dissociation energy by assuming a Lennard Jones potential function: 10. 4 k. Jmol 1 b RCM=3. 65 Å J. Chem. Phys. 1983, 78, 3501. Can. J. Phys. 1975, 53, 2007. J. Am. Chem. Soc. 1963, 85, 1715. a 11

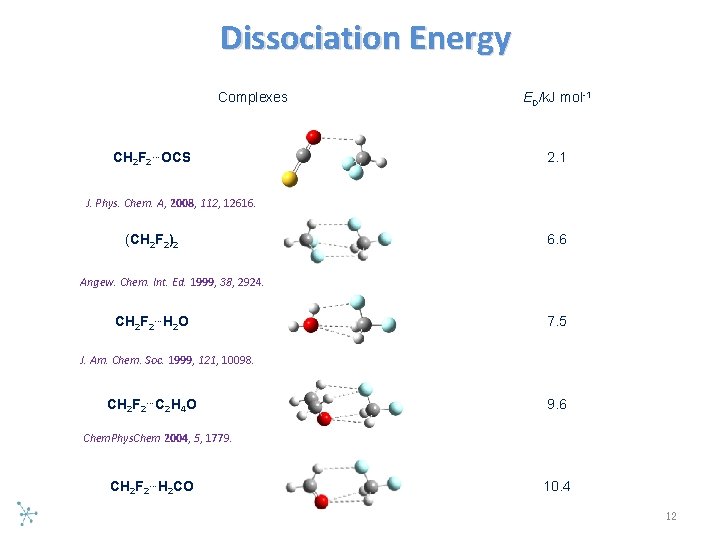
Dissociation Energy Complexes CH 2 F 2…OCS ED/k. J mol-1 2. 1 J. Phys. Chem. A, 2008, 112, 12616. (CH 2 F 2)2 6. 6 Angew. Chem. Int. Ed. 1999, 38, 2924. CH 2 F 2…H 2 O 7. 5 J. Am. Chem. Soc. 1999, 121, 10098. CH 2 F 2…C 2 H 4 O 9. 6 Chem. Phys. Chem 2004, 5, 1779. CH 2 F 2…H 2 CO 10. 4 12

II. Chlorofluoromethane− Formaldehyde CH 2 FCl − H 2 CO 13

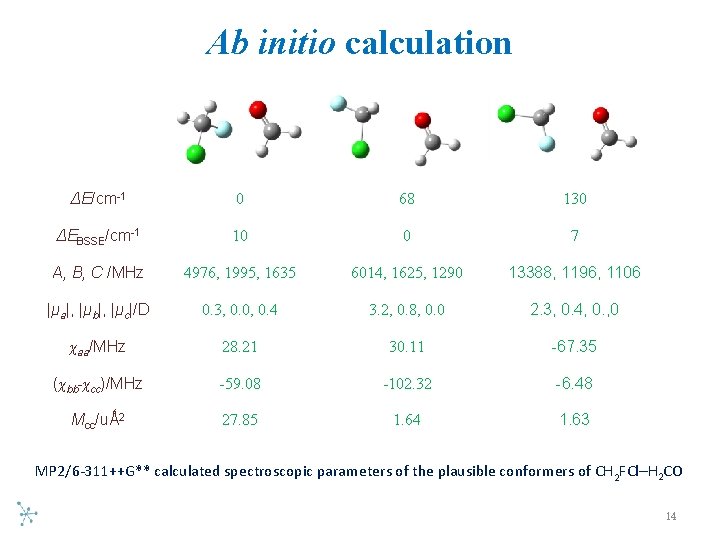
Ab initio calculation ΔE/cm-1 0 68 130 ΔEBSSE/cm-1 10 0 7 A, B, C /MHz 4976, 1995, 1635 6014, 1625, 1290 13388, 1196, 1106 |μa|, |μb|, |μc|/D 0. 3, 0. 0, 0. 4 3. 2, 0. 8, 0. 0 2. 3, 0. 4, 0. , 0 χaa/MHz 28. 21 30. 11 -67. 35 (χbb-χcc)/MHz -59. 08 -102. 32 -6. 48 Mcc/uǺ 2 27. 85 1. 64 1. 63 MP 2/6 311++G** calculated spectroscopic parameters of the plausible conformers of CH 2 FCl–H 2 CO 14

