Organic compounds containing oxygen nitrogen and sulfur Biologically

Organic compounds containing oxygen, nitrogen and sulfur Biologically important heterocycles Martina Srbová

Organic compounds containing oxygen
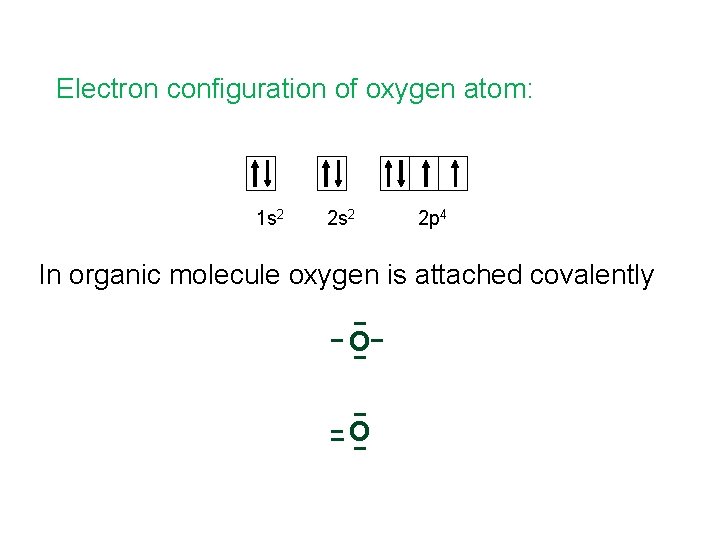
Electron configuration of oxygen atom: 1 s 2 2 p 4 In organic molecule oxygen is attached covalently O O

q Alcohols and phenols (hydroxy derivates) – compounds with the hydroxyl (-OH) R-O-H q Ethers – compounds with alcoxyl group (-OR) R-O-R
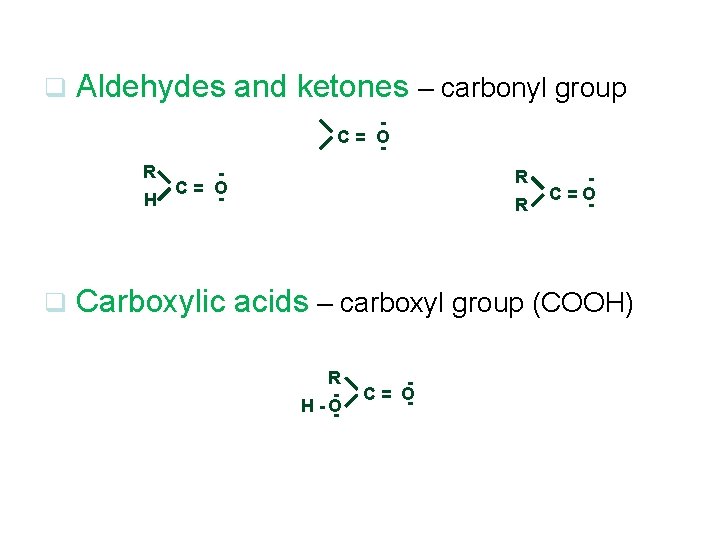
q Aldehydes and ketones – carbonyl group - C= O R C= O H q R R - C=O Carboxylic acids – carboxyl group (COOH) R H -O - - C = O-

Alcohols Classification In 1 o alcohol, only one carbon atom is attached to the carbon carrying the -OH group (primary carbon). q In 2 o alcohol two carbon atoms are attached to the carbon carrying the -OH group (secondary carbon). q In 3 o alcohol three other carbon atoms are attached to the carbon atom carrying the -OH group (tertiary carbon). q

The number of hydroxyl groups, there are: Monohydroxyderivatives (monohydroxy alcohols) Polyhydroxy alcohols q Diols (dihydroxyderivatives, ) q q Triols (trihydroxyderivatives) Tetrols (tetrahydroxyderivatives) Phenols -OH attached primary to aromatic ring
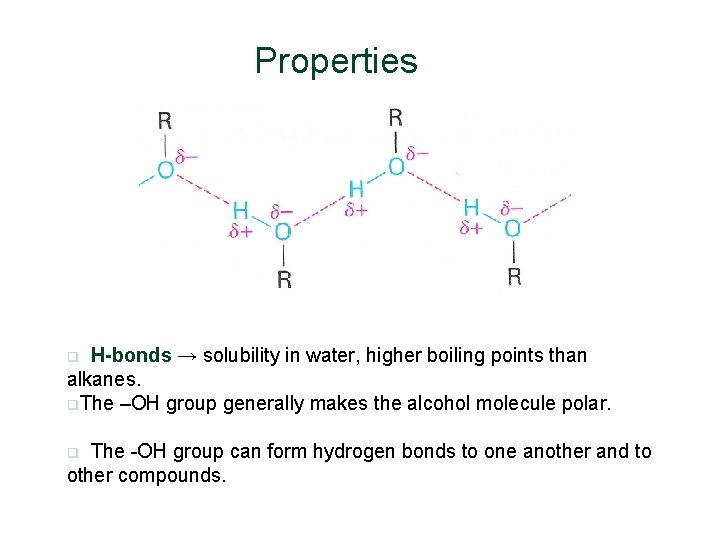
Properties H-bonds → solubility in water, higher boiling points than alkanes. q. The –OH group generally makes the alcohol molecule polar. q The -OH group can form hydrogen bonds to one another and to other compounds. q
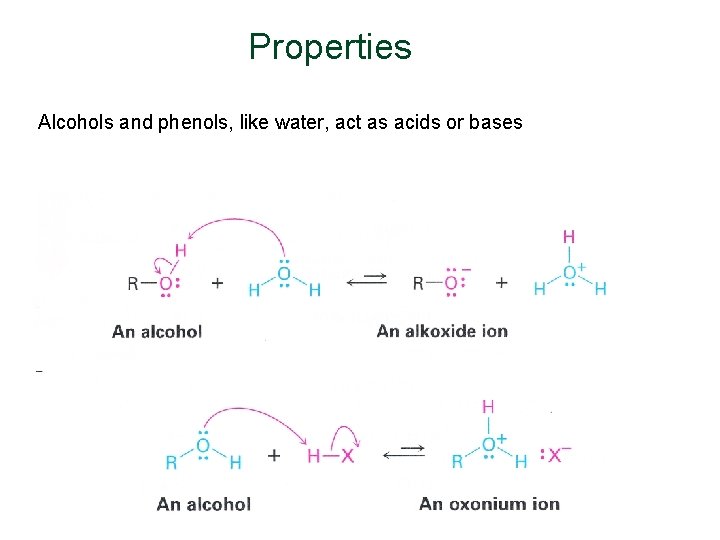
Properties Alcohols and phenols, like water, act as acids or bases

