TimeResolved Raman Studies of the Electron Adducts of









- Slides: 26

Time-Resolved Raman Studies of the Electron Adducts of Benzoate Anion in Water Deanna O’Donnell University of Notre Dame Radiation Laboratory Department of Chemistry and Biochemistry June 16 th, 2008 1

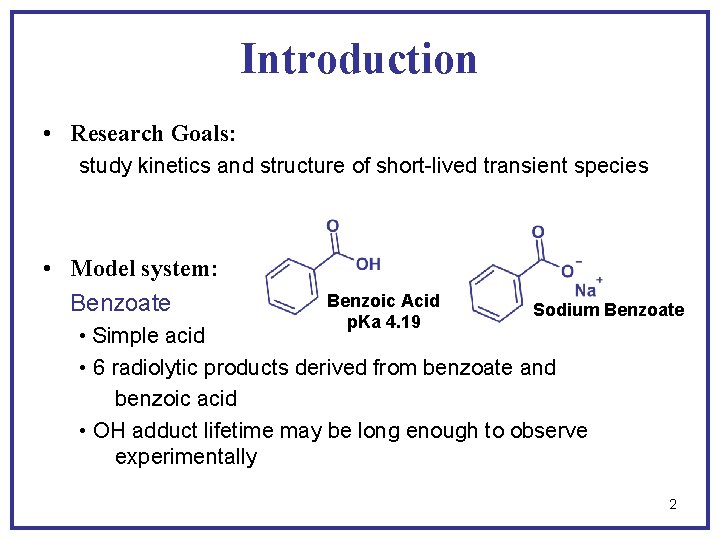
Introduction • Research Goals: study kinetics and structure of short-lived transient species • Model system: Benzoate • Simple acid Benzoic Acid p. Ka 4. 19 Sodium Benzoate • 6 radiolytic products derived from benzoate and benzoic acid • OH adduct lifetime may be long enough to observe experimentally 2

Problems with Benzoate • Preservative in Food Industry for over 80 years (E 211) • 1993 - Lawrence 1 provided mechanism for benzoate conversion to benzene • May 2006 - FDA released report 2 listing 100 soft drinks containing benzene – 10 exceeded the EPA drinking water legal limit • May 2008 - Coca Cola Company released a statement that they will phase out sodium benzoate in their soft drink in the UK 3 1 Lawrence, G. J. Agric. Food Chem. , 41, 1993, 693. FDA, 2006. "Data on Benzene in Soft Drinks and Other Beverages, " United States Food and Drug Administration. Accessed June 2 nd at: http: //www. cfsan. fda. gov/~dms/benzdata. html 3 3 http: //www. Beverage. Daily. com 2

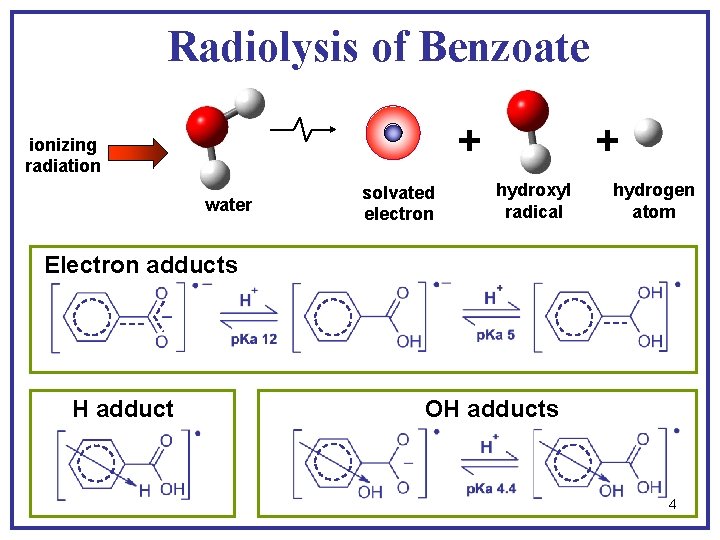
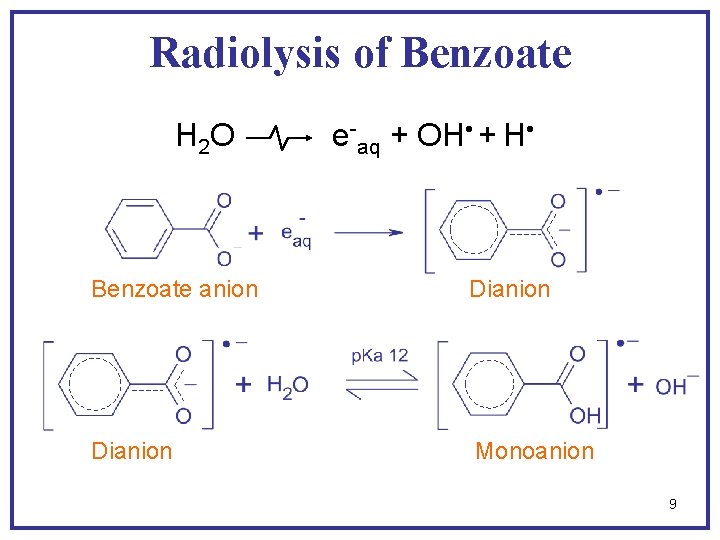
Radiolysis of Benzoate + ionizing radiation water solvated electron + hydroxyl radical hydrogen atom Electron adducts H adduct OH adducts 4