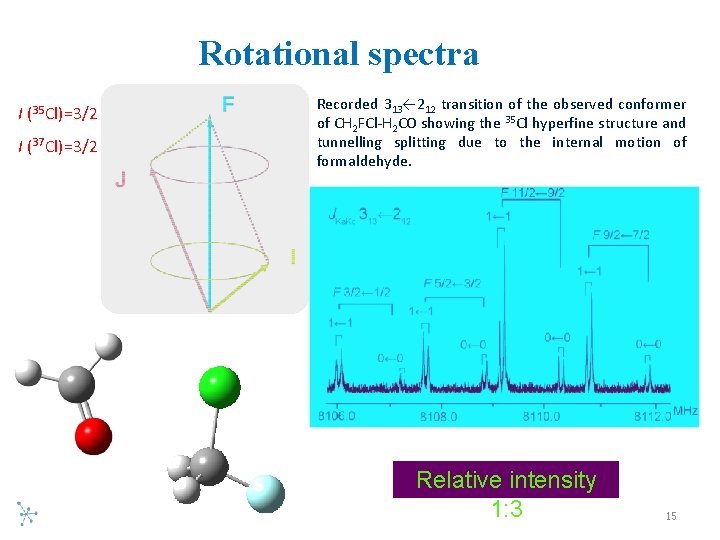
Rotational spectra I (35 Cl)=3/2 I (37 Cl)=3/2 Recorded 313← 212 transition of the observed conformer of CH 2 FCl H 2 CO showing the 35 Cl hyperfine structure and tunnelling splitting due to the internal motion of formaldehyde. Relative intensity 1: 3 15

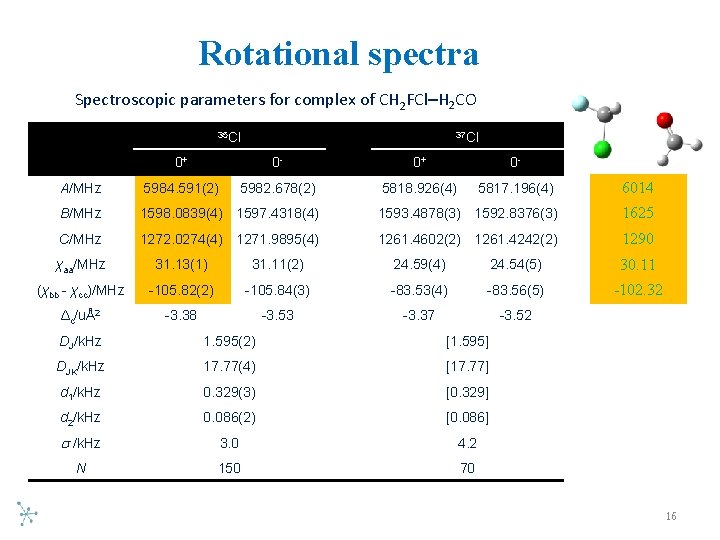
Rotational spectra Spectroscopic parameters for complex of CH 2 FCl–H 2 CO 35 Cl 37 Cl 0+ 0 - A/MHz 5984. 591(2) 5982. 678(2) 5818. 926(4) 5817. 196(4) 6014 B/MHz 1598. 0839(4) 1597. 4318(4) 1593. 4878(3) 1592. 8376(3) 1625 C/MHz 1272. 0274(4) 1271. 9895(4) 1261. 4602(2) 1261. 4242(2) 1290 χaa/MHz 31. 13(1) 31. 11(2) 24. 59(4) 24. 54(5) 30. 11 (χbb - χcc)/MHz -105. 82(2) -105. 84(3) -83. 53(4) -83. 56(5) -102. 32 Δc/uÅ2 -3. 38 -3. 53 -3. 37 -3. 52 DJ/k. Hz 1. 595(2) [1. 595] DJK/k. Hz 17. 77(4) [17. 77] d 1/k. Hz 0. 329(3) [0. 329] d 2/k. Hz 0. 086(2) [0. 086] /k. Hz 3. 0 4. 2 N 150 70 16