Methanol CH 3 -OH Toxic substance, temporary blindness (15 ml), permanent blindness or death (30 ml) Ethanol CH 3 -CH 2 -OH q. Obtained by fermentation from sugar juices in the presence of yeast and temperature of less than 37°C . q Lethal dose is 6 -8 g/kg ( 1 L of vodka)
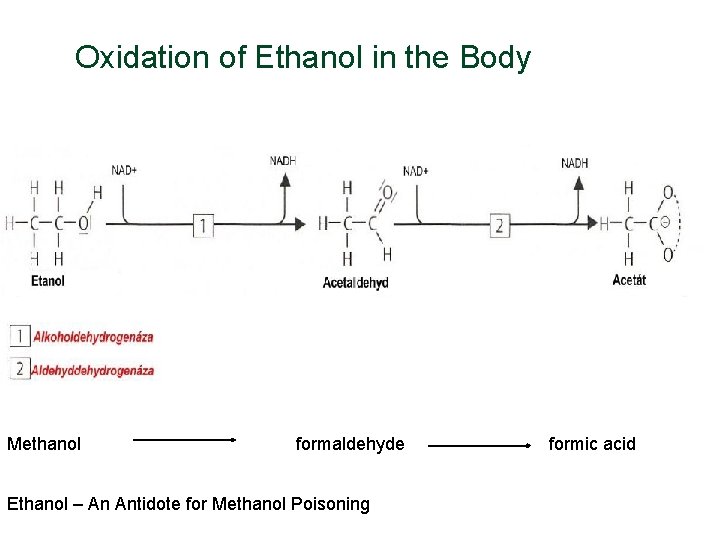
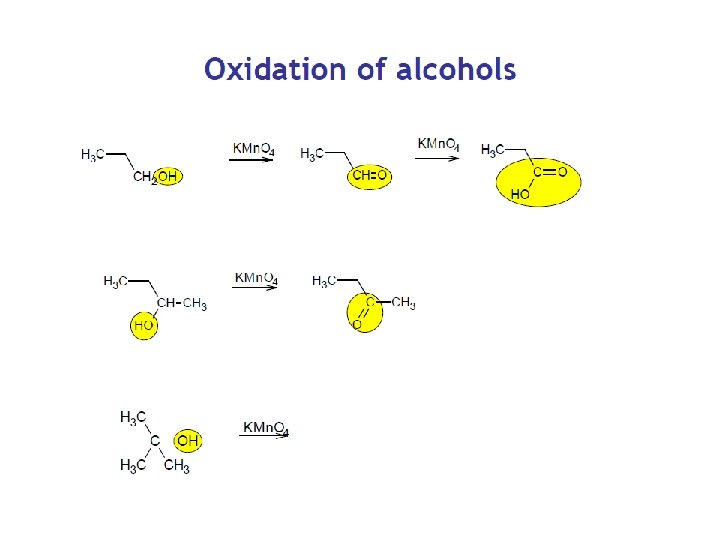
Oxidation of Ethanol in the Body Methanol formaldehyde Ethanol – An Antidote for Methanol Poisoning formic acid
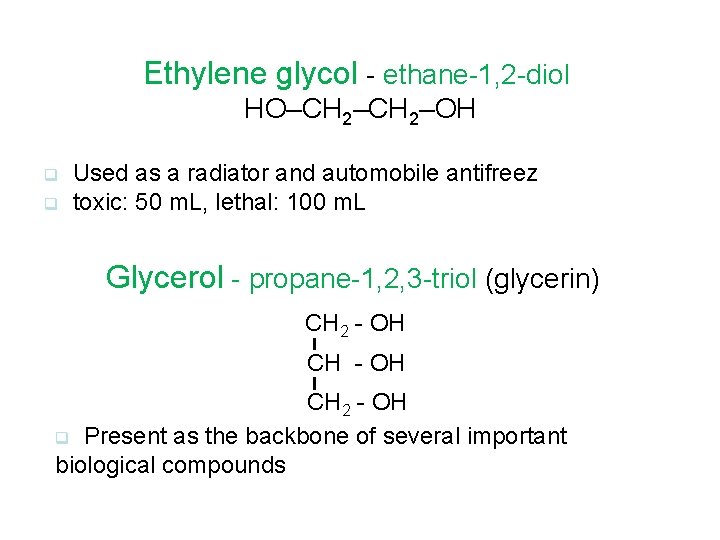
Ethylene glycol - ethane-1, 2 -diol HO–CH 2–OH q q Used as a radiator and automobile antifreez toxic: 50 m. L, lethal: 100 m. L Glycerol - propane-1, 2, 3 -triol (glycerin) CH 2 - OH q Present as the backbone of several important biological compounds
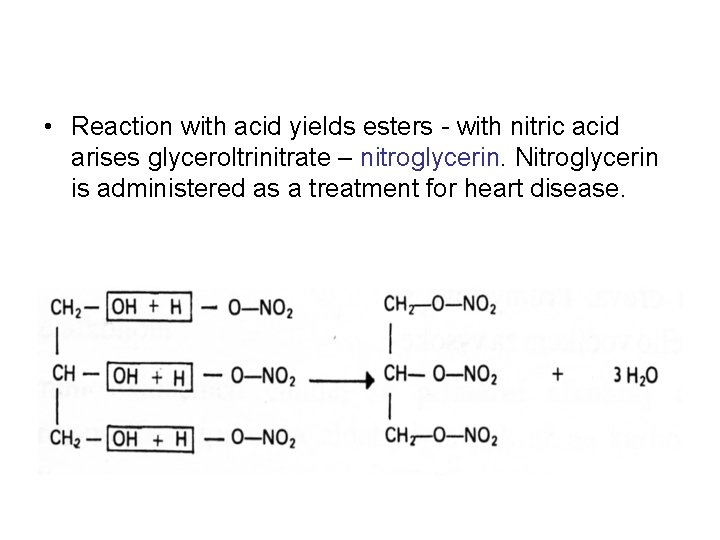
• Reaction with acid yields esters - with nitric acid arises glyceroltrinitrate – nitroglycerin. Nitroglycerin is administered as a treatment for heart disease.
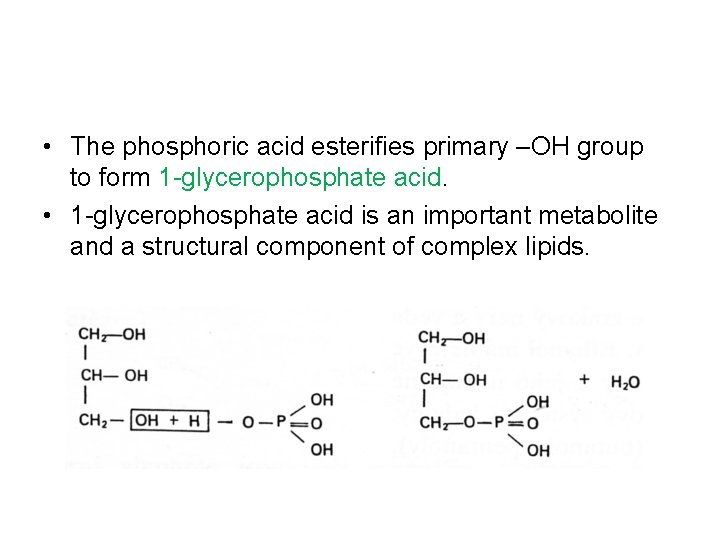
• The phosphoric acid esterifies primary –OH group to form 1 -glycerophosphate acid. • 1 -glycerophosphate acid is an important metabolite and a structural component of complex lipids.
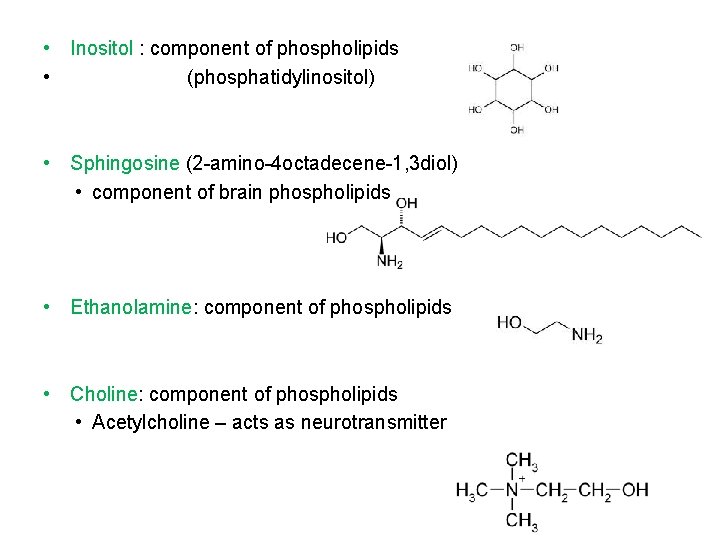
• Inositol : component of phospholipids • (phosphatidylinositol) • Sphingosine (2 -amino-4 octadecene-1, 3 diol) • component of brain phospholipids • Ethanolamine: component of phospholipids • Choline: component of phospholipids • Acetylcholine – acts as neurotransmitter
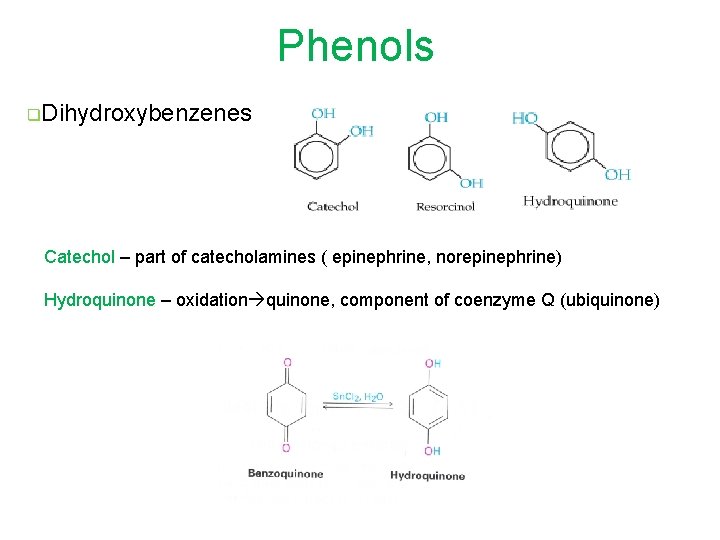
Phenols q. Dihydroxybenzenes Catechol – part of catecholamines ( epinephrine, norepinephrine) Hydroquinone – oxidation quinone, component of coenzyme Q (ubiquinone)