LINAC – Transient Absorption Benzoate Dianion e 445 = 8000 M-1 cm-1 Benzoate Monoanion e 435 = 5200 M-1 cm-1 Computer Controlled RF Linac System 5

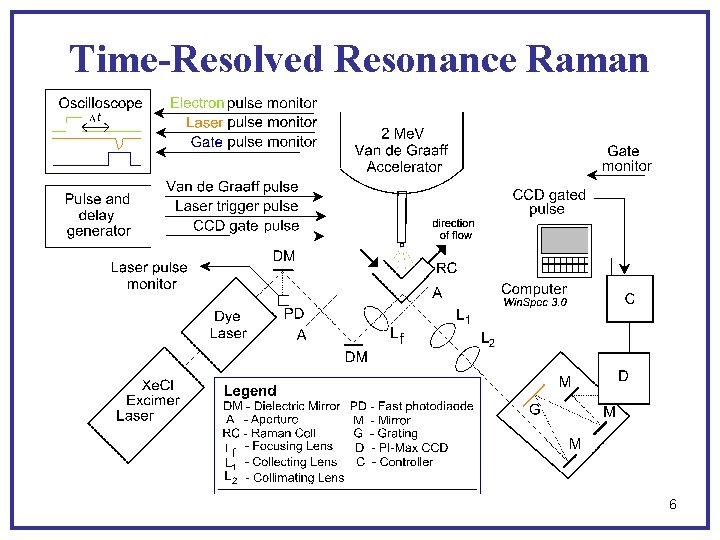
Time-Resolved Resonance Raman 6

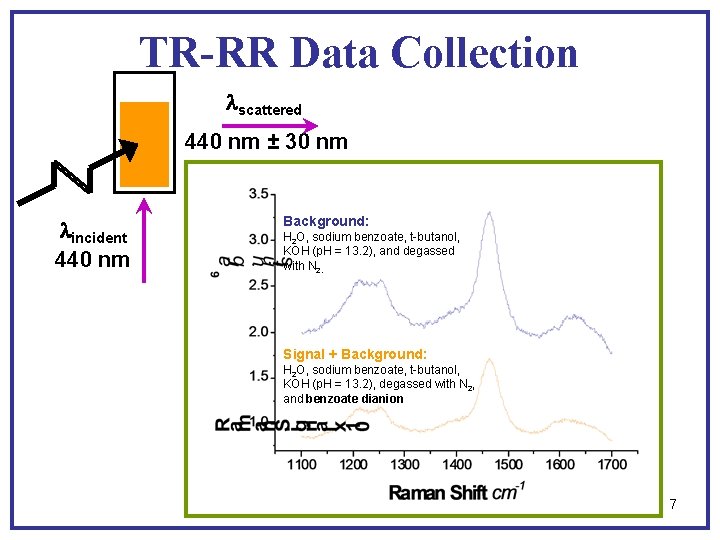
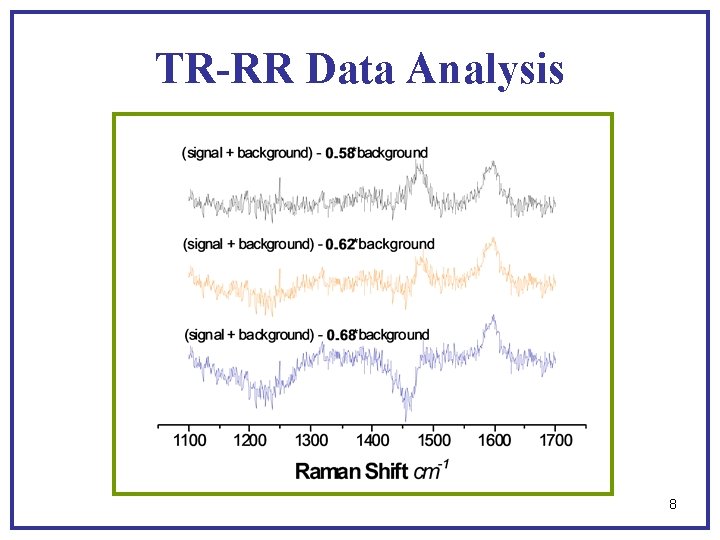
TR-RR Data Collection lscattered 440 nm ± 30 nm lincident 440 nm Background: H 2 O, sodium benzoate, t-butanol, KOH (p. H = 13. 2), and degassed with N 2. Signal + Background: H 2 O, sodium benzoate, t-butanol, KOH (p. H = 13. 2), degassed with N 2, and benzoate dianion 7