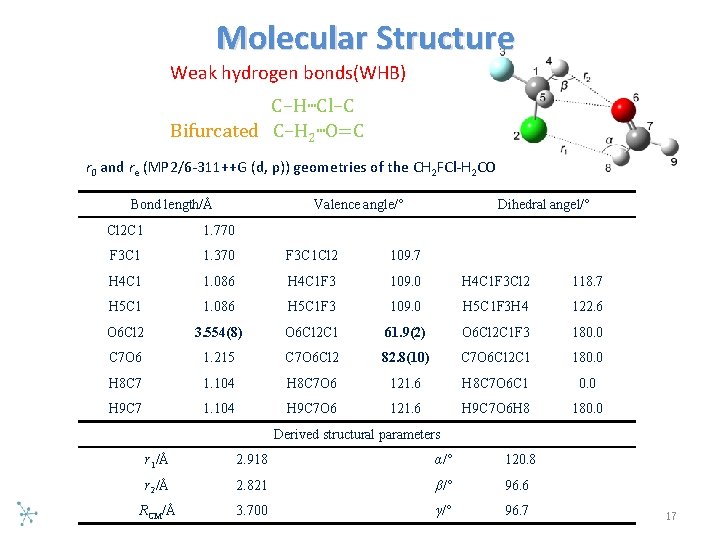
Molecular Structure Weak hydrogen bonds(WHB) C–H…Cl–C Bifurcated C–H 2…O=C r 0 and re (MP 2/6 311++G (d, p)) geometries of the CH 2 FCl H 2 CO Bond length/Å Valence angle/° Dihedral angel/° Cl 2 C 1 1. 770 F 3 C 1 1. 370 F 3 C 1 Cl 2 109. 7 H 4 C 1 1. 086 H 4 C 1 F 3 109. 0 H 4 C 1 F 3 Cl 2 118. 7 H 5 C 1 1. 086 H 5 C 1 F 3 109. 0 H 5 C 1 F 3 H 4 122. 6 O 6 Cl 2 3. 554(8) O 6 Cl 2 C 1 61. 9(2) O 6 Cl 2 C 1 F 3 180. 0 C 7 O 6 1. 215 C 7 O 6 Cl 2 82. 8(10) C 7 O 6 Cl 2 C 1 180. 0 H 8 C 7 1. 104 H 8 C 7 O 6 121. 6 H 8 C 7 O 6 C 1 0. 0 H 9 C 7 1. 104 H 9 C 7 O 6 121. 6 H 9 C 7 O 6 H 8 180. 0 Derived structural parameters r 1/Å 2. 918 α/° 120. 8 r 2/Å 2. 821 β/° 96. 6 RCM/Å 3. 700 γ/° 96. 7 17

