Aldehydes and ketons Formaldehyde Acetaldehyde Chloral hydrate Benzaldehyde
Aldehydes and ketons ü Formaldehyde. ü Acetaldehyde. ü Chloral hydrate. ü Benzaldehyde. ü Aceton. ü Acetophenone.

It contains variety of chemical compound including benzoate, ceosol, , benzaldehyde, y-terpineol, nerolidol, isohytol, phytol and acetic acid Chloral Hydrate is a sedative/hypnotic drug used to treat insomnia
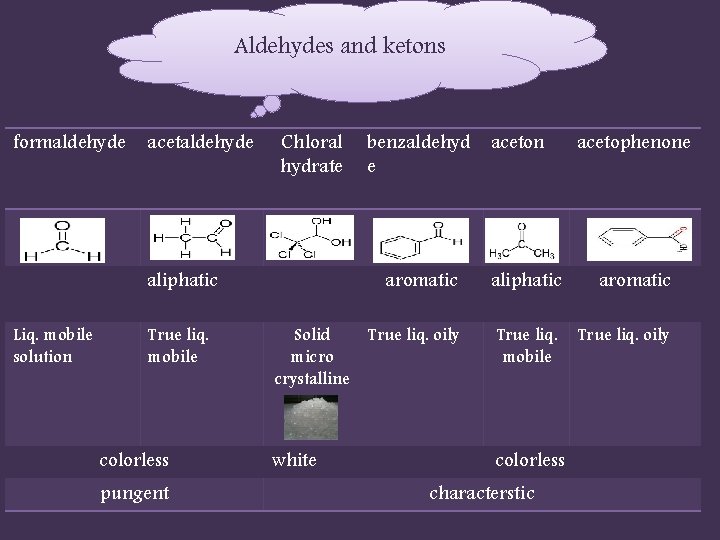
Aldehydes and ketons formaldehyde acetaldehyde Chloral hydrate aliphatic Liq. mobile solution True liq. mobile colorless pungent benzaldehyd aceton e acetophenone aromatic aliphatic aromatic Solid True liq. oily micro crystalline True liq. mobile True liq. oily white colorless characterstic
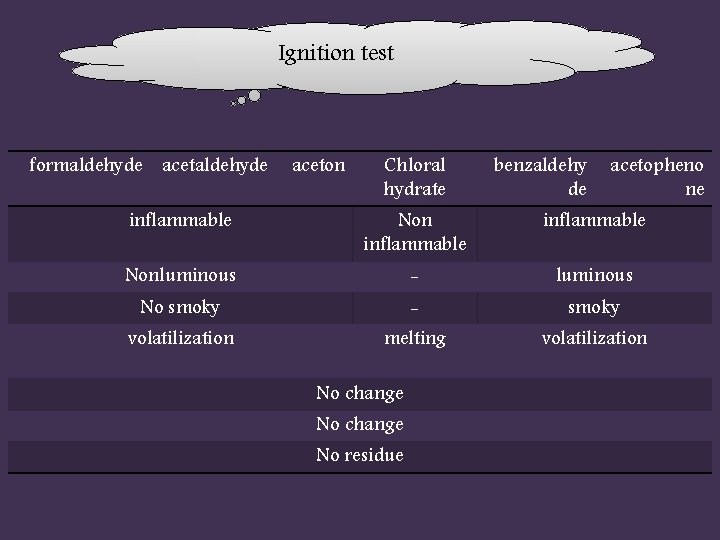
Ignition test formaldehyde aceton Chloral hydrate benzaldehy de acetopheno ne inflammable Nonluminous - luminous No smoky - smoky volatilization melting volatilization No change No residue
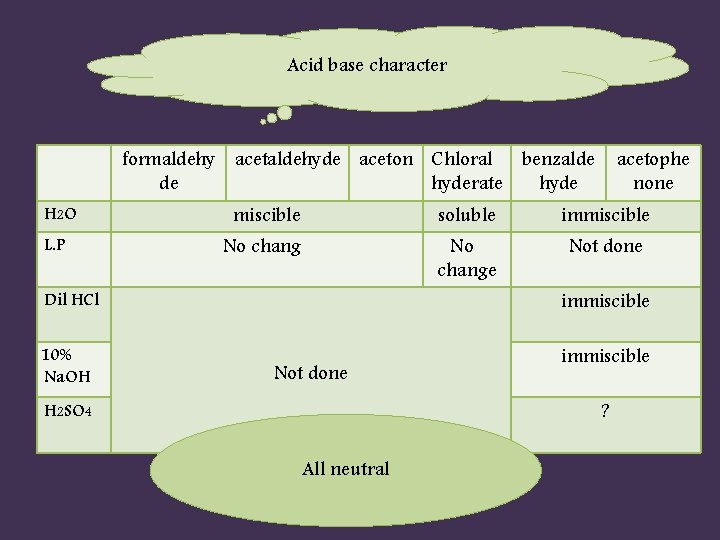
Acid base character formaldehy acetaldehyde aceton Chloral benzalde de hyderate hyde H 2 O L. P acetophe none miscible soluble immiscible No change Not done Dil HCl immiscible 10% Na. OH immiscible Not done ? H 2 SO 4 All neutral