TR-RR Data Analysis 8

Radiolysis of Benzoate H 2 O e-aq + OH + H Benzoate anion Dianion Monoanion 9

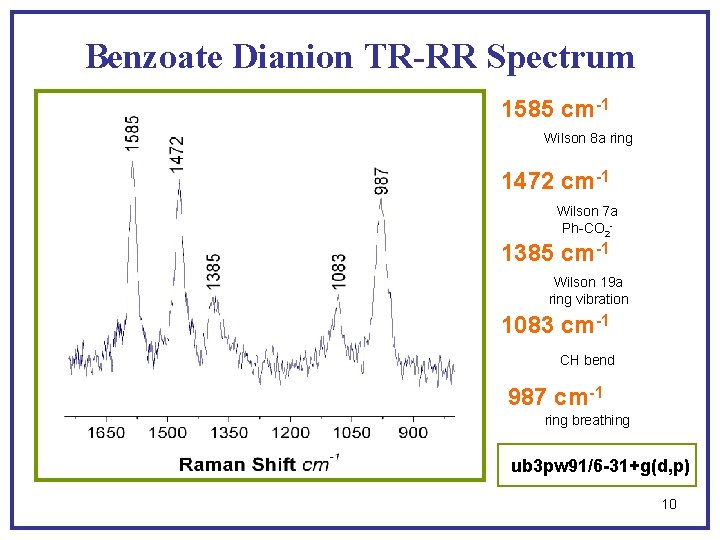
Benzoate Dianion TR-RR Spectrum 1585 cm-1 Wilson 8 a ring 1472 cm-1 Wilson 7 a Ph-CO 2 - 1385 cm-1 Wilson 19 a ring vibration 1083 cm-1 CH bend 987 cm-1 ring breathing ub 3 pw 91/6 -31+g(d, p) 10

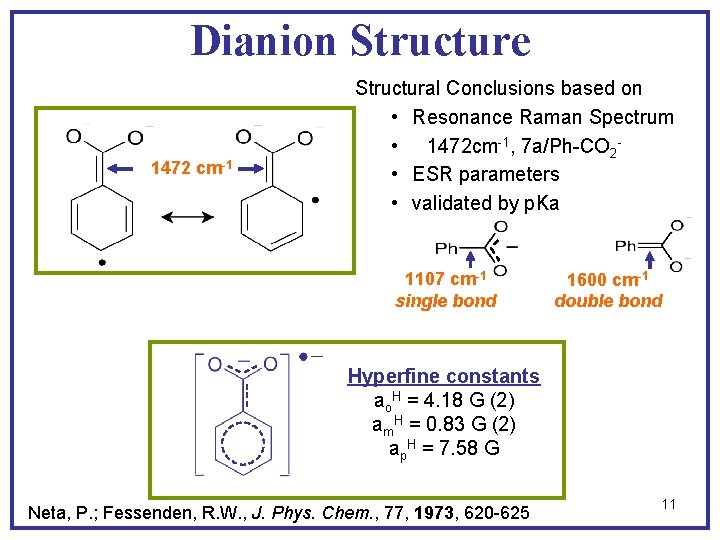
Dianion Structure 1472 cm-1 Structural Conclusions based on • Resonance Raman Spectrum • 1472 cm-1, 7 a/Ph-CO 2 • ESR parameters • validated by p. Ka 1107 cm-1 single bond 1600 cm-1 double bond Hyperfine constants ao. H = 4. 18 G (2) am. H = 0. 83 G (2) ap. H = 7. 58 G Neta, P. ; Fessenden, R. W. , J. Phys. Chem. , 77, 1973, 620 -625 11

