The Mole Formulas Dr Ron Rusay Mole Mass



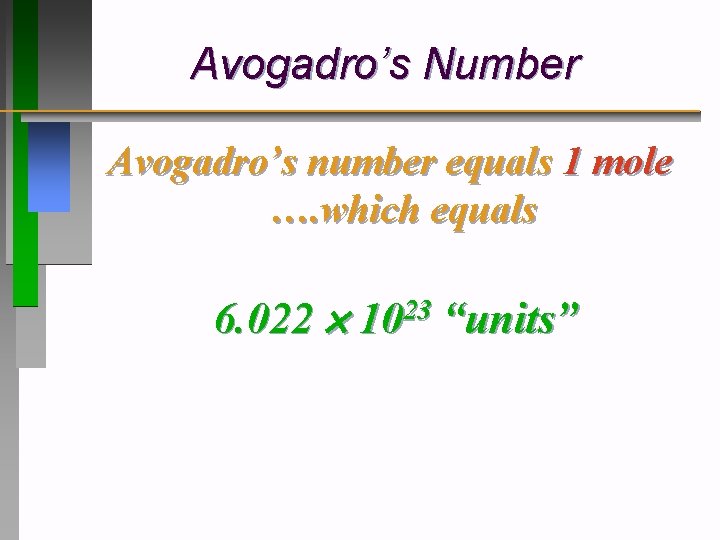



































- Slides: 44

The Mole & Formulas Dr. Ron Rusay

Mole - Mass Relationships Chemical Reactions Stoichiometry The Mole • % Composition: Determining the Formula of an Unknown Compound • Writing and Balancing Chemical Equations • Calculating the amounts of Reactant and Product • Limiting Reactant

The Mole • The number of carbon atoms in exactly 12 grams of pure 12 C. The number equals 6. 02 10 23 ð 1 mole of anything = 6. 02 10 23 units • 6. 02 10 23 “units” of anything: atoms, people, stars, $s, etc. = 1 mole
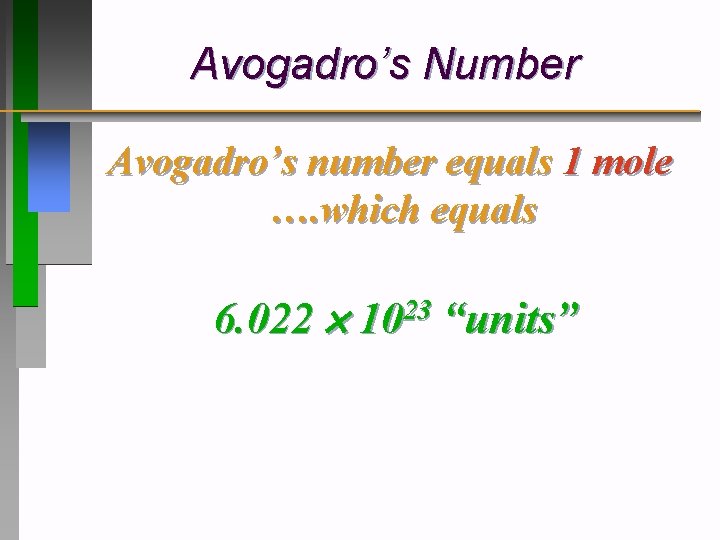
Avogadro’s Number Avogadro’s number equals 1 mole …. which equals 6. 022 23 10 “units”

Counting by Weighing 12 red marbles @ 7 g each = 84 g 12 yellow marbles @4 g each=48 g 55. 85 g Fe = 6. 022 x 1023 atoms Fe 32. 07 g S = 6. 022 x 1023 atoms S

Relative Masses of 1 Mole Ca. CO 3 100. 09 g Oxygen 32. 00 g Copper 63. 55 g Water 18. 02 g

Atomic and Molecular Weights Mass Measurements • 1 H weighs 1. 6735 x 10 -24 g and 16 O 2. 6560 x 10 -23 g. • DEFINITION: mass of 12 C = exactly 12 amu. • Using atomic mass units: • 1 amu = 1. 66054 x 10 -24 g • 1 g = 6. 02214 x 1023 amu

QUESTION

ANSWER – 22 E) 1. 06 ´ 10 g A mole of copper atoms has a mass of 63. 55 g. The mass of 1 copper atom is 23 – 22 63. 55 g/(6. 022 10 ) = 1. 06 10 g.