Preliminary test Soda lime test: Only for solid Few chlorahydrate +few soda lime in mortor →sweety odor of chloroform odor Do not smell
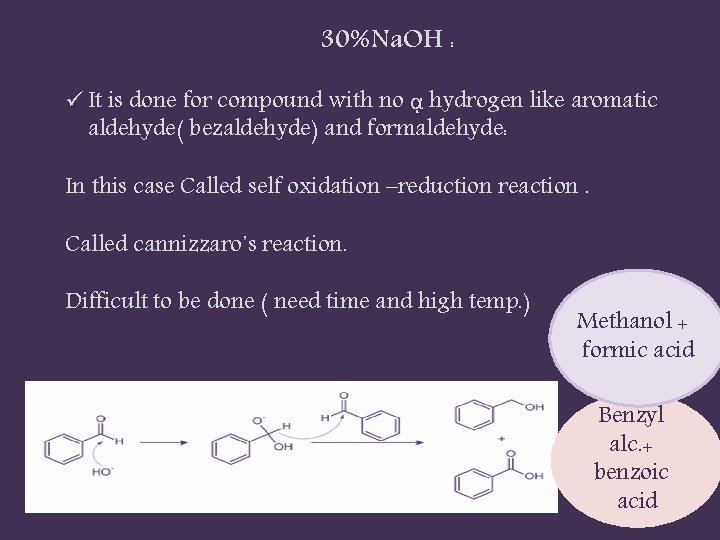
30%Na. OH : ü It is done for compound with no ᾳ hydrogen like aromatic aldehyde( bezaldehyde) and formaldehyde: In this case Called self oxidation –reduction reaction. Called cannizzaro᾿s reaction. Difficult to be done ( need time and high temp. ) Methanol + formic acid Benzyl alc. + benzoic acid
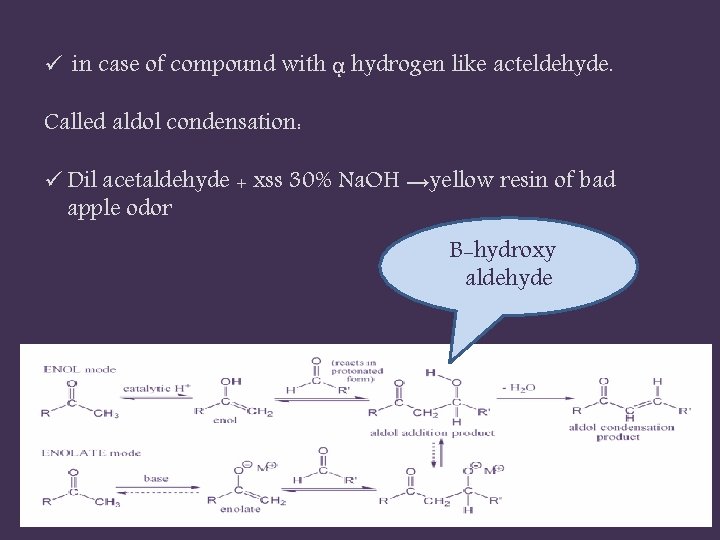
ü in case of compound with ᾳ hydrogen like acteldehyde. Called aldol condensation: ü Dil acetaldehyde + xss 30% Na. OH →yellow resin of bad apple odor B-hydroxy aldehyde