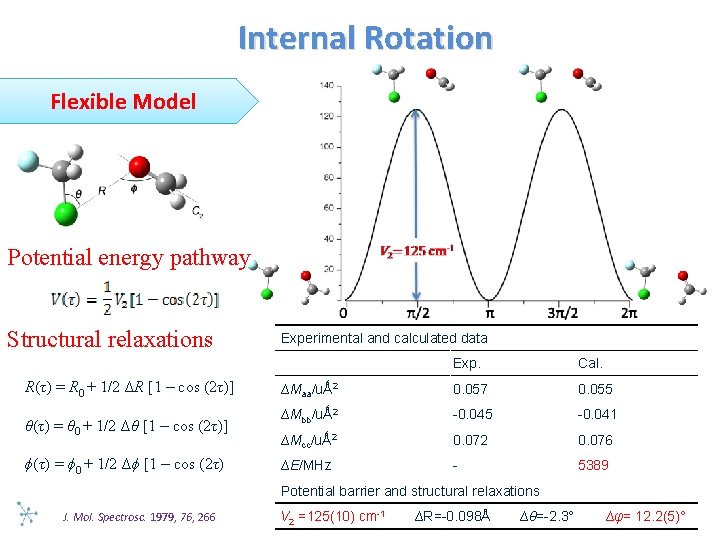
Internal Rotation Flexible Model Potential energy pathway Structural relaxations R(τ) = R 0 + 1/2 ΔR [1 – cos (2τ)] θ(τ) = θ 0 + 1/2 Δθ [1 – cos (2τ)] ϕ(τ) = ϕ 0 + 1/2 Δϕ [1 – cos (2τ) Experimental and calculated data Exp. Cal. ∆Maa/uǺ 2 0. 057 0. 055 ∆Mbb/uǺ 2 -0. 045 -0. 041 ∆Mcc/uǺ 2 0. 076 ∆E/MHz - 5389 Potential barrier and structural relaxations J. Mol. Spectrosc. 1979, 76, 266 V 2 =125(10) cm-1 ∆R=-0. 098Å ∆θ=-2. 3° ∆φ= 12. 2(5)°

Dissociation Energy Stretching force constant: Dissociation energy by assuming a Lennard Jones potential function: complex ks/Nm-1 EB/k. Jmol-1 CH 2 FCl…H 2 O 8. 6 8. 5 CH 2 FCl…H 2 CO 8. 6 9. 8 J. Chem. Phys. 1983, 78, 3501. Can. J. Phys. 1975, 53, 2007. J. Am. Chem. Soc. 1963, 85, 1715.

III. Chlorotrifluoromethane − Formaldehyde CF 3 Cl − H 2 CO 20

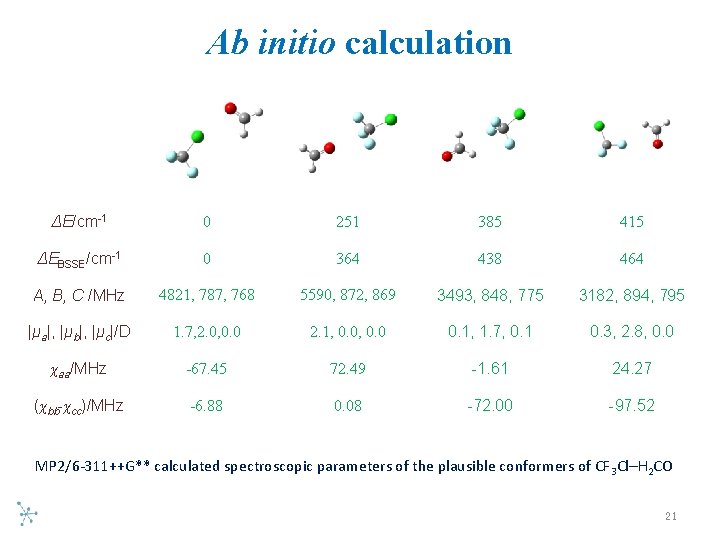
Ab initio calculation ΔE/cm-1 0 251 385 415 ΔEBSSE/cm-1 0 364 438 464 A, B, C /MHz 4821, 787, 768 5590, 872, 869 3493, 848, 775 3182, 894, 795 |μa|, |μb|, |μc|/D 1. 7, 2. 0, 0. 0 2. 1, 0. 0 0. 1, 1. 7, 0. 1 0. 3, 2. 8, 0. 0 χaa/MHz -67. 45 72. 49 -1. 61 24. 27 (χbb-χcc)/MHz -6. 88 0. 08 -72. 00 -97. 52 MP 2/6 311++G** calculated spectroscopic parameters of the plausible conformers of CF 3 Cl–H 2 CO 21