Atomic and Molecular Weights • Formula Weight a. k. a. Molecular Weight • Formula weights (FW): sum of Atomic Weights (AW) for atoms in formula. • FW (H 2 SO 4) = 2 AW(H) + AW(S) + 4 AW(O) • = 2(1. 0 amu) + (32. 0 amu) + 4(16. 0) • = 98. 0 amu

Atomic and Molecular Weights • Molecular weight (MW) is the weight of the molecular formula in amu. • MW of sugar (C 6 H 12 O 6 ) = ? • MW = 6(12. 0 amu) + 12(1. 0 amu) + 6(16. 0 amu) • = 180 amu

Molar Mass • A substance’s molar mass (equal to the formula weight: atomic or molecular weight in grams) is the mass in grams of one mole of the element or compound. ð C = 12. 01 grams per mole (g/mol) ð CO 2 ð 44. 01 = ? ? grams per mole (g/mol) 12. 01 + 2(16. 00) = 44. 01

QUESTION

ANSWER C) 46. 07 The molar mass is the sum of masses of all the atoms in the molecule. 2 ´ 12. 01 + 6 ´ 1. 008 + 1 ´ 16. 00 = 46. 07

QUESTION

ANSWER C) CH 3 Cl The molar mass has units of g/mol. (12. 8 g/0. 256 mol) = 50. 0 g/mol. The molecule with the closest molar mass is CH 3 Cl.

QUESTION

ANSWER B) 278 g The molar mass of sodium hydroxide, Na. OH, is 22. 99 g/mol + 16. 00 g/mol + 1. 008 g/mol = 40. 00 g/mol. Convert to grams: 6. 94 mol (40. 00 g/mol) = 278 g.

Percent Composition • Mass percent of an element: • For iron in (Fe 2 O 3), iron (III) oxide = ?

QUESTION

ANSWER E) All of these The ratio of C to H in C 8 H 8 is 1: 1. This is the same ratio found for each of the compounds, so all have the same percent composition by mass.

QUESTION Morphine, derived from opium plants, has the potential for use and abuse. It’s formula is C 17 H 19 NO 3. What percent, by mass, is the carbon in this compound? A. 42. 5% B. 27. 9% C. 71. 6% D. This cannot be solved until the mass of the sample is given.

ANSWER Morphine, derived from opium plants, has the potential for use and abuse. It’s formula is C 17 H 19 NO 3. What percent, by mass, is the carbon in this compound? A. 42. 5% B. 27. 9% C. 71. 6% D. This cannot be solved until the mass of the sample is given. (17 12) / ((17 12) + (19 1) + (1 14) + 3 16)) = 0. 716 100 = 71. 6 %

QUESTION

ANSWER C) 4. 04 g The molar mass of K 2 Cr. O 7 is 2 39. 10 + 52. 00 + 7 16. 00 = 242. 2. The mass fraction of potassium is (2 39. 10)/242. 2 = 0. 3229 12. 5 g = 4. 04 g.

Formulas: Dalton’s Law • Dalton’s law of multiple proportions: When two elements form different compounds, the mass ratio of the elements in one compound is related to the mass ratio in the other by a small whole number.

Formulas: Multiple Proportions

Formulas & Multiple Proportions Components of acid rain, SO 2(g) and SO 3(g) • Compound A contains: 1. 000 g Sulfur & 1. 500 g Oxygen • Compound B contains: 1. 000 g Sulfur & 1. 000 g Oxygen • Mass ratio A: 2 to 3; Mass ratio B: 1 to 1 • Adjusting for atomic mass differences: AW sulfur is 2 x the AW oxygen; the atom ratios therefore are S 1 O 3 and S 1 O 2 respectively

Formulas & Molecular Representations ð molecular formula = C 6 H 6 Benzene ð empirical formula = CH = C 6/6 H 6/6 ð molecular formula = (empirical formula)n [n = integer] (CH)6 • Other representations: Lewis Dot formulas, structural formulas, 2 -D, 3 -D

Formulas & Molecular Representations

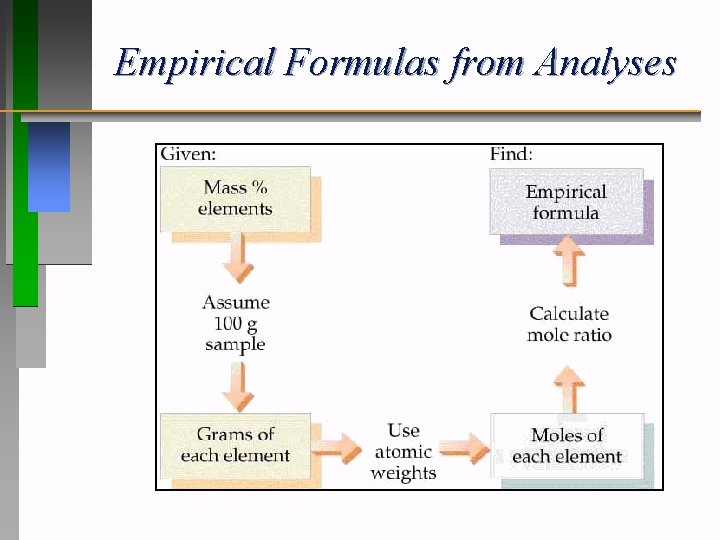
Empirical Formulas from Analyses

Empirical Formula Determination • 1. Use percent analysis. Let 100 % = 100 grams of compound. • 2. Determine the moles of each element. (Element % = grams of element. ) • 3. Divide each value of moles by the smallest of • the mole values. 4. Multiply each number by an integer to obtain all whole numbers.

