STRUCTURAL CHARACTERIZATION OF HYDROXYL RADICAL ADDUCTS IN AQUEOUS

![Hemibonding of OH and halide ion in aqueous solution Lambda max [nm] Epsilon/103 [M-1 Hemibonding of OH and halide ion in aqueous solution Lambda max [nm] Epsilon/103 [M-1](https://slidetodoc.com/presentation_image_h2/cfd01aceee2688a8e51f7e33c1be5d7d/image-11.jpg)



- Slides: 17

STRUCTURAL CHARACTERIZATION OF HYDROXYL RADICAL ADDUCTS IN AQUEOUS MEDIA Irek Janik, GNR Tripathi Notre Dame Radiation Laboratory

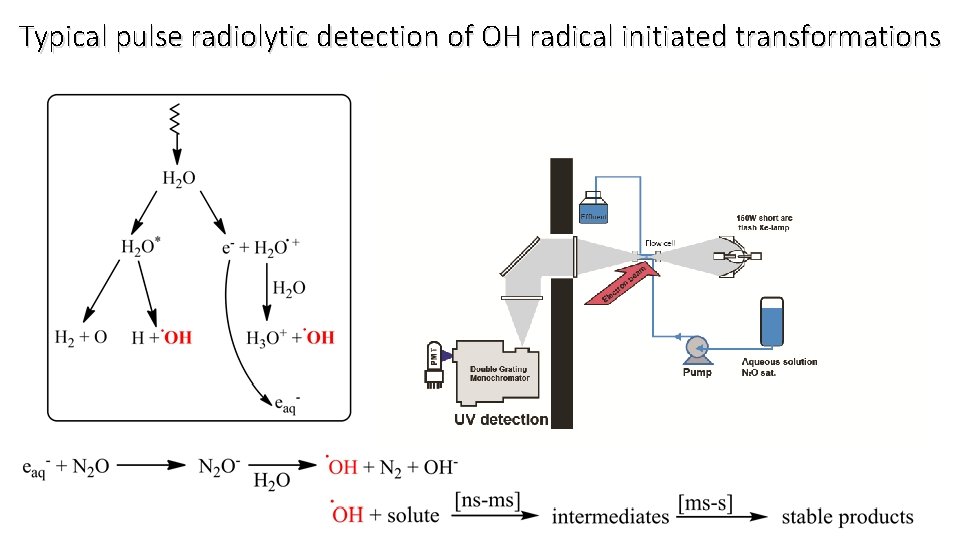
Typical pulse radiolytic detection of OH radical initiated transformations

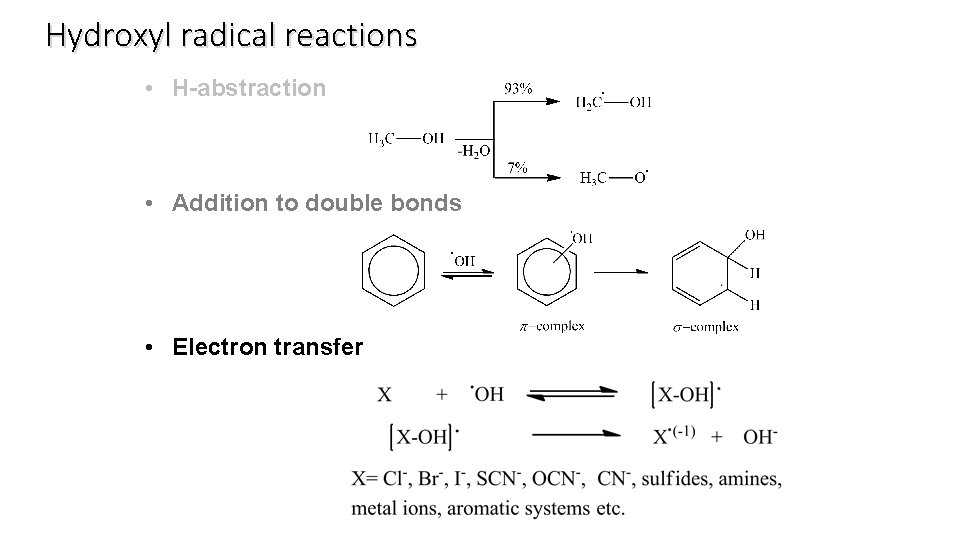
Hydroxyl radical reactions • H-abstraction • Addition to double bonds • Electron transfer

