Raman Spectra SEM IV Dr DEBDAS MANDAL Raman

Raman Spectra SEM – IV Dr. DEBDAS MANDAL

Raman spectra deals with the scattering of light and not with its absorption. Homo nuclear diatomic molecule such as H 2, N 2, O 2 etc. which do not show IR spectra since they do not possess a permanent dipole moment do show Raman spectra since their vibration is accompanied by a change in the polarsibility of the molecule.

A monochromatic beam of light illuminates the sample and the scattered light is observed. If the incident light and scattered light are found to have the same frequency (say υ0) i. e no change in frequency , it gives the Rayleigh Scattering. If the frequency of the scattered light is less than that of the incident radiation, it gives the Stokes line (Raman Scattering). But if the frequency of the scattered light is higher than that of the incident radiation, it gives the Anti -stokes line (Raman Scattering).

Quantum theory of Raman effect The occurrence of Raman scattering may be most easily understood in terms of the quantum theory of radiation. This treats radiation of frequency υ as consisting of a stream of particle (called photons) having energy hυ where h is plancks constant. Photons can be imagined to undergo collision with molecule, and if the collision is perfectly elastic they will be unchanged. However, it may happen that energy is exchanged between photon and molecule during the collision. Such collisions are called inelastic.

The molecule can gain or lose amount of energy only in accordance with the quantal laws. If the molecule gains energy ΔE, the photons will be scattered with energy hυ – ΔE and the equivalent radiation will have a frequency υ –ΔE/h. Conversely if the molecule loses energy ΔE , the scattered frequency will be υ + ΔE/h. Radiation Scattered with a frequency lower than that of the incident beam is referred to as Stokes radiation, while that at higher frequency is called Anti-stokes radiation. Since the former is accompanied by an increase in molecular energy while latter involves a decrease.
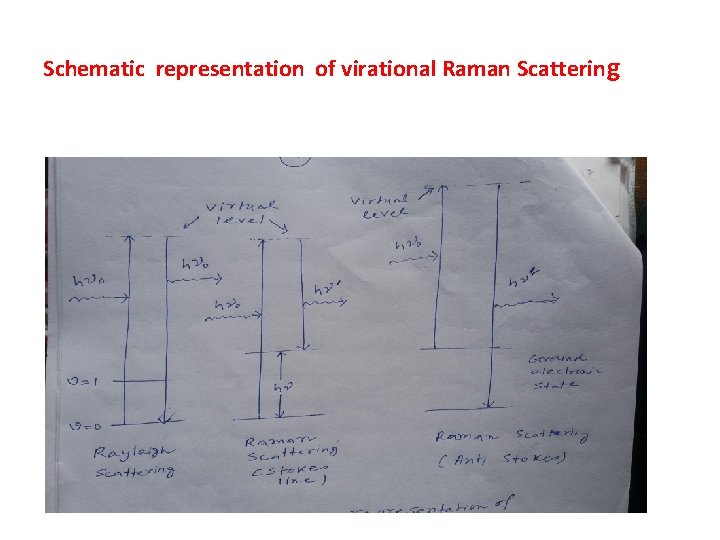
Schematic representation of virational Raman Scattering

When the incident light (υ0 frequency) includes a vibrational transition ( say v = 0 to v =1, the energy requirement for this vibrational transition = hυ) and frequency of the scattered light becomes (υ0 – υ) i. e. Stokes line. If the vibrational excited molecule (v =1) interacts with the incident radiation and transfers the excitation energy (hυ) to come back to the ground state ( v = 0) , then the incident radiation gains the additional energy hυ and if this photon scattered it will possess the frequency υ0 + υ (i. e. Anti-stokes line)

There are many other ways for returning from the excited state (v =1) to the ground state ( i. e. v =0) and consequently, intensity of the Anti-stokes line is much lower than that of the Stokes line. Rayleigh Scattering is the result of elastic collision while Raman scattering is the result of inelastic collisions

When a molecule is put into a static electric field it suffers some distortion, the positively charged nuclei being attracted towards the negative pole of the field , the electrons to the positive pole. This separation of charge centers causes an induced electric dipole moment to be set up in the molecule and the molecule is said to be polarized. The induced dipole moment μ depends both on the magnitude of the applied field, E, and on the ease with which the molecule can be distorted. We may write μ = αE where α is the polarisability of the molecule. Raman spectroscopy deals with the scattering of light and not with its absorption. Homo nuclear diatomic molecules such as H 2, N 2, O 2 etc. which do not show IR spectra since they do not possess a permanent dipole moment, but show Raman spectra since their vibration is accompanied by a change in polarisibility of the molecules. As a consequence of the change in polarisibility there occurs a change in the induced moment at the vibrational frequency.

Raman spectroscopy despite difficulties with sensitivities, has several advantages. It gives information about molecular vibration that are inactive in the IR region because of molecular symmetry. According to the mutual exclusion rules for centro symmetric molecules (such as H 2, N 2, O 2 etc. ) which are inactive in IR (since they are not accompanied by a change in dipole moment which is already zero) are observed in Raman spectra. Also the symmetric stretching vibration of CO 2 that is inactive in IR, is active in Raman Spectra.

Difference between Raman Spectra and IR spectra Raman Spectra IR spectra (a) It is due to scattering of (a) It is the result of light by the vibrating absorption of light by molecules vibrating molecule (b) Polarisibility of the molecule will decide whether the Raman spectra will be observed or not. (b) The presence of permanent dipole moment in a molecule may be regarded as a criterion of infra red spectra.

The rule of mutual exclusion in molecular spectroscopy relates the observation of molecular vibration to molecular symmetry. It states that no normal modes can be both Infrared and Raman active in a molecule that possesses a centre of symmetry.

Application Structure of CO 2 : If it is assumed to be a symmetrical linear molecule O-C-O , one should expect two fundamental lines (υ2 and υ3) in IR and one (υ1) in the Raman. This has been experimentally confirmed and the following assignments are made υ1 = 1340 cm-1, υ2 = 667 cm-1 and υ3 = 2349 cm-1. Structure of N 2 O : If it is assumed that N 2 O is like CO 2 i. e. NO-N, one should expect a similar type of behaviour as in CO 2. If it is unsymmetrical, i. e. N-N-O , υ1 should become active in IR as well as in Raman. The additional fundamental has been observed in both IR and Raman spectra. This proves that the structure of N 2 O is N-N-O.

Structure of mercurous salts : When a Raman spectrum of an aqueous solution of mercurous nitrate is recorded. It shows a line which is absent in the other metal nitrates. This line may be attributed to the vibration of Hg-Hg covalent bond in the diatomic molecule Hg 22+.

Many problem of physical chemistry concerning electrolytic dissociation, hydrolysis and transition from crystalline to amorphous state have been studied and following results have been obtained. The amorphous state of a substance gives rise to broad and diffused band while crystalline state gives fine sharp line. In the case of the phenomenon of electrolytic dissociation , the intensity of Raman lines enable us to determine the number and nature of ions produced. Therefore we can decide whether the dissociation is complete or partial. Raman spectra provides the information about the following facts. The presence and absence of specific linkage in a molecule The structure of simple compound Study of isomers Classification of the compound
- Slides: 15