QUESTION The dye indigo is a compound with tremendous economic importance (blue jeans wouldn’t be blue without it. ) Indigo’s percent composition is: 73. 27% C; 3. 84% H; 10. 68%N and 12. 21% O. What is the empirical formula of indigo? A. C 6 H 4 NO B. C 8 H 3 NO C. C 8 H 5 NO D. I know this should be whole numbers for each atom, but I do not know how to accomplish that.

ANSWER The dye indigo is a compound with tremendous economic importance (blue jeans wouldn’t be blue without it. ) Indigo’s percent composition is: 73. 27% C; 3. 84% H; 10. 68%N and 12. 21% O. What is the empirical formula of indigo? A. C 6 H 4 NO B. C 8 H 3 NO C. C 8 H 5 NO D. I know this should be whole numbers for each atom, but I do not know how to accomplish that.

Empirical & Molecular Formula Determination Quinine: C 74. 05%, H 7. 46%, N 8. 63%, O 9. 86% • 74. 05/12. 01, 7. 46/1. 008, 8. 63/14. 01, 9. 86/16. 00 C 6. 166 H 7. 40 N 0. 616 O 0. 616 • Empirical Formula: C 10 H 12 N 1 O 1 Empirical Formula Weight = ? • Molecular Weight = 324. 42 Molecular Formula = 2 x empirical formula • Molecular Formula = C 20 H 24 N 2 O 2

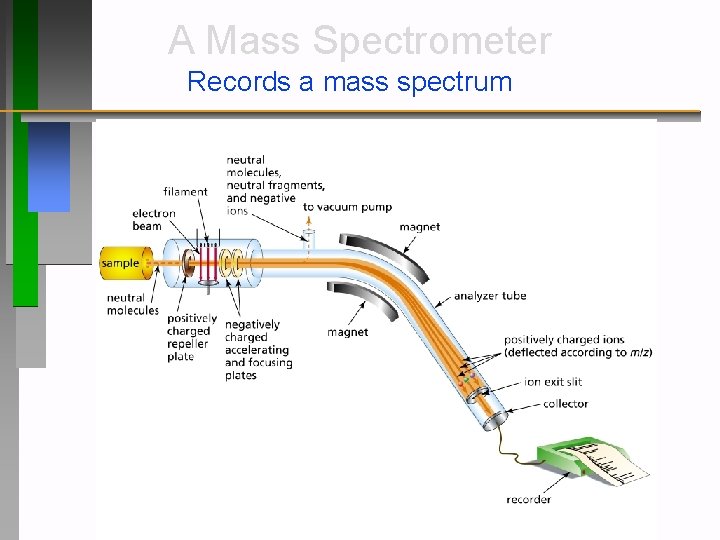
A Mass Spectrometer Records a mass spectrum

http: //www. colby. edu/chemistry/OChem/demoindex. html#table

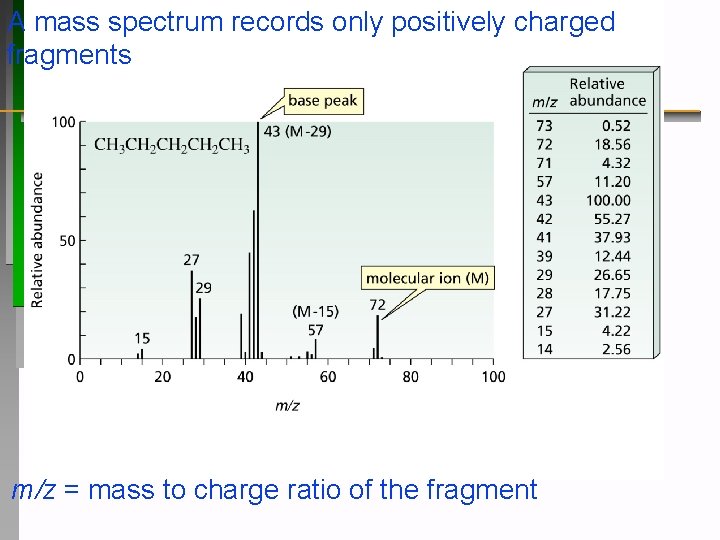
A mass spectrum records only positively charged fragments m/z = mass to charge ratio of the fragment