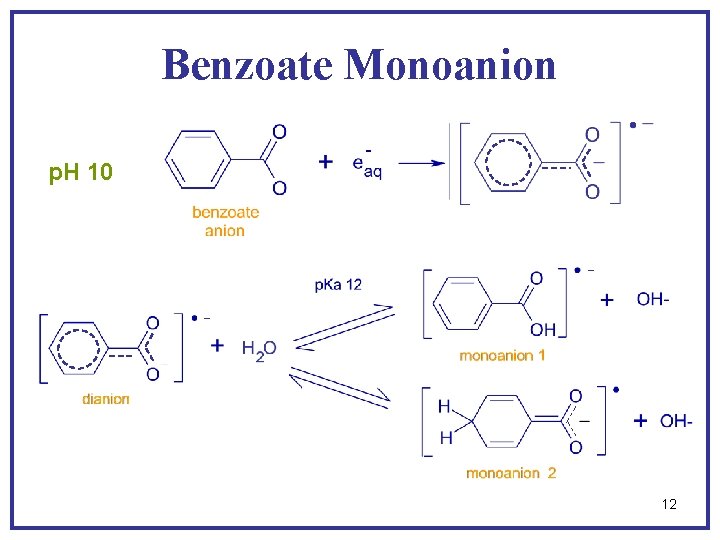
Benzoate Monoanion p. H 10 12

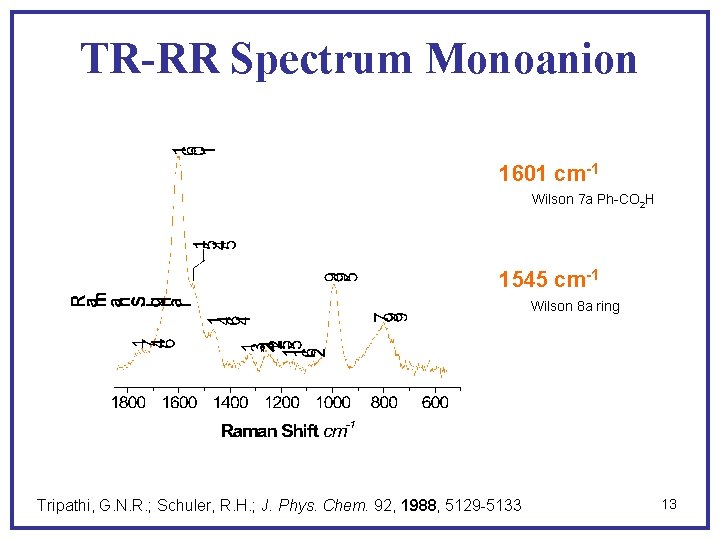
TR-RR Spectrum Monoanion 1601 cm-1 Wilson 7 a Ph-CO 2 H 1545 cm-1 Wilson 8 a ring Tripathi, G. N. R. ; Schuler, R. H. ; J. Phys. Chem. 92, 1988, 5129 -5133 13