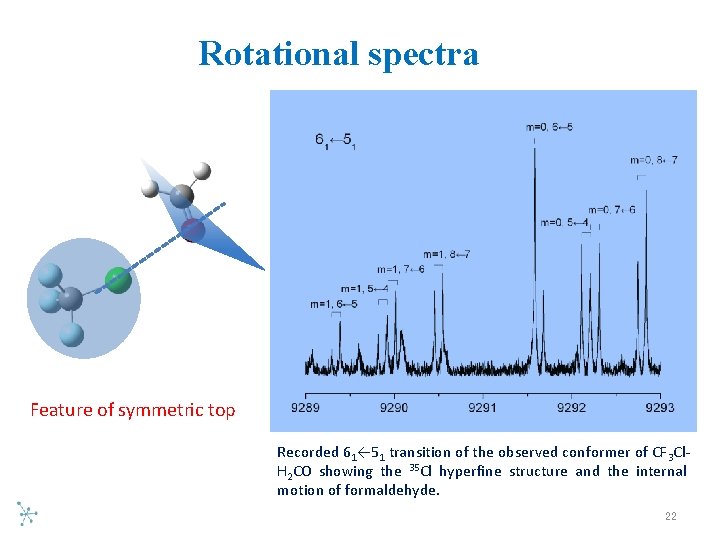
Rotational spectra Feature of symmetric top Recorded 61← 51 transition of the observed conformer of CF 3 Cl H 2 CO showing the 35 Cl hyperfine structure and the internal motion of formaldehyde. 22

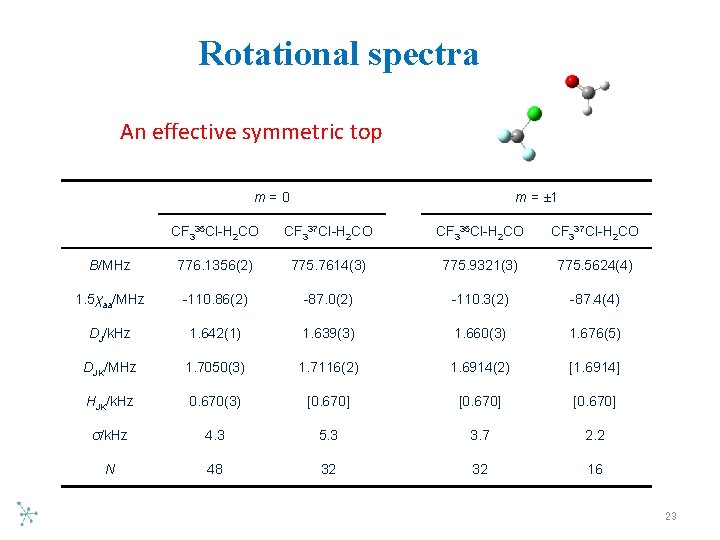
Rotational spectra An effective symmetric top m=0 m = ± 1 CF 335 Cl-H 2 CO CF 337 Cl-H 2 CO B/MHz 776. 1356(2) 775. 7614(3) 775. 9321(3) 775. 5624(4) 1. 5χaa/MHz -110. 86(2) -87. 0(2) -110. 3(2) -87. 4(4) DJ/k. Hz 1. 642(1) 1. 639(3) 1. 660(3) 1. 676(5) DJK/MHz 1. 7050(3) 1. 7116(2) 1. 6914(2) [1. 6914] HJK/k. Hz 0. 670(3) [0. 670] σ/k. Hz 4. 3 5. 3 3. 7 2. 2 N 48 32 32 16 23