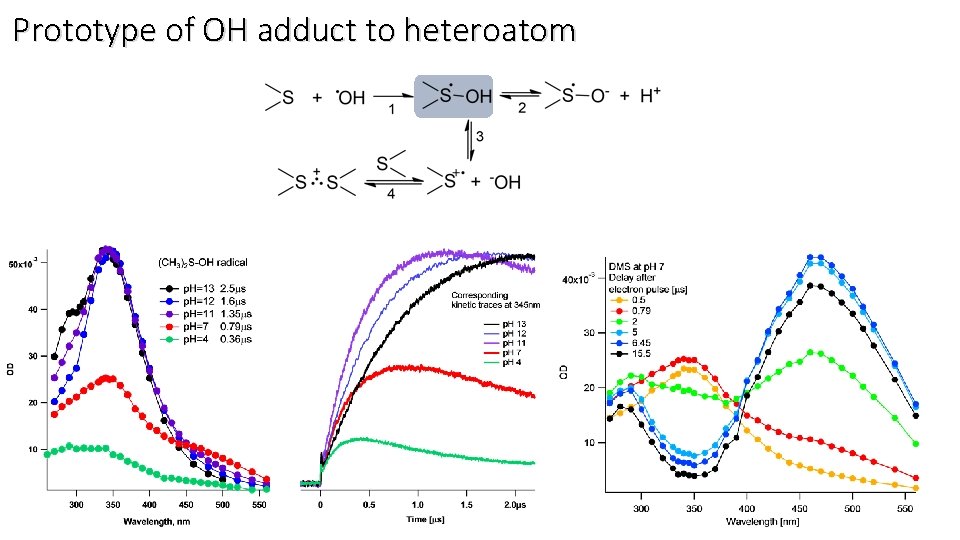
Prototype of OH adduct to heteroatom

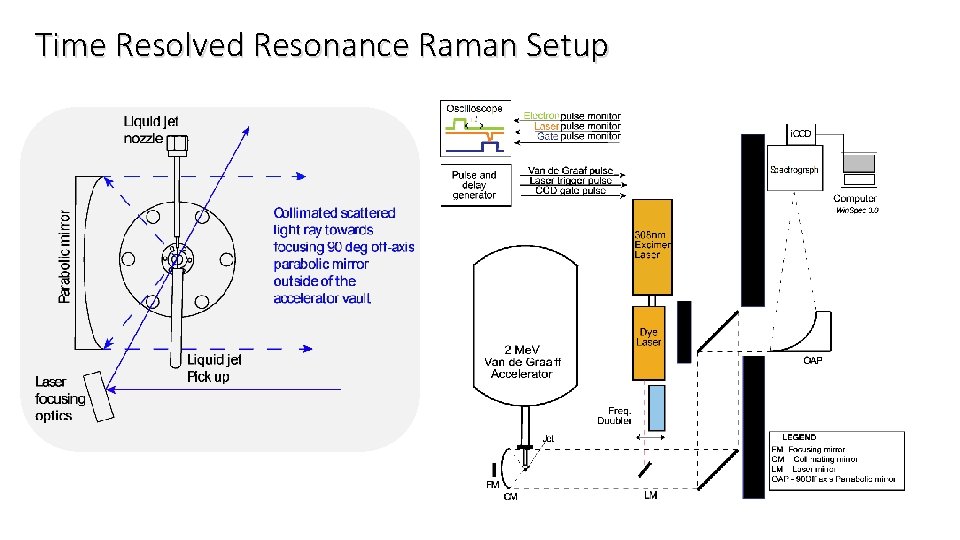
Time Resolved Resonance Raman Setup

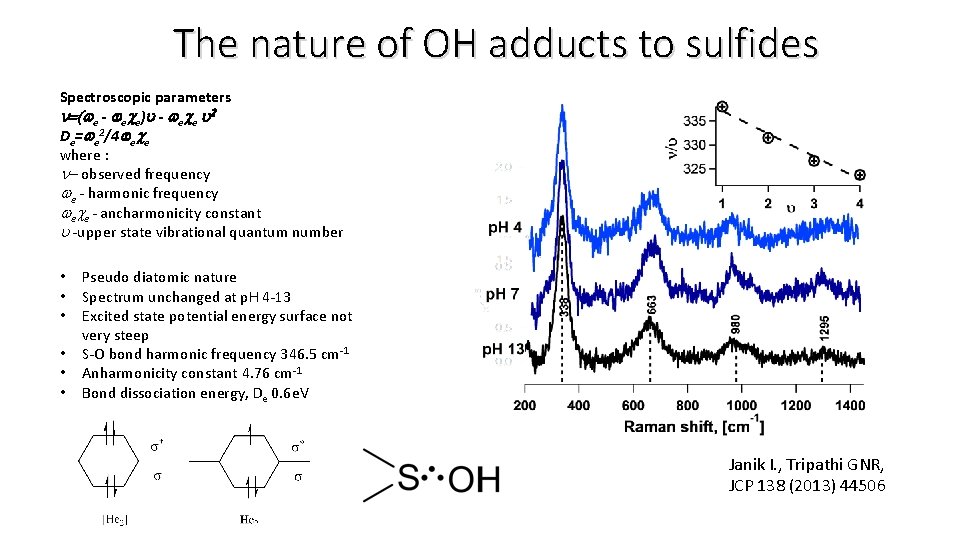
The nature of OH adducts to sulfides Spectroscopic parameters n=(we - wece)u - wece u 2 De=we 2/4 wece where : n- observed frequency we - harmonic frequency wece - ancharmonicity constant u -upper state vibrational quantum number • • • Pseudo diatomic nature Spectrum unchanged