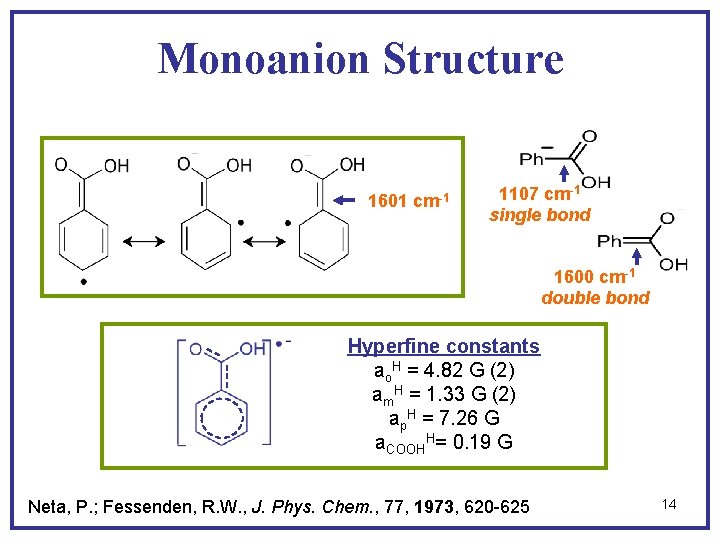
Monoanion Structure 1601 cm-1 1107 cm-1 single bond 1600 cm-1 double bond Hyperfine constants ao. H = 4. 82 G (2) am. H = 1. 33 G (2) ap. H = 7. 26 G a. COOHH= 0. 19 G Neta, P. ; Fessenden, R. W. , J. Phys. Chem. , 77, 1973, 620 -625 14

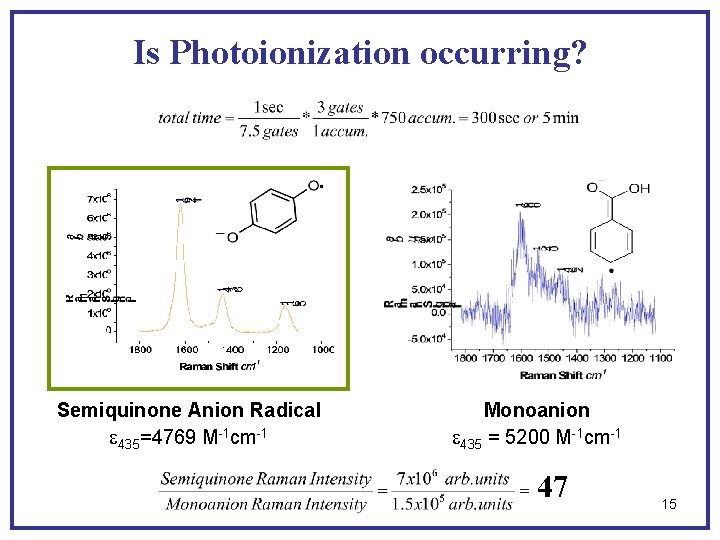
Is Photoionization occurring? Semiquinone Anion Radical e 435=4769 M-1 cm-1 Monoanion e 435 = 5200 M-1 cm-1 47 15

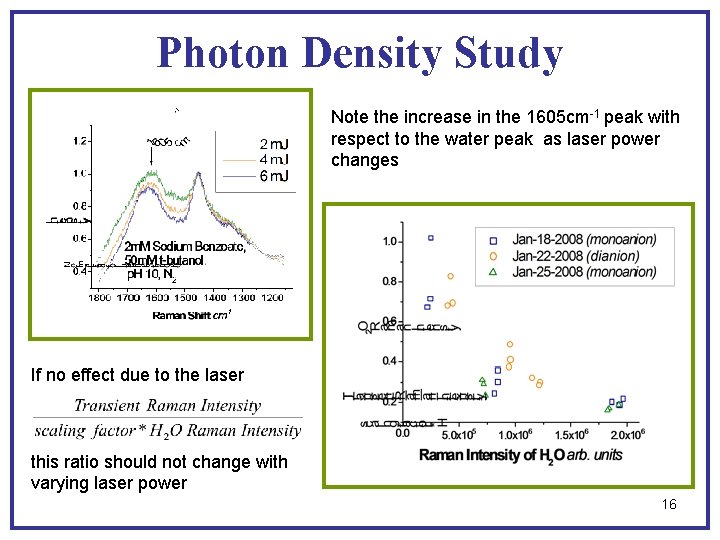
Photon Density Study Note the increase in the 1605 cm-1 peak with respect to the water peak as laser power changes If no effect due to the laser this ratio should not change with varying laser power 16

Summary • Benzoate electron adducts – Benzoate Dianion Radical – Benzoate Monoanion Radical • Photoionization • Future Work includes: – OH adduct – product analysis – para-substituted benzoate 17

Acknowledgments • Dr. G. N. R. Tripathi • Dr. Ian Carmichael • Dr. Irek Janik • This work was sponsored by the Department of Energy 18