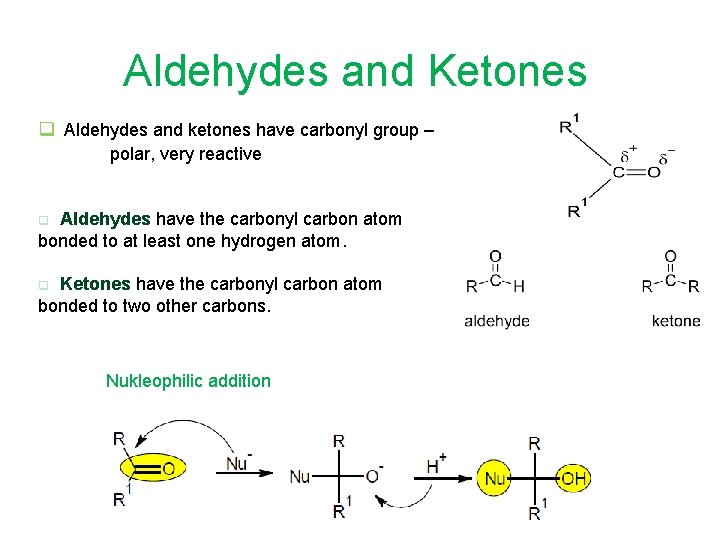
Aldehydes and Ketones q Aldehydes and ketones have carbonyl group – polar, very reactive Aldehydes have the carbonyl carbon atom bonded to at least one hydrogen atom. q Ketones have the carbonyl carbon atom bonded to two other carbons. q Nukleophilic addition
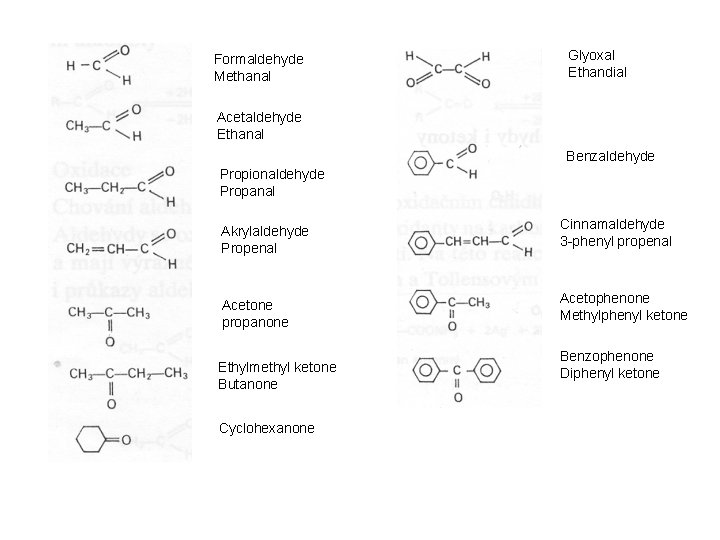
Formaldehyde Methanal Glyoxal Ethandial Acetaldehyde Ethanal Benzaldehyde Propionaldehyde Propanal Akrylaldehyde Propenal Cinnamaldehyde 3 -phenyl propenal Acetone propanone Acetophenone Methylphenyl ketone Ethylmethyl ketone Butanone Cyclohexanone Benzophenone Diphenyl ketone
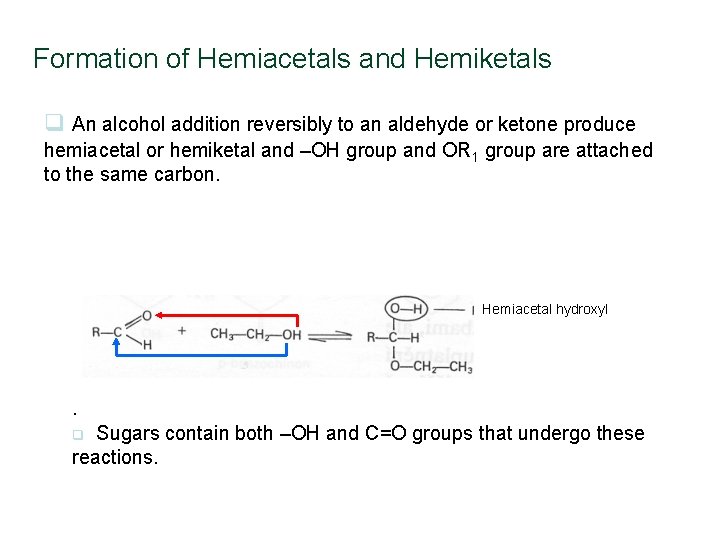
Formation of Hemiacetals and Hemiketals q An alcohol addition reversibly to an aldehyde or ketone produce hemiacetal or hemiketal and –OH group and OR 1 group are attached to the same carbon. Hemiacetal hydroxyl . Sugars contain both –OH and C=O groups that undergo these reactions. q
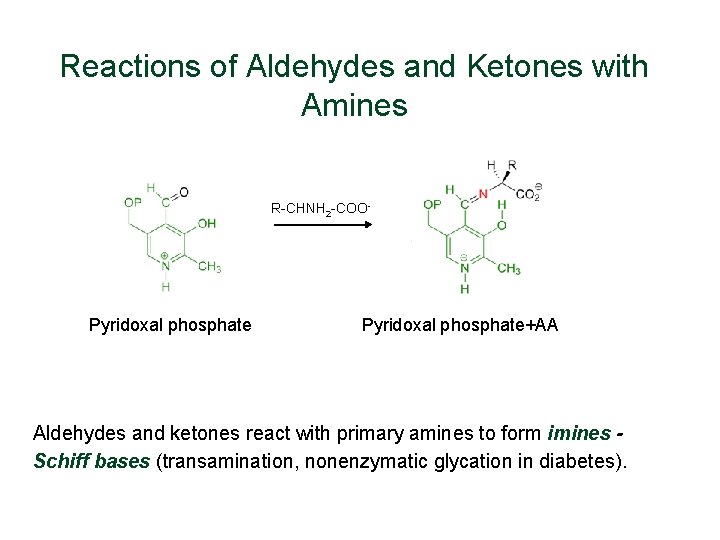
Reactions of Aldehydes and Ketones with Amines R-CHNH 2 -COO- Pyridoxal phosphate+AA Aldehydes and ketones react with primary amines to form imines Schiff bases (transamination, nonenzymatic glycation in diabetes).
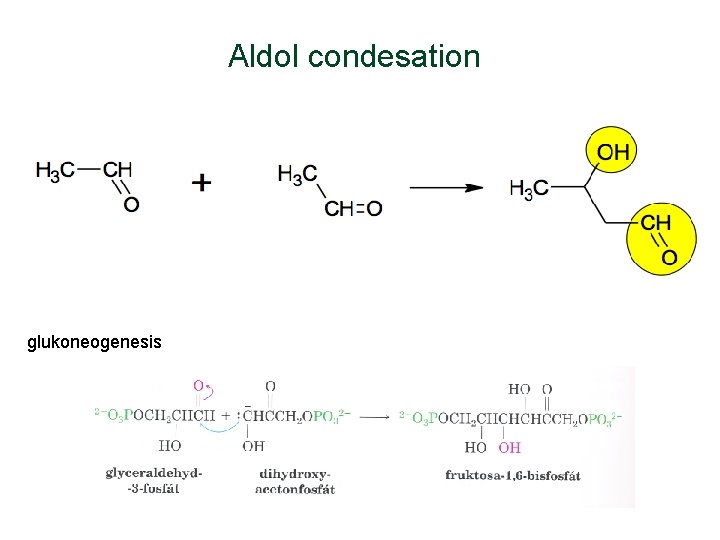
Aldol condesation glukoneogenesis
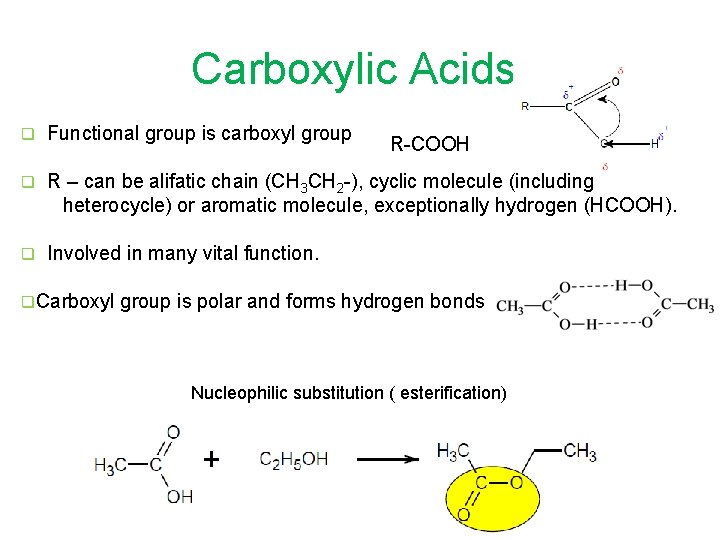
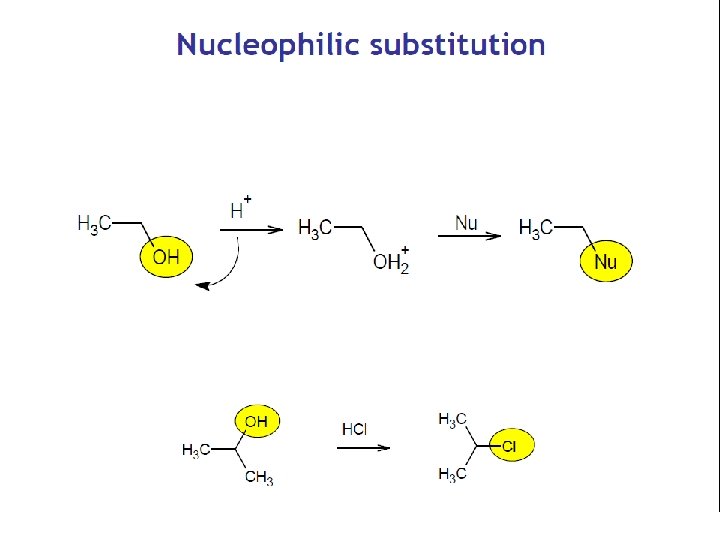
Carboxylic Acids q Functional group is carboxyl group q R – can be alifatic chain (CH 3 CH 2 -), cyclic molecule (including heterocycle) or aromatic molecule, exceptionally hydrogen (HCOOH). q Involved in many vital function. q. Carboxyl R-COOH group is polar and forms hydrogen bonds Nucleophilic substitution ( esterification)