at p. H 4 -13 Excited state potential energy surface not very steep S-O bond harmonic frequency 346. 5 cm-1 Anharmonicity constant 4. 76 cm-1 Bond dissociation energy, De 0. 6 e. V Janik I. , Tripathi GNR, JCP 138 (2013) 44506

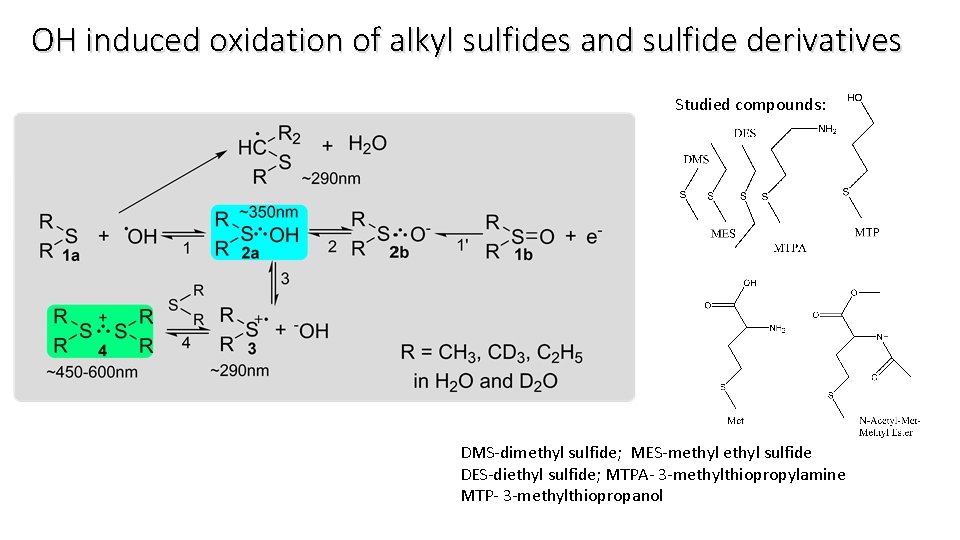
OH induced oxidation of alkyl sulfides and sulfide derivatives Studied compounds: DMS-dimethyl sulfide; MES-methyl sulfide DES-diethyl sulfide; MTPA- 3 -methylthiopropylamine MTP- 3 -methylthiopropanol