19

Extra Slides 20

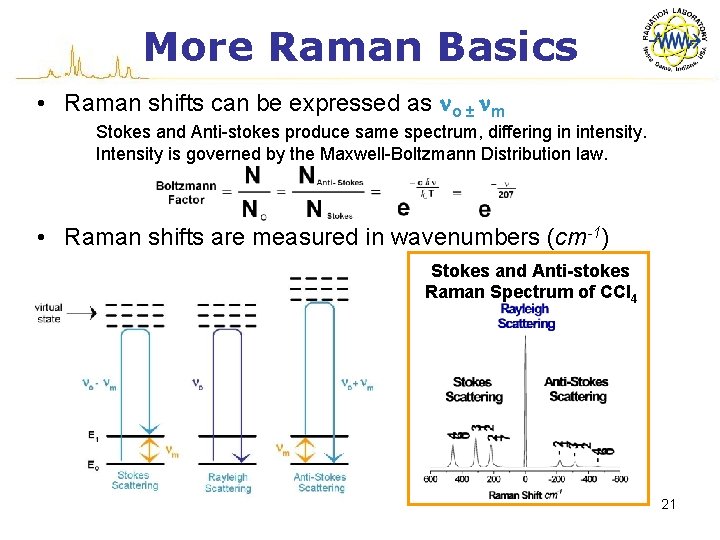
More Raman Basics • Raman shifts can be expressed as no ± nm Stokes and Anti-stokes produce same spectrum, differing in intensity. Intensity is governed by the Maxwell-Boltzmann Distribution law. • Raman shifts are measured in wavenumbers (cm-1) Stokes and Anti-stokes Raman Spectrum of CCl 4 21

Signal Enhancement • Common method to enhance the Raman scattering is • Resonance Raman • Occurs when no nem • Enhancement is on the order of 103 to 108 mi = aij Ej mi = induced electric dipole aij = polarizability E = electric field of the iiiiielectromagnetic radiation Imn = Io(no-nmn)4 S|(aij)mn|2 (aij)mn (nem-no)-1 22

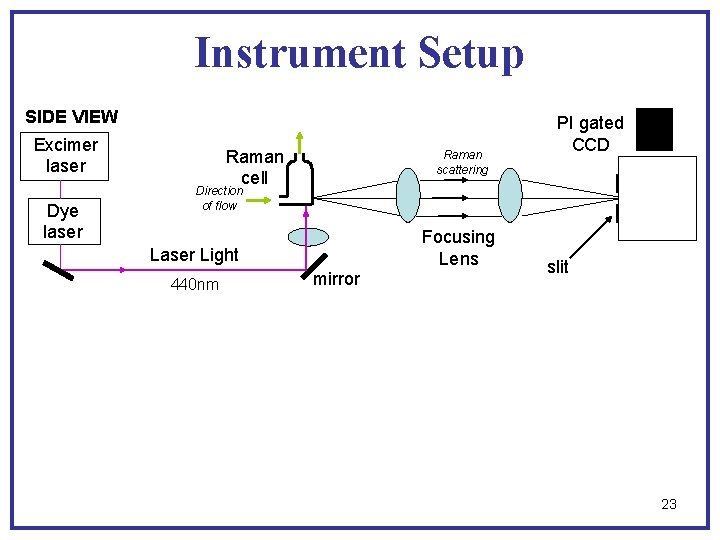
Instrument Setup SIDE VIEW Excimer laser Dye laser Raman cell Raman scattering PI gated CCD Direction of flow Focusing Lens Laser Light 440 nm mirror slit 23

Electron Spin Resonance • Same principles as NMR • Measures resonance of electron rather than nuclei • Species must be paramagnetic E ms = + ½ E = E +½ - E -½ = gem. BBo E = hn = gm. BBo ms = - ½ Bo=0 Bo≠ 0 Magnetic Field Hyperfine Constant • analogous to J-coupling in NMR • when me interact with m. N, allow additional energy states • spacing between lines is hyperfine constant 24