Examples of monocarboxylic acids Formic acid Methanoic acid Stearic acid Octadecanoic acid Acetic acid Ethanoic acid Oleic acid Cis-9 -octadecanoic acid Propionic acid Propanoic acid Butyric acid Butanoic acid Isobutyric acid Isobutanoic acid Valeric acid Pentanoic acid Palmitic acid Hexadecanoic acid Acrylic acid Propenoic acid Crotonic acid trans-2 -butenoic acid Benzencarboxylic acid b-naphtoic acid 2 -naphtalenecarboxylic acid
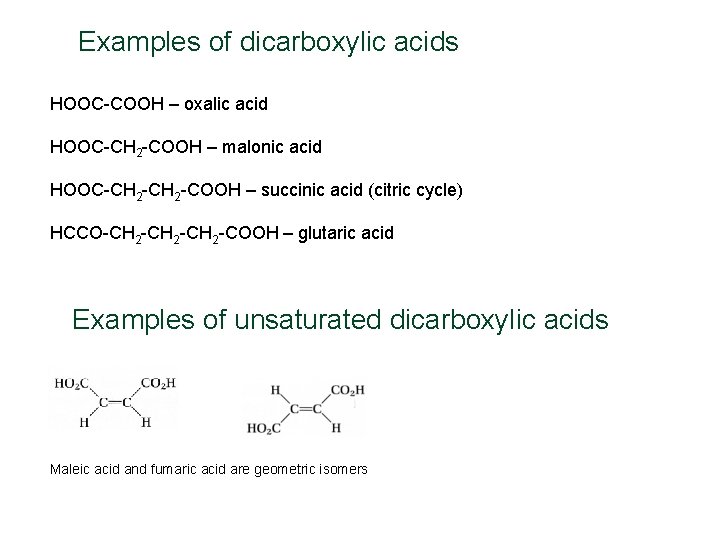
Examples of dicarboxylic acids HOOC-COOH – oxalic acid HOOC-CH 2 -COOH – malonic acid HOOC-CH 2 -COOH – succinic acid (citric cycle) HCCO-CH 2 -CH 2 -COOH – glutaric acid Examples of unsaturated dicarboxylic acids Maleic acid and fumaric acid are geometric isomers
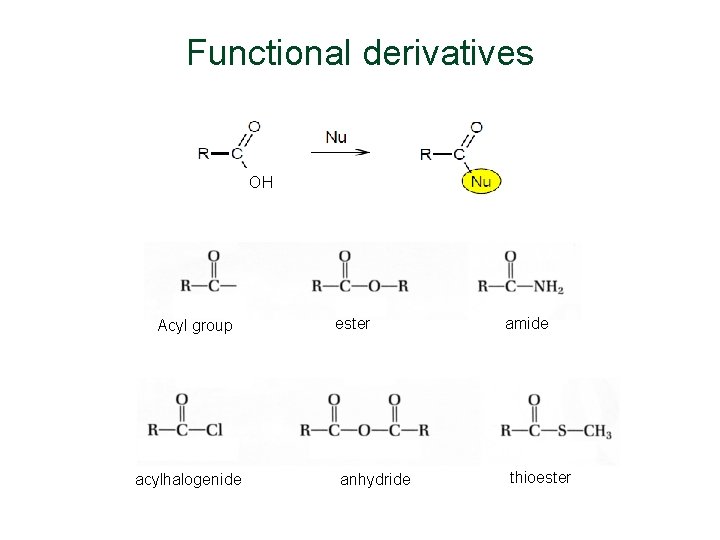
Functional derivatives OH Acyl group acylhalogenide ester anhydride amide thioester
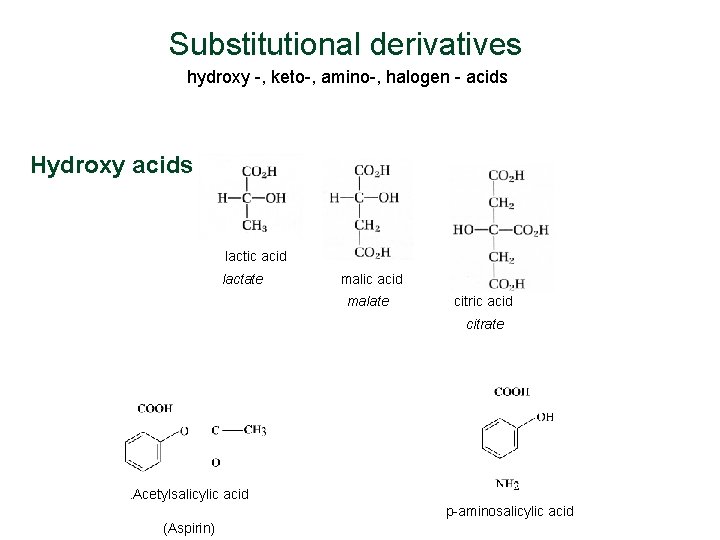
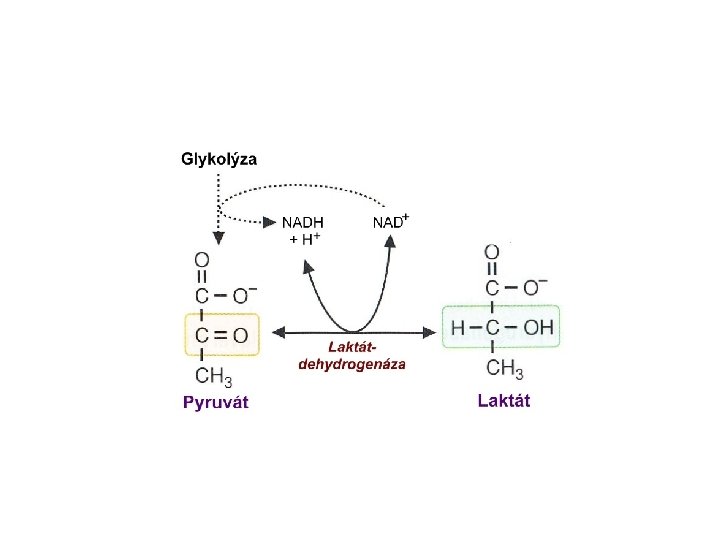
Substitutional derivatives hydroxy -, keto-, amino-, halogen - acids Hydroxy acids lactic acid lactate malic acid malate citric acid citrate . Acetylsalicylic acid p-aminosalicylic acid (Aspirin)
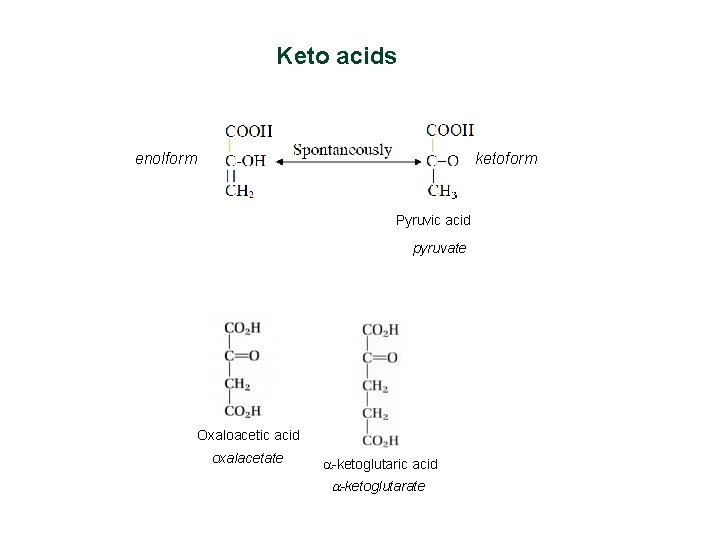
Keto acids enolform ketoform Pyruvic acid pyruvate Oxaloacetic acid oxalacetate a-ketoglutaric acid a-ketoglutarate

Organic compounds containing nitrogen
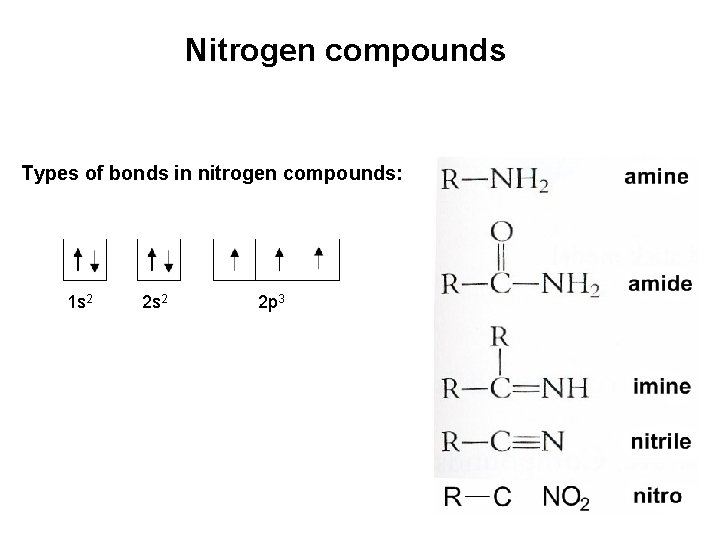
Nitrogen compounds Types of bonds in nitrogen compounds: 1 s 2 2 p 3
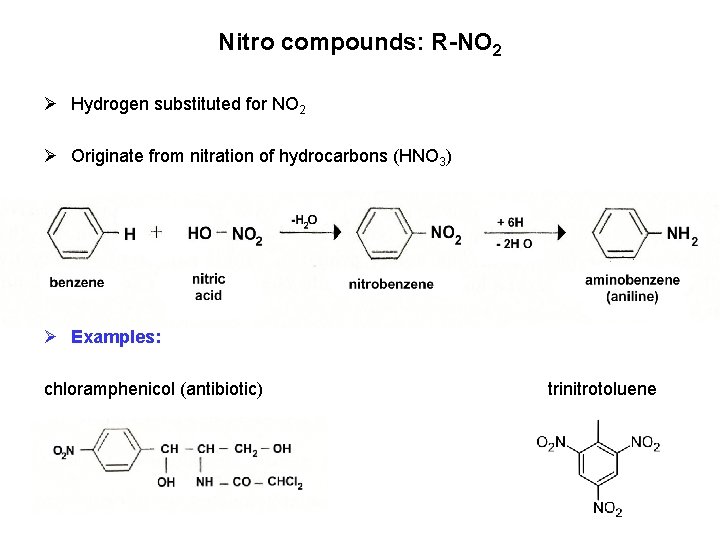
Nitro compounds: R-NO 2 Ø Hydrogen substituted for NO 2 Ø Originate from nitration of hydrocarbons (HNO 3) Ø Examples: chloramphenicol (antibiotic) trinitrotoluene