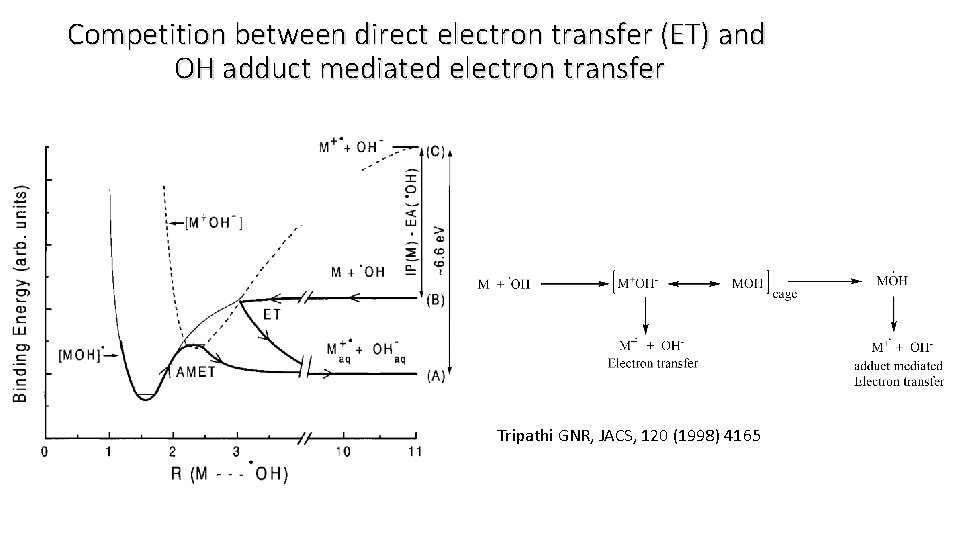
Competition between direct electron transfer (ET) and OH adduct mediated electron transfer Tripathi GNR, JACS, 120 (1998) 4165

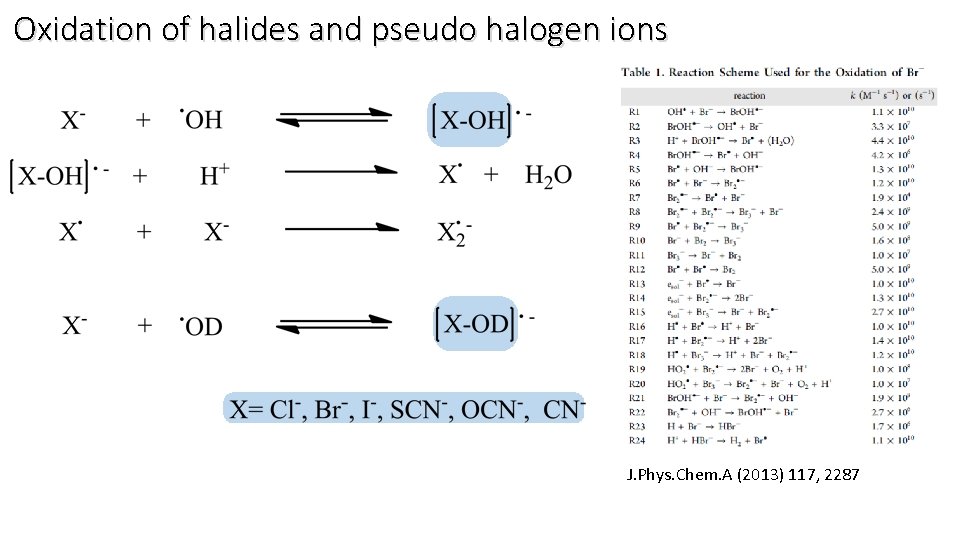
Oxidation of halides and pseudo halogen ions J. Phys. Chem. A (2013) 117, 2287