Chlorahydrate: chlor. +30%Na. OH on cold but some times need to warm → sweety odor of chloroform +oily droplet. Aceton and acetophenone: no reaction. Fecl 3: All no reaction. Conc. H 2 SO 4: ü no reaction with all except with chlorahydrate. ü Few chlorahydrate + Conc. H 2 SO 4 on cold →sepration of oily layer of chloral

General test ü 2, 4 dinitrophenyl hydrazine test: ü silver mirror test (tollen᾿s reagent test or ammonical silver nitrate test: ü fehling reagent test or fehling reduction test
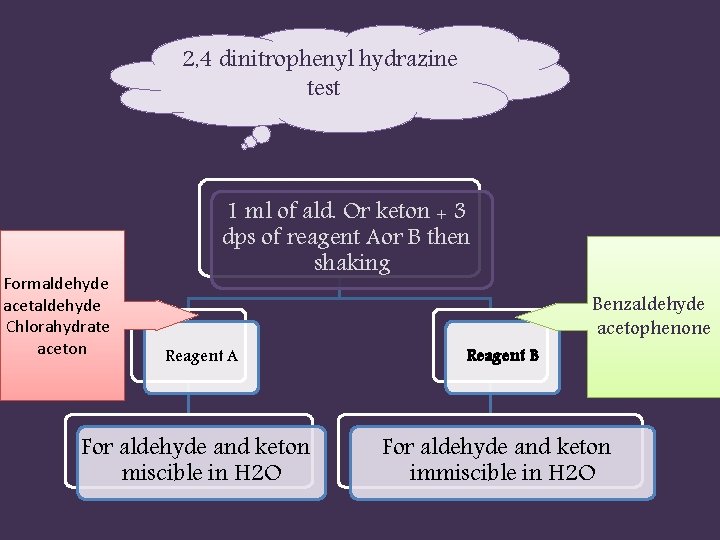
2, 4 dinitrophenyl hydrazine test Formaldehyde acetaldehyde Chlorahydrate aceton 1 ml of ald. Or keton + 3 dps of reagent Aor B then shaking Reagent A For aldehyde and keton miscible in H 2 O Reagent B Benzaldehyde acetophenone For aldehyde and keton immiscible in H 2 O

formaldehyde: yellow ppt. Acetaldehyde: yellow orange ppt. Chlorahydrate: no reaction ( yellow color) Aceton: orange yellow ppt. Benzaldehyde: orange ppt. Acetophenone: orange ppt.
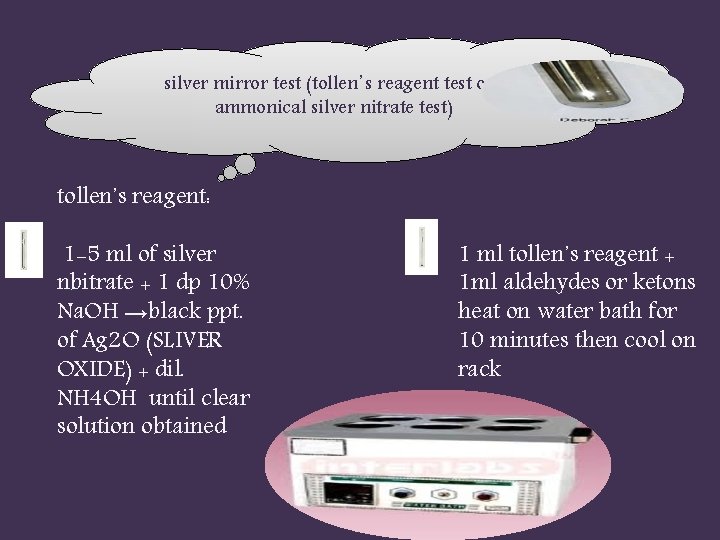
silver mirror test (tollen᾿s reagent test or ammonical silver nitrate test) tollen᾿s reagent: 1 -5 ml of silver nbitrate + 1 dp 10% Na. OH →black ppt. of Ag 2 O (SLIVER OXIDE) + dil. NH 4 OH until clear solution obtained 1 ml tollen᾿s reagent + 1 ml aldehydes or ketons heat on water bath for 10 minutes then cool on rack
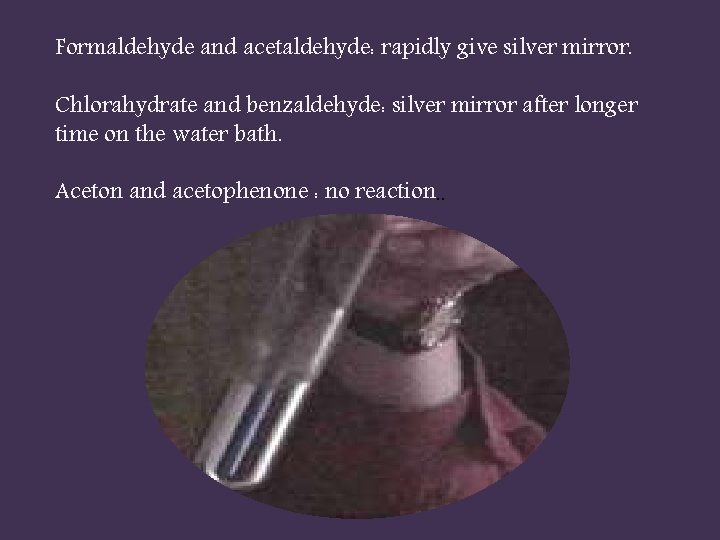
Formaldehyde and acetaldehyde: rapidly give silver mirror. Chlorahydrate and benzaldehyde: silver mirror after longer time on the water bath. Aceton and acetophenone : no reaction. .