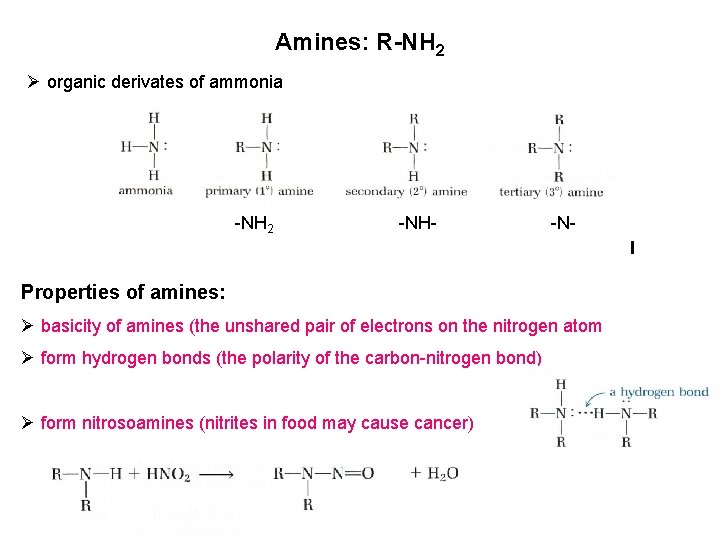
Amines: R-NH 2 Ø organic derivates of ammonia -NH 2 -NH- -N- Properties of amines: Ø basicity of amines (the unshared pair of electrons on the nitrogen atom Ø form hydrogen bonds (the polarity of the carbon-nitrogen bond) Ø form nitrosoamines (nitrites in food may cause cancer)

Importance of amines: The most important organic bases In biochemistry: Ø Quaternary ammonium salts Ø Biogenic amines Ø Amino acids Ø Proteins Ø Alkaloids
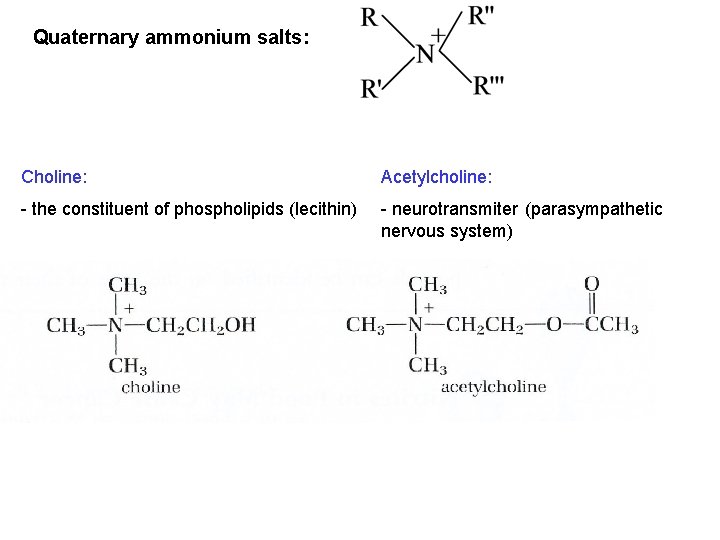
Quaternary ammonium salts: Choline: Acetylcholine: - the constituent of phospholipids (lecithin) - neurotransmiter (parasympathetic nervous system)

Acetylcholine - the transmitter of the parasympathetic and sympathetic system, at neuromuscular junctions, involved in learning and memory 1. The synthesis: from choline and acetyl-Co. A in the neurons 2. Hydrolysis: in the synaptict cleft (restores the resting potential in the postsynaptic membrane)
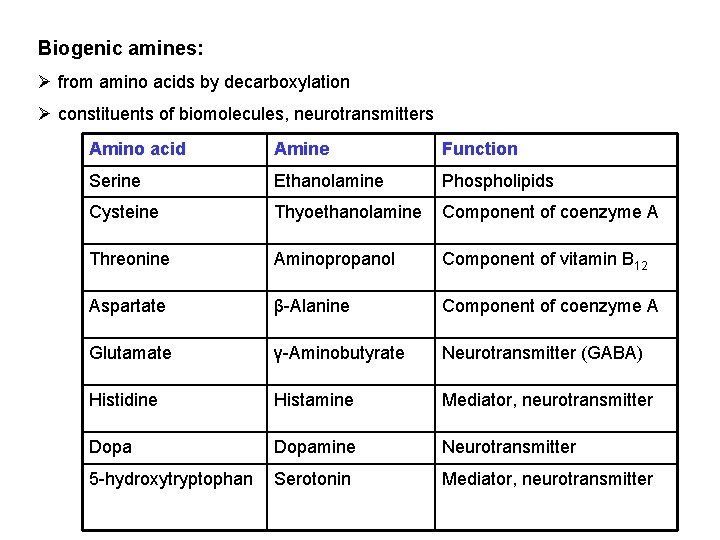
Biogenic amines: Ø from amino acids by decarboxylation Ø constituents of biomolecules, neurotransmitters Amino acid Amine Function Serine Ethanolamine Phospholipids Cysteine Thyoethanolamine Component of coenzyme A Threonine Aminopropanol Component of vitamin B 12 Aspartate β-Alanine Component of coenzyme A Glutamate γ-Aminobutyrate Neurotransmitter (GABA) Histidine Histamine Mediator, neurotransmitter Dopamine Neurotransmitter 5 -hydroxytryptophan Serotonin Mediator, neurotransmitter
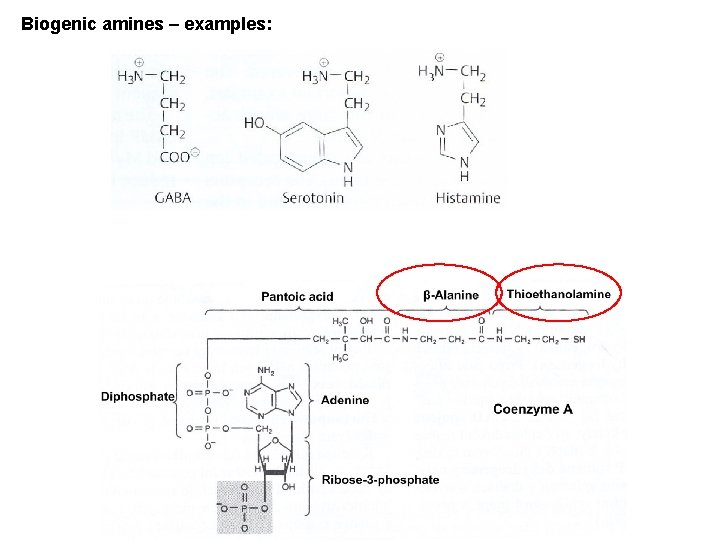
Biogenic amines – examples:
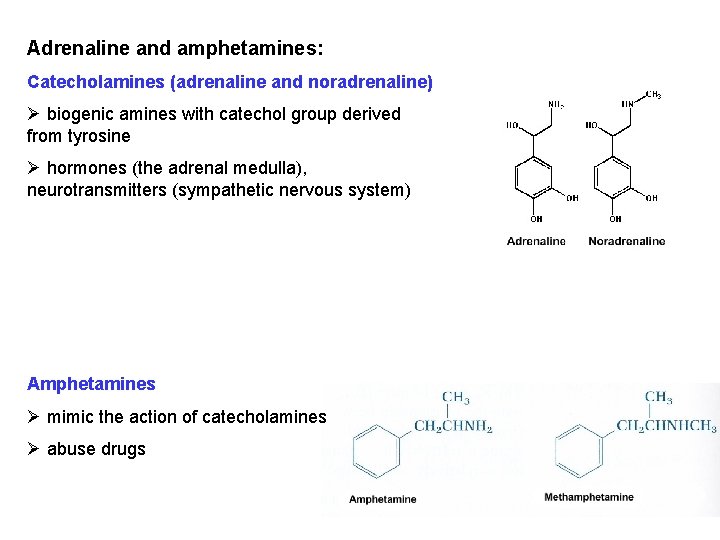