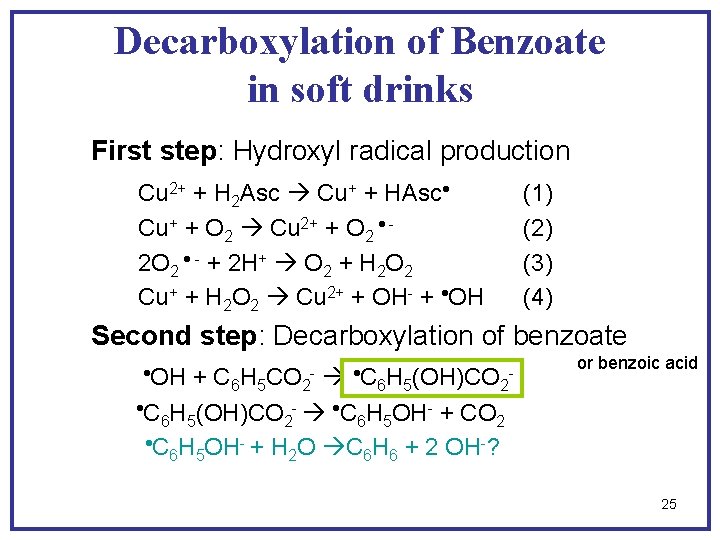
Decarboxylation of Benzoate in soft drinks First step: Hydroxyl radical production Cu 2+ + H 2 Asc Cu+ + HAsc Cu+ + O 2 Cu 2+ + O 2 2 O 2 - + 2 H+ O 2 + H 2 O 2 Cu+ + H 2 O 2 Cu 2+ + OH- + OH (1) (2) (3) (4) Second step: Decarboxylation of benzoate OH C + C 6 H 5 CO 2 6 H 5(OH)CO 2 C H (OH)CO - C H OH- + CO 6 5 2 C H OH- + H O C H + 2 OH-? 6 5 2 6 6 - - or benzoic acid 25

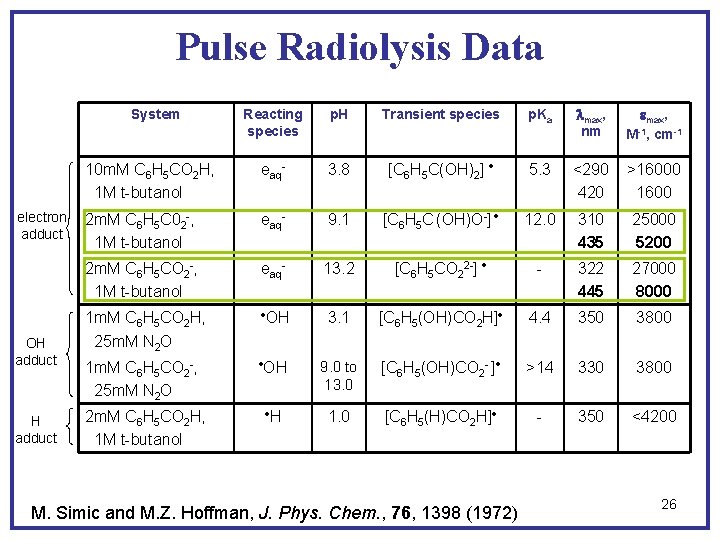
Pulse Radiolysis Data Reacting species p. H Transient species p. Ka lmax, nm emax, M-1, cm-1 10 m. M C 6 H 5 CO 2 H, 1 M t-butanol eaq- 3. 8 [C 6 H 5 C(OH)2] 5. 3 <290 420 >16000 1600 2 m. M C 6 H 5 C 02 -, 1 M t-butanol eaq- 9. 1 [C 6 H 5 C (OH)O-] 12. 0 310 435 25000 5200 2 m. M C 6 H 5 CO 2 -, 1 M t-butanol eaq- 13. 2 [C 6 H 5 CO 22 -] - 322 445 27000 8000 1 m. M C 6 H 5 CO 2 H, 25 m. M N 2 O OH 3. 1 [C 6 H 5(OH)CO 2 H] 4. 4 350 3800 1 m. M C 6 H 5 CO 2 -, 25 m. M N 2 O OH 9. 0 to 13. 0 [C 6 H 5(OH)CO 2 - ] >14 330 3800 H 1. 0 [C 6 H 5(H)CO 2 H] - 350 <4200 System electron adduct OH adduct 2 m. M C 6 H 5 CO 2 H, 1 M t-butanol M. Simic and M. Z. Hoffman, J. Phys. Chem. , 76, 1398 (1972) 26