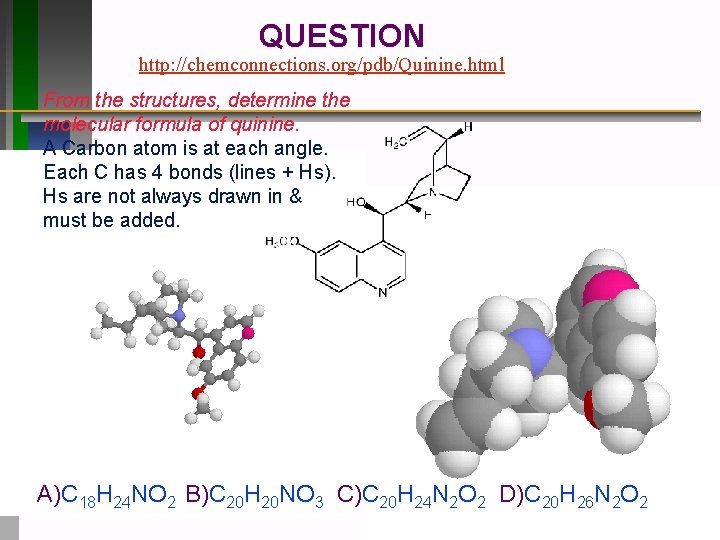
QUESTION http: //chemconnections. org/pdb/Quinine. html From the structures, determine the molecular formula of quinine. A Carbon atom is at each angle. Each C has 4 bonds (lines + Hs). Hs are not always drawn in & must be added. A)C 18 H 24 NO 2 B)C 20 H 20 NO 3 C)C 20 H 24 N 2 O 2 D)C 20 H 26 N 2 O 2

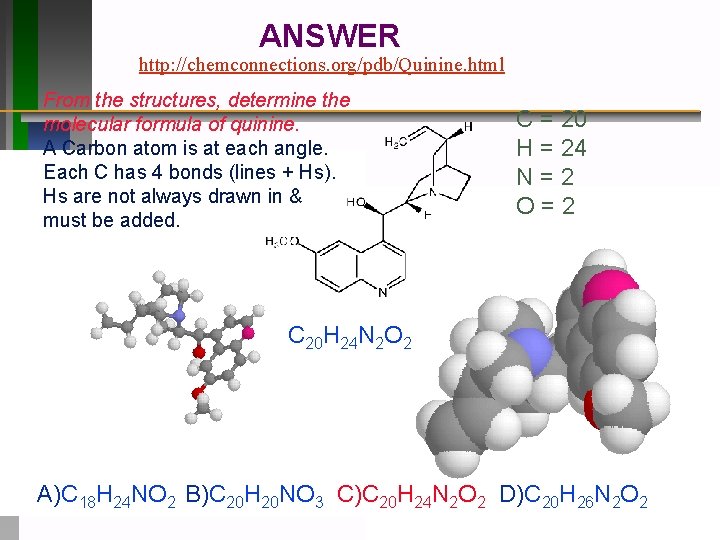
ANSWER http: //chemconnections. org/pdb/Quinine. html From the structures, determine the molecular formula of quinine. A Carbon atom is at each angle. Each C has 4 bonds (lines + Hs). Hs are not always drawn in & must be added. C = 20 H = 24 N=2 O=2 C 20 H 24 N 2 O 2 A)C 18 H 24 NO 2 B)C 20 H 20 NO 3 C)C 20 H 24 N 2 O 2 D)C 20 H 26 N 2 O 2

QUESTION

ANSWER B) C 8 H 8 The mass of CH is 13. 018 divides into 104. 1 about 8 times. Therefore there are 8 CH groups in this compound. C 8 H 8 is the molecular formula.

QUESTION Examine the condensed structural formulas shown below: I) Acetic acid (main ingredient in vinegar), CH 3 COOH II) Formaldehyde (used to preserve biological specimens), HCHO III) Ethanol (alcohol in beer and wine), CH 3 CH 2 OH For which molecule(s) are the empirical formula(s) and the molecular formula(s) the same? A) II B) I and II C) II and III D) I, II, and III

ANSWER Examine the condensed structural formulas shown below: I) Acetic acid (main ingredient in vinegar), CH 3 COOH II) Formaldehyde (used to preserve biological specimens), HCHO III) Ethanol (alcohol in beer and wine), CH 3 CH 2 OH For which molecule(s) are the empirical formula(s) and the molecular formula(s) the same? A) II B) I and II C) II and III D) I, II, and III