Adrenaline and amphetamines: Catecholamines (adrenaline and noradrenaline) Ø biogenic amines with catechol group derived from tyrosine Ø hormones (the adrenal medulla), neurotransmitters (sympathetic nervous system) Amphetamines Ø mimic the action of catecholamines Ø abuse drugs
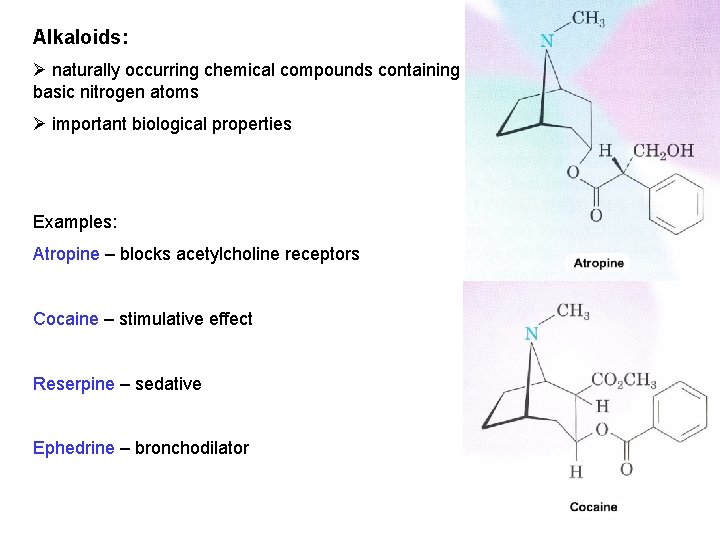
Alkaloids: Ø naturally occurring chemical compounds containing basic nitrogen atoms Ø important biological properties Examples: Atropine – blocks acetylcholine receptors Cocaine – stimulative effect Reserpine – sedative Ephedrine – bronchodilator
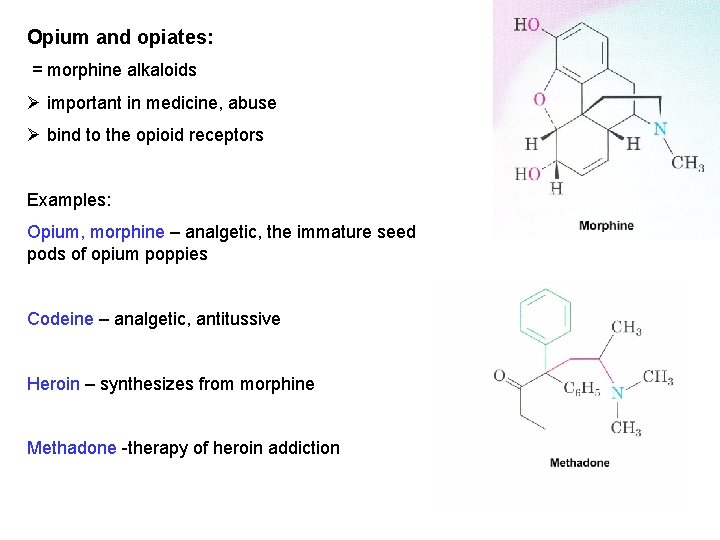
Opium and opiates: = morphine alkaloids Ø important in medicine, abuse Ø bind to the opioid receptors Examples: Opium, morphine – analgetic, the immature seed pods of opium poppies Codeine – analgetic, antitussive Heroin – synthesizes from morphine Methadone -therapy of heroin addiction
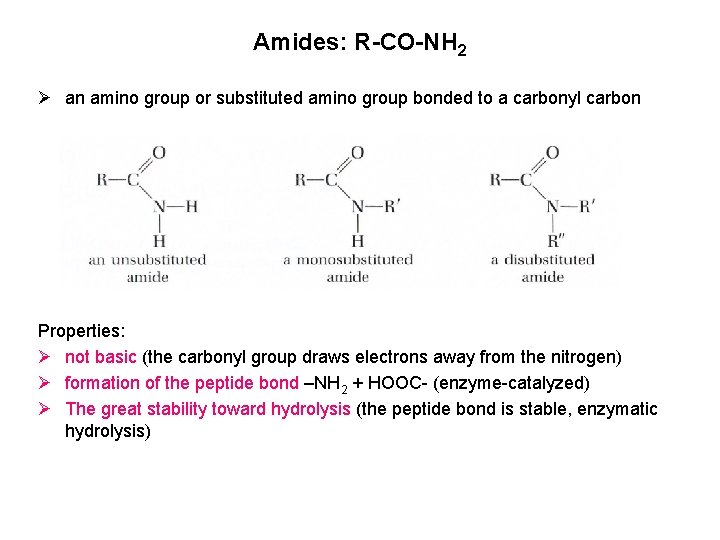
Amides: R-CO-NH 2 Ø an amino group or substituted amino group bonded to a carbonyl carbon Properties: Ø not basic (the carbonyl group draws electrons away from the nitrogen) Ø formation of the peptide bond –NH 2 + HOOC- (enzyme-catalyzed) Ø The great stability toward hydrolysis (the peptide bond is stable, enzymatic hydrolysis)
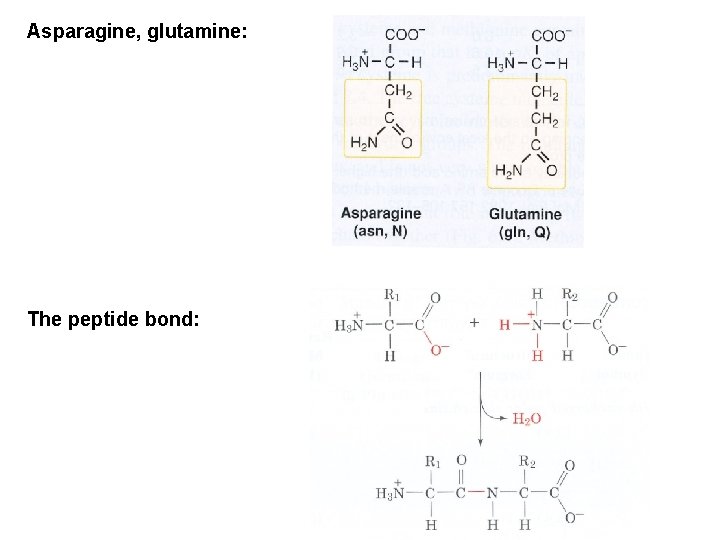
Asparagine, glutamine: The peptide bond:
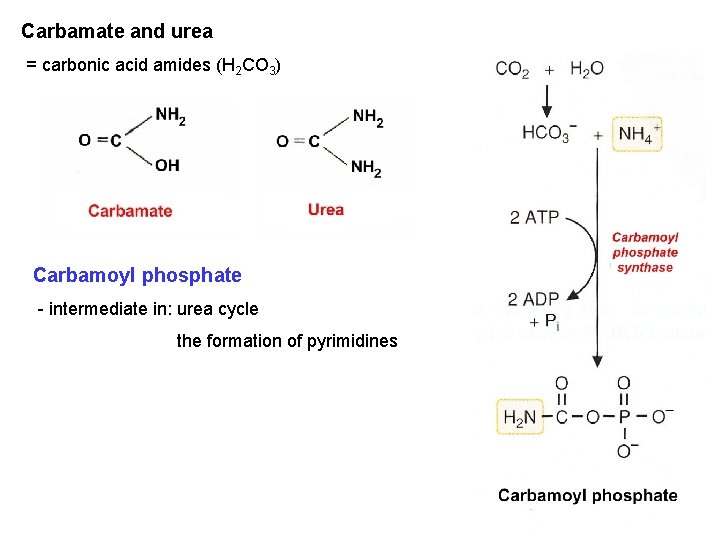
Carbamate and urea = carbonic acid amides (H 2 CO 3) Carbamoyl phosphate - intermediate in: urea cycle the formation of pyrimidines
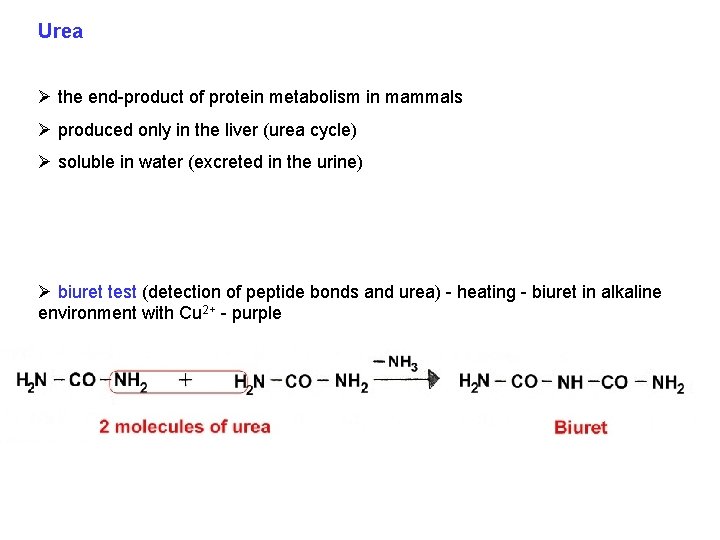