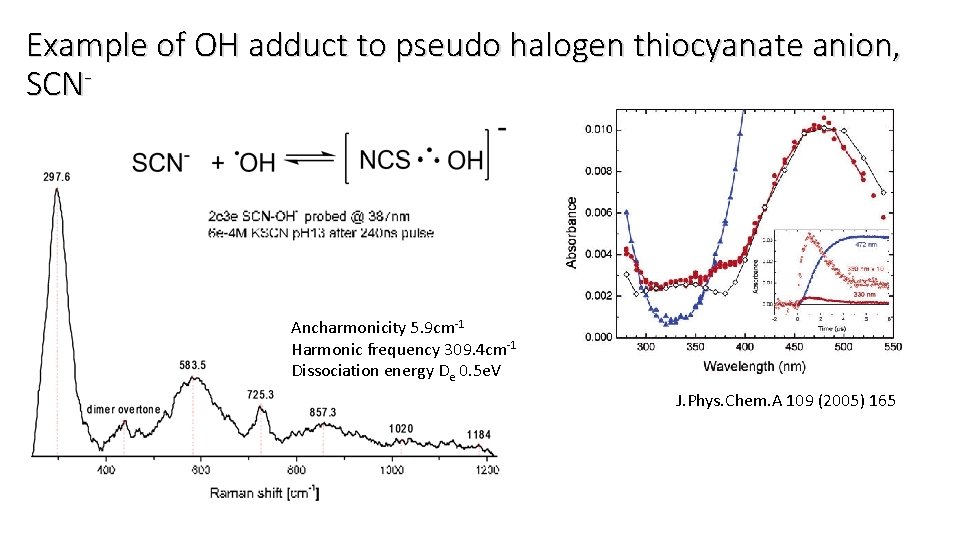
Example of OH adduct to pseudo halogen thiocyanate anion, SCN- Ancharmonicity 5. 9 cm-1 Harmonic frequency 309. 4 cm-1 Dissociation energy De 0. 5 e. V J. Phys. Chem. A 109 (2005) 165
![Hemibonding of OH and halide ion in aqueous solution Lambda max nm Epsilon103 M1 Hemibonding of OH and halide ion in aqueous solution Lambda max [nm] Epsilon/103 [M-1](https://slidetodoc.com/presentation_image_h2/cfd01aceee2688a8e51f7e33c1be5d7d/image-11.jpg)
Hemibonding of OH and halide ion in aqueous solution Lambda max [nm] Epsilon/103 [M-1 cm-1] X-O stretching [cm-1] R (X-O) [A] Cl-OH 350 (348) 3. 7 (5. 7) 162, 180 2. 417 Br-OH 360 (343) 8 (7. 1) 285 2. 402 I-OH 335 (324) 4. 5 (6. 9) 277 2. 485 DFT with range-separated hybrid (RSH) exchange-correlation functional M. Yamaguchi, J. Phys. Chem. A 2011, 115, 14620

OH adduct to bromide anion, Br- Ancharmonicity 7. 75 cm-1 Harmonic frequency 309 cm-1 Bond dissociation energy De 0. 38 e. V J. Phys. Chem. A (2013) 117, 2287 R 2 est. ~ 2. 5 x 107 s-1

Oxidation of transition metal cations

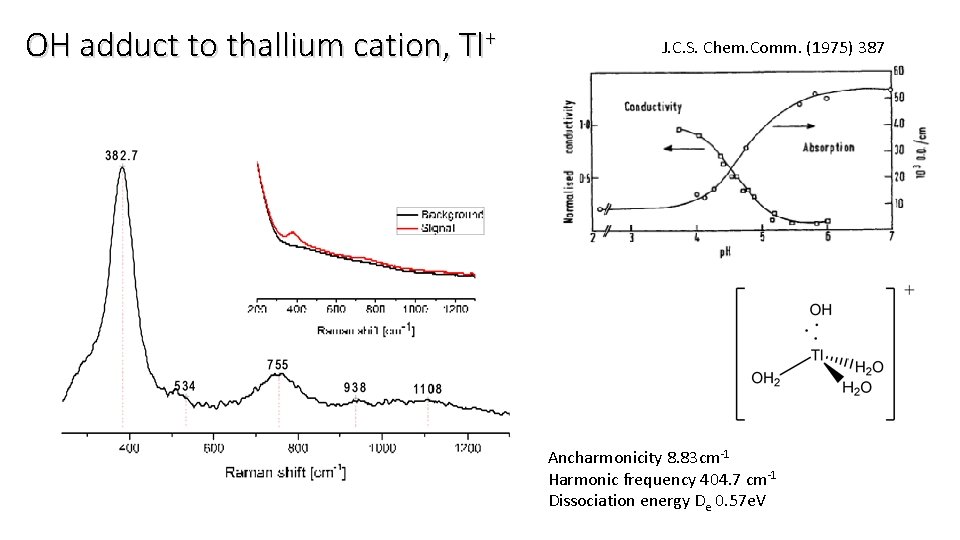
OH adduct to thallium cation, Tl+ J. C. S. Chem. Comm. (1975) 387 Ancharmonicity 8. 83 cm-1 Harmonic frequency 404. 7 cm-1 Dissociation energy De 0. 57 e. V

