Organic Chemistry Oxygen and Sulfur in Organic Compounds









- Slides: 16

Organic Chemistry Oxygen and Sulfur in Organic Compounds 1

Chapter 12 Part 06 Ethers Page 104 2

1. Ethers - contain the R-O-R functional group - the –OR group is termed alkoxy - derivatives of water where both hydrogens are replaced by R groups H O H R O R Ar O Ar 3

2. Classification - can be symmetrical or unsymmetrical - symmetrical ethers contain R O two identical R groups - unsymmetrical ethers contain R two different R groups R O R’ 4

3. Nomenclature - IUPAC - Determine main chain (longest continuous chain that does NOT contain the –OR group - Identify the remaining short chain as an alkoxy group CH 3 O is methoxy C 2 H 5 O is ethoxy, etc. - Number the chain so the C to which the –OR is attached has the lowest number - Place the name of the alkoxy group in front of the name of the main chain indicate the position of the alkoxy group on the main chain with a number 5

3. Nomenclature - Common - Identify the 2 substituents, either alkyl or aryl, attached to the ether linkage. - Name the substituents alphabetically followed by the word ether - If the ether is symmetrical (2 substituents are the same), the prefix di- is used 6

3. Nomenclature Name the following ethers by either the IUPAC system or the common name. Indicate if it is symmetrical or unsymmetrical. 7

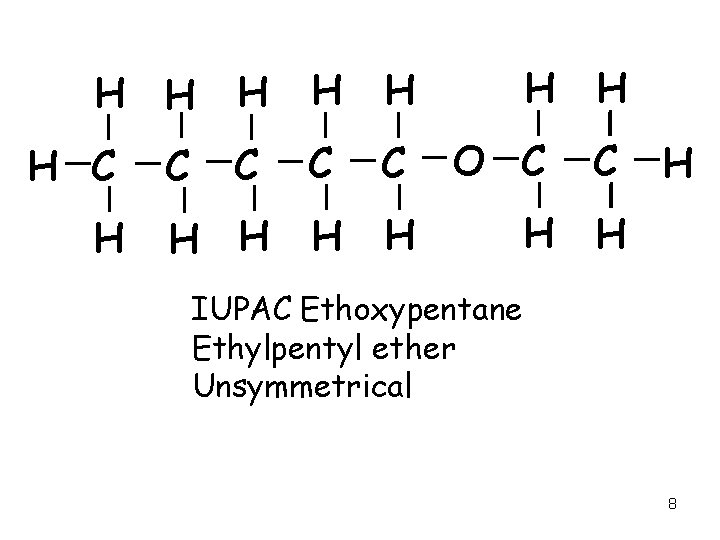
H H H C C C O C C H H H H H C H IUPAC Ethoxypentane Ethylpentyl ether Unsymmetrical 8

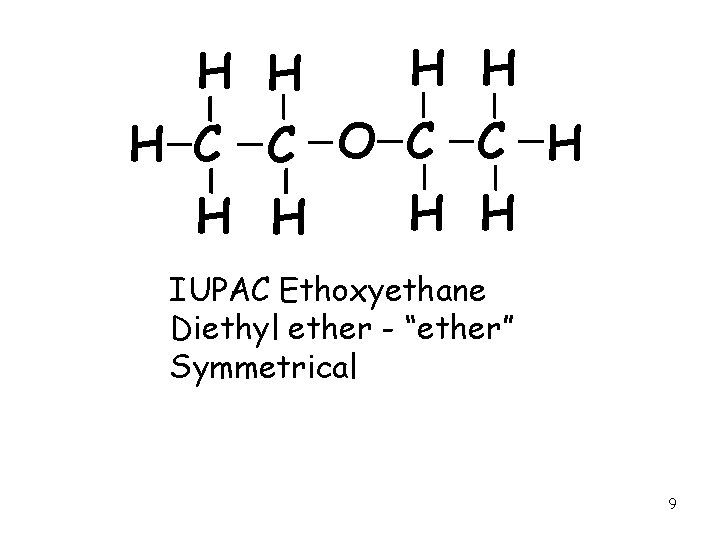
H H H C C O C C H H H IUPAC Ethoxyethane Diethyl ether - “ether” Symmetrical 9