Urea Ø the end-product of protein metabolism in mammals Ø produced only in the liver (urea cycle) Ø soluble in water (excreted in the urine) Ø biuret test (detection of peptide bonds and urea) - heating - biuret in alkaline environment with Cu 2+ - purple
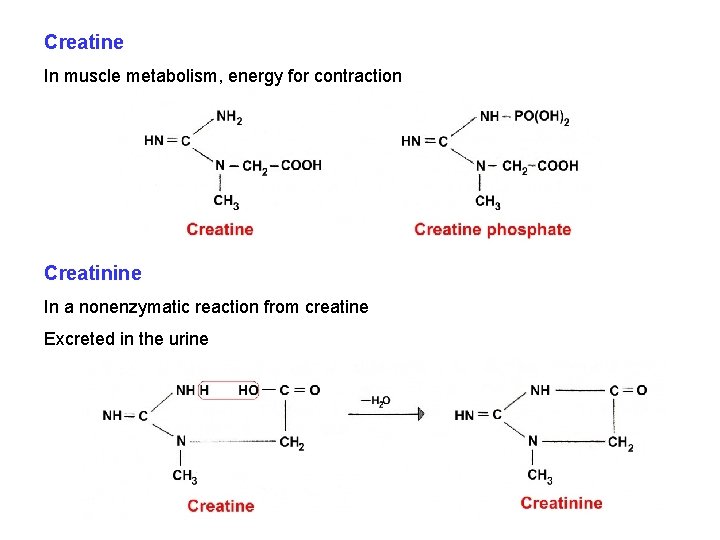
Creatine In muscle metabolism, energy for contraction Creatinine In a nonenzymatic reaction from creatine Excreted in the urine
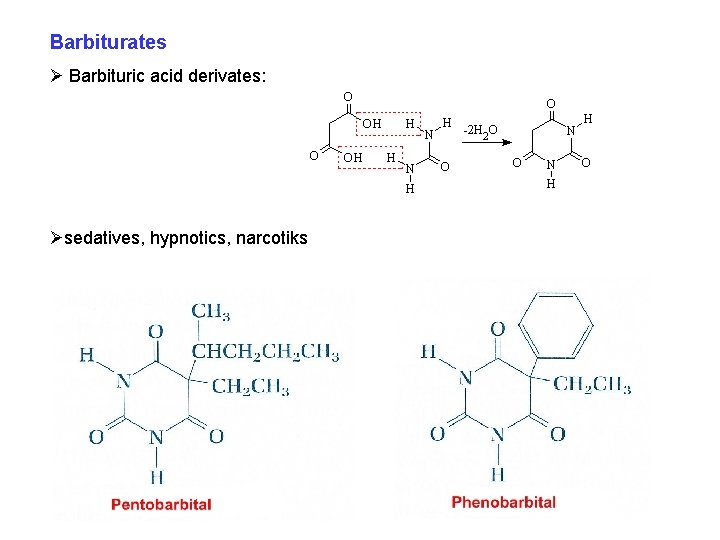
Barbiturates Ø Barbituric acid derivates: Øsedatives, hypnotics, narcotiks
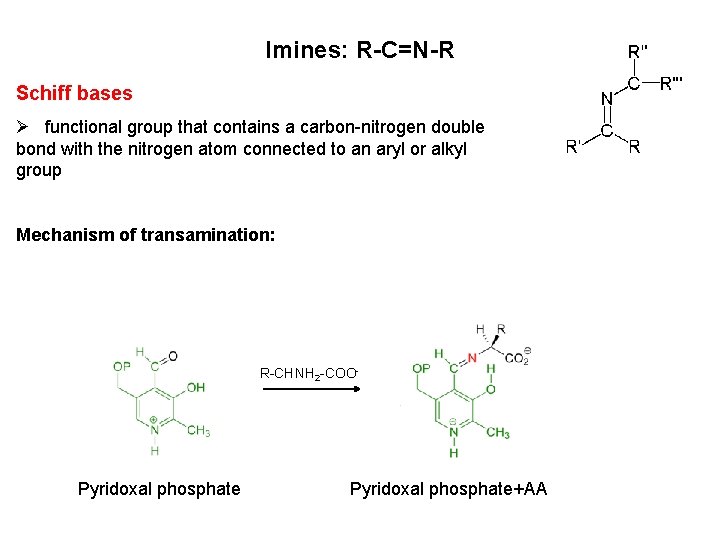
Imines: R-C=N-R Schiff bases Ø functional group that contains a carbon-nitrogen double bond with the nitrogen atom connected to an aryl or alkyl group Mechanism of transamination: R-CHNH 2 -COO- Pyridoxal phosphate+AA

Organic compounds containing sulfur

Sulfur compounds Structurally similar to oxygen compound
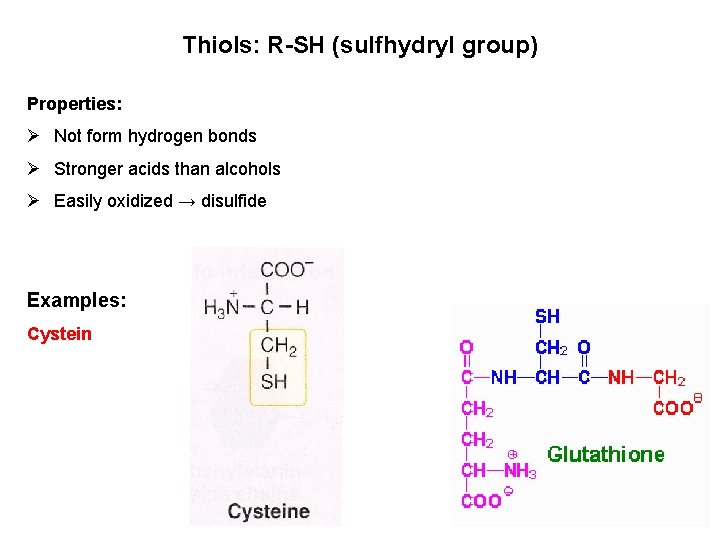
Thiols: R-SH (sulfhydryl group) Properties: Ø Not form hydrogen bonds Ø Stronger acids than alcohols Ø Easily oxidized → disulfide Examples: Cystein

Disulfides: R-S-S-R Disulfide-bonded cystein residues Ø The disulfide bond forms when the two thiol groups are oxidized (reverse reaction) Ø Protein conformation!
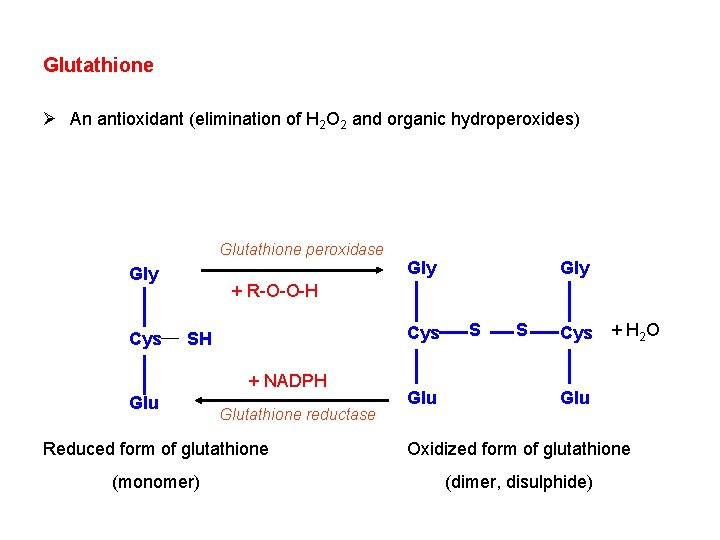
Glutathione Ø An antioxidant (elimination of H 2 O 2 and organic hydroperoxides) Glutathione peroxidase Gly Cys Gly + R-O-O-H Cys SH + NADPH Glutathione reductase Glu S S Cys + H 2 O Glu Reduced form of glutathione Oxidized form of glutathione (monomer) (dimer, disulphide)
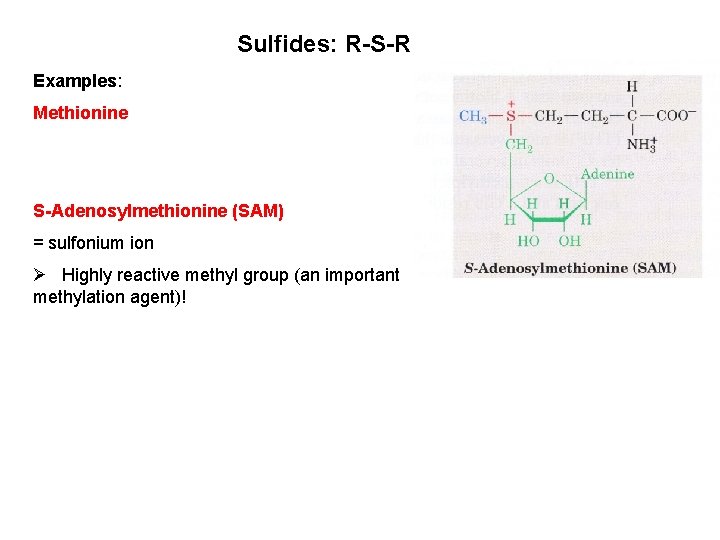
Sulfides: R-S-R Examples: Methionine S-Adenosylmethionine (SAM) = sulfonium ion Ø Highly reactive methyl group (an important methylation agent)!