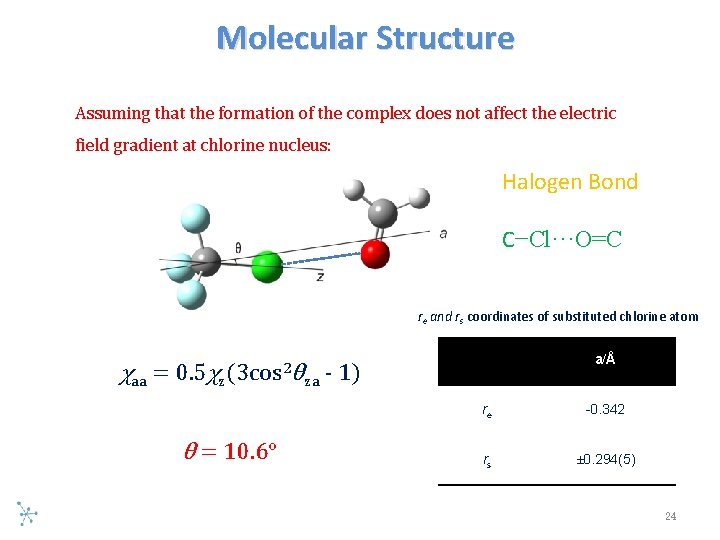
Molecular Structure Assuming that the formation of the complex does not affect the electric field gradient at chlorine nucleus: Halogen Bond C−Cl···O=C re and rs coordinates of substituted chlorine atom χaa = 0. 5χz (3 cos 2 θ = 10. 6° a/Å θza - 1) re -0. 342 rs ± 0. 294(5) 24

Dissociation Energy Stretching force constant: ks = 128π4(μRCM)2 B 4/h. DJ Dissociation energy by assuming a Lennard Jones potential function: EB = 1/72 ks. R 2 CM complex ks/Nm-1 EB/k. Jmol-1 CF 3 Cl…H 2 O 5. 0 7. 7 CF 3 Cl…H 2 CO 6. 3 11. 0 J. Chem. Phys. 1983, 78, 3501. Can. J. Phys. 1975, 53, 2007. J. Am. Chem. Soc. 1963, 85, 1715.

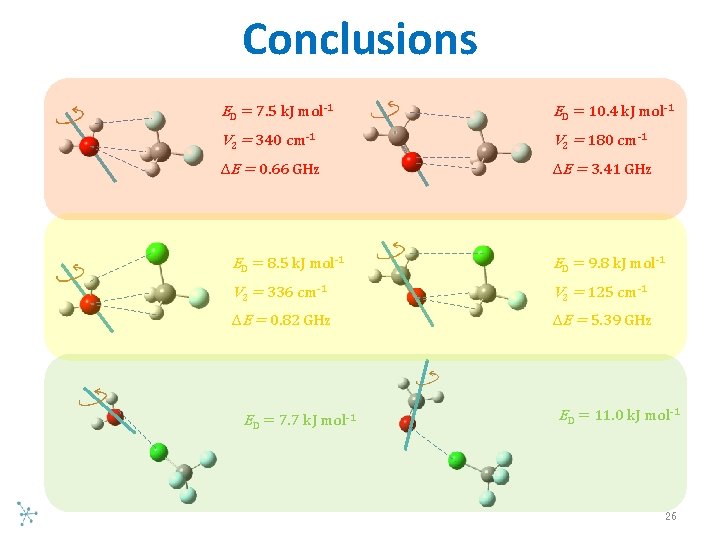
Conclusions ED = 7. 5 k. J mol-1 ED = 10. 4 k. J mol-1 V 2 = 340 cm-1 V 2 = 180 cm-1 ∆E = 0. 66 GHz ∆E = 3. 41 GHz ED = 8. 5 k. J mol-1 ED = 9. 8 k. J mol-1 V 2 = 336 cm-1 V 2 = 125 cm-1 ∆E = 0. 82 GHz ∆E = 5. 39 GHz ED = 7. 7 k. J mol-1 ED = 11. 0 k. J mol-1 26

Acknowledgement Prof. W. Caminati Dr. L. B. Favero Prof. S. Melandri Dr. A. Maris Dr. L. Evangelisti Thanks for attention! Dr. F. Gang Ms. C. Calabrese Mr. L. Spada 27