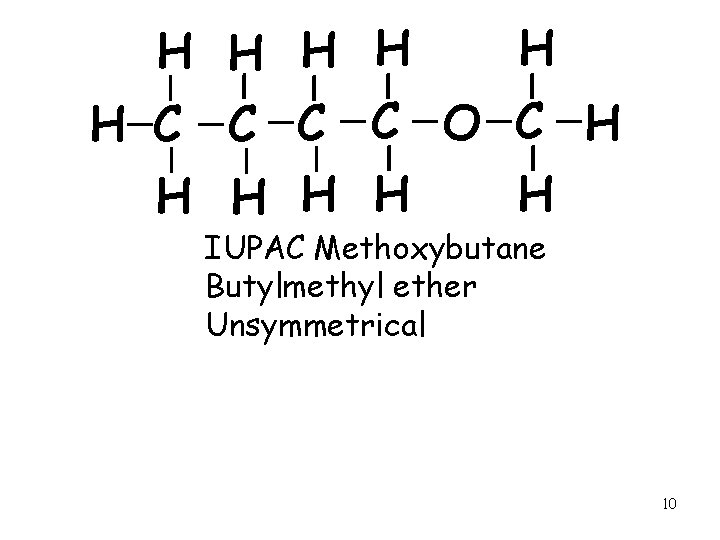
H H H C C O C H H H IUPAC Methoxybutane Butylmethyl ether Unsymmetrical 10

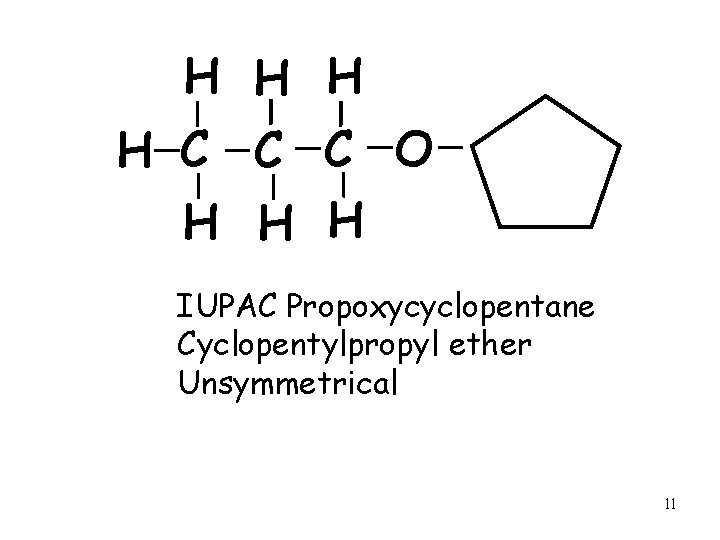
H H C C C O H H H IUPAC Propoxycyclopentane Cyclopentylpropyl ether Unsymmetrical 11

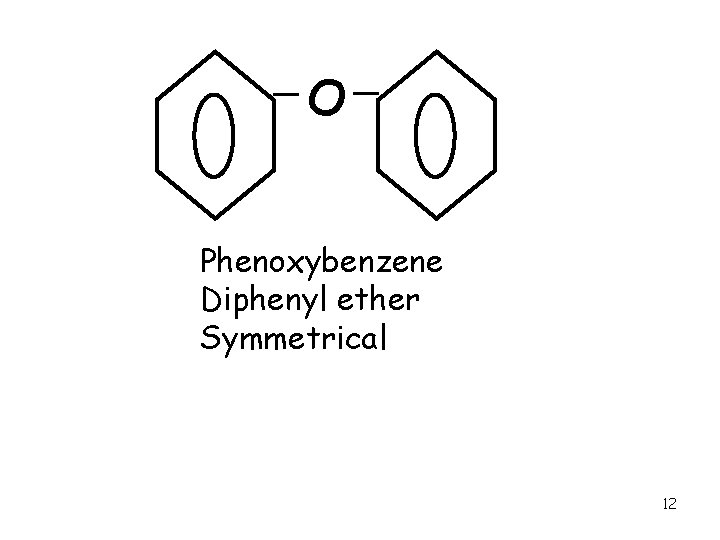
O Phenoxybenzene Diphenyl ether Symmetrical 12