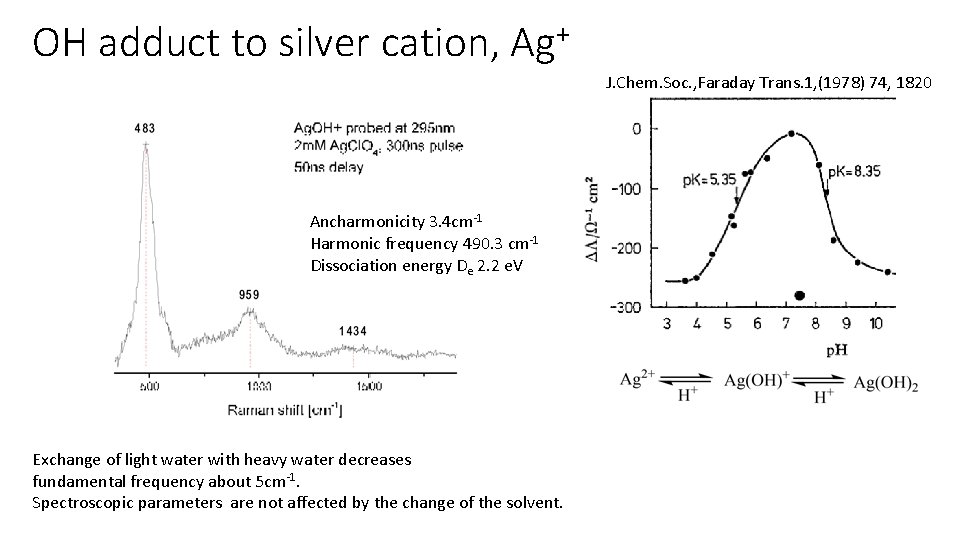
OH adduct to silver cation, + Ag J. Chem. Soc. , Faraday Trans. 1, (1978) 74, 1820 Ancharmonicity 3. 4 cm-1 Harmonic frequency 490. 3 cm-1 Dissociation energy De 2. 2 e. V Exchange of light water with heavy water decreases fundamental frequency about 5 cm-1. Spectroscopic parameters are not affected by the change of the solvent.

Conclusions • Hydroxyl radical adducts to sulfur containing organic compounds, halide ions as well as some metal cations have been observed for the first time using pulse radiolysis with TRRR detection technique • In majority of observed adduct intermediates bond between hydroxyl radical oxygen and parent molecule has two-center three-electron character recognized by its fundamental vibration and progression of overtones • The formation of OH adduct intermediates is determined by the rate of their vibrational relaxation (reorganization) competing with the rate of solvation of products formed in alternative electron transfer process • Assuming a first order anharmonicity in stretching motion of studied adduct intermediates and applying Birge-Sponer extrapolation bond dissociation energies of studied adduct intermediates have been determined • Observed results may additionally serve as a experimental reference for the applicability of existing computational methods in determination of spectroscopic parameters of 2 c-3 e bond intermediates

Acknowledgements Students Daniel Stuart Rohit Chikkaraddy Faculty Gordon Hug Ian Carmichael