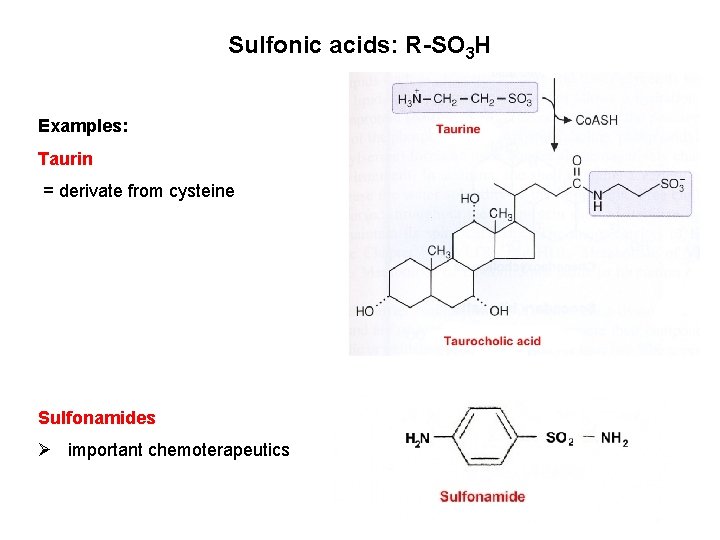
Sulfonic acids: R-SO 3 H Examples: Taurin = derivate from cysteine Sulfonamides Ø important chemoterapeutics

Biologically important heterocycles
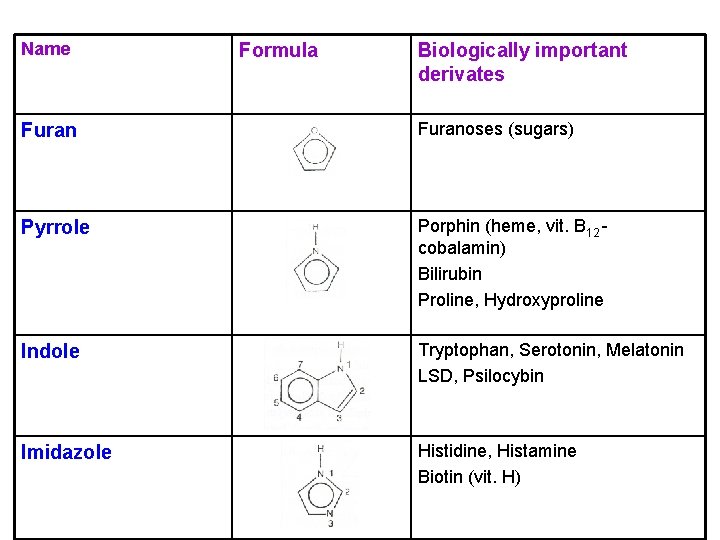
Name Formula Biologically important derivates Furanoses (sugars) Pyrrole Porphin (heme, vit. B 12 cobalamin) Bilirubin Proline, Hydroxyproline Indole Tryptophan, Serotonin, Melatonin LSD, Psilocybin Imidazole Histidine, Histamine Biotin (vit. H)
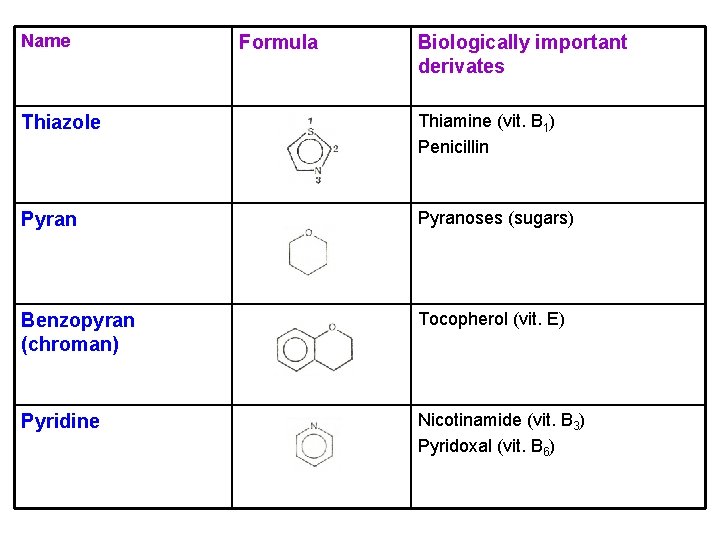
Name Formula Biologically important derivates Thiazole Thiamine (vit. B 1) Penicillin Pyranoses (sugars) Benzopyran (chroman) Tocopherol (vit. E) Pyridine Nicotinamide (vit. B 3) Pyridoxal (vit. B 6)
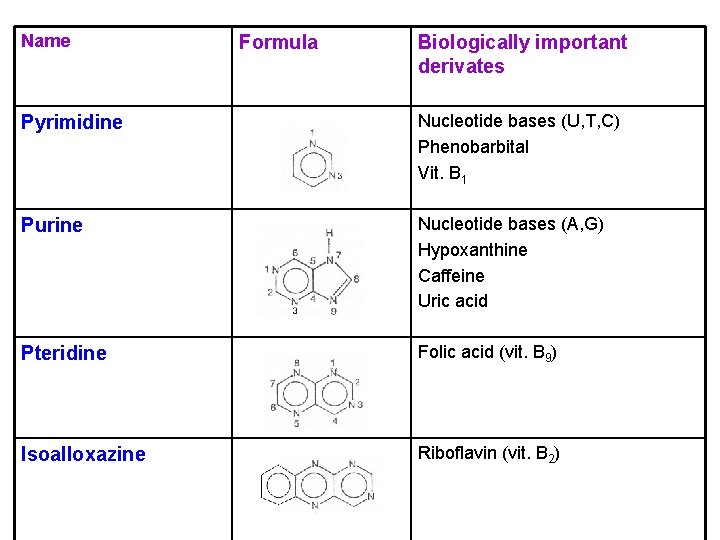
Name Formula Biologically important derivates Pyrimidine Nucleotide bases (U, T, C) Phenobarbital Vit. B 1 Purine Nucleotide bases (A, G) Hypoxanthine Caffeine Uric acid Pteridine Folic acid (vit. B 9) Isoalloxazine Riboflavin (vit. B 2)
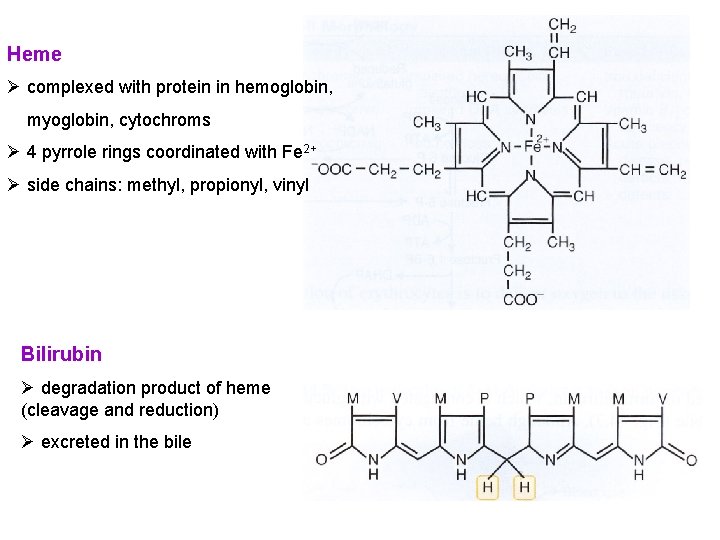
Heme Ø complexed with protein in hemoglobin, myoglobin, cytochroms Ø 4 pyrrole rings coordinated with Fe 2+ Ø side chains: methyl, propionyl, vinyl Bilirubin Ø degradation product of heme (cleavage and reduction) Ø excreted in the bile
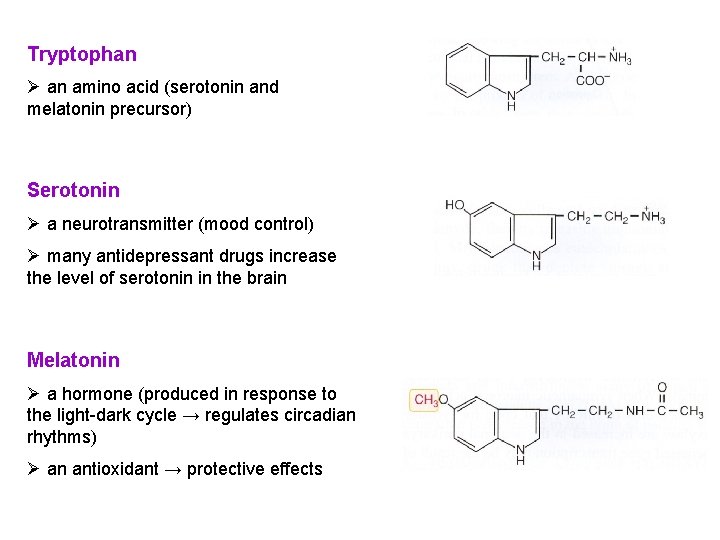
Tryptophan Ø an amino acid (serotonin and melatonin