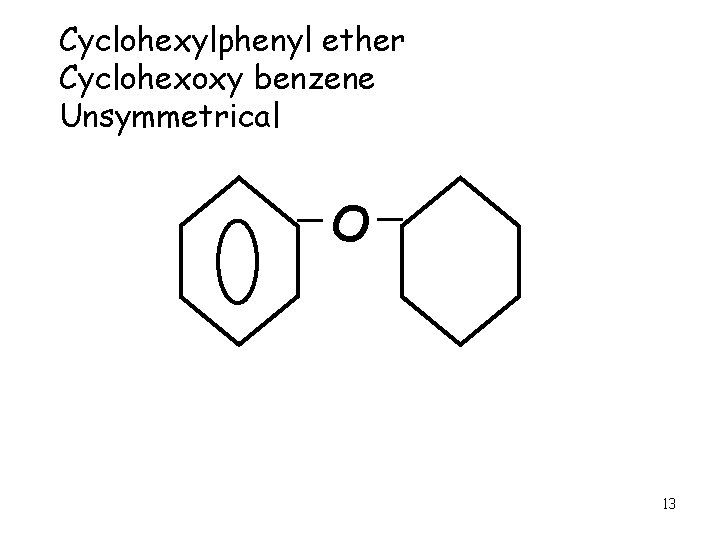
Cyclohexylphenyl ether Cyclohexoxy benzene Unsymmetrical O 13

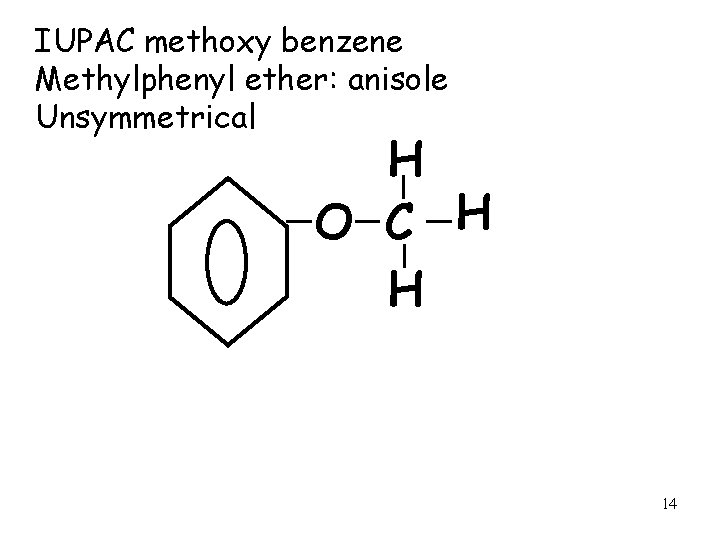
IUPAC methoxy benzene Methylphenyl ether: anisole Unsymmetrical H O C H H 14

4. Properties and Uses - prepared from dehydration of 2 alcohols - colorless, - slightly soluble in water - very soluble in organic solvents - density & boiling points similar to hydrocarbons of similar mass 15

4. Properties and Uses - largely unreactive (stable in acids and bases; resistant to oxidizing and reducing agents) - excellent solvents for chemical reactions - highly flammable - form explosive organic peroxides on standing Diethyl ether and tetrahydrofuran – most widely used organic solvents 16