fehling reagent test or fehling reduction test Mix equal amount of fehling A + B to give blue color of cupric ion Few aldehydes or ketons + few Na 2 CO 3 + fehling mix and put it in water bath Formaldehyde actaldehyde : rapidly change in color. Chlorahydrat and benzaldehyde: take longer time to change the color. yellow or red ppt. of cuprous oxide Aceton and acetophenone: no reaction
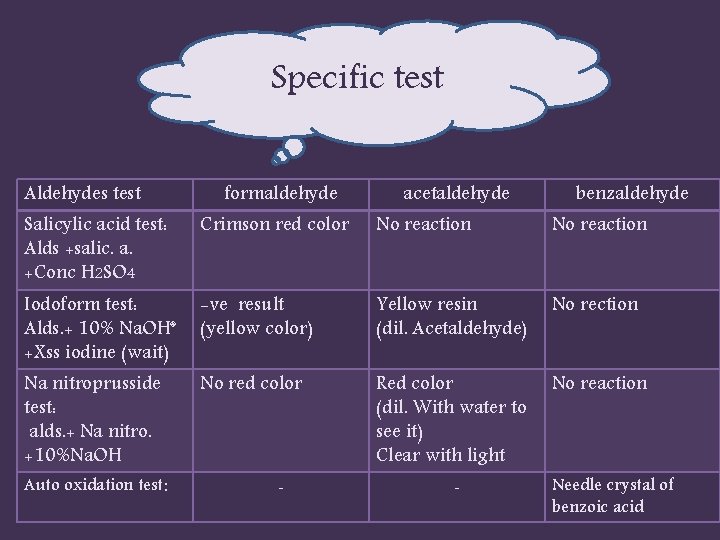
Specific test Aldehydes test formaldehyde acetaldehyde benzaldehyde Salicylic acid test: Alds +salic. a. +Conc H 2 SO 4 Crimson red color No reaction Iodoform test: Alds. + 10% Na. OH* +Xss iodine (wait) -ve result (yellow color) Yellow resin (dil. Acetaldehyde) No rection Na nitroprusside test: alds. + Na nitro. +10%Na. OH No red color Red color (dil. With water to see it) Clear with light No reaction Auto oxidation test: - - Needle crystal of benzoic acid

Ketones test Iodoform test: Na nitroprusside test: Meta dinitro benzen test: Kets. + meta dinitro benzene+ Xss 10% Na. OH (Shake) aceton acetophenone Yellow ppt. Red color Deep violet color slowly fade Violet color in organic layer (dissolve acetophenone in ethanol) chlorahydrate ü Soda lime test: sweety odor of chloroform. ü Conc. H 2 SO 4: sepration of chloral layer. ü 30% Na. OH: oily droplet and chloroform odor

IR location ü C-H stretching band of aliphatic under 3000 cm -1. ü C-H stretching band of aromatic above 3000 cm-1. ü Carbonyl group stretching band of aldehyde at 1725 cm-1. ü Carbonyl group stretching band of ketone at 1715 cm-1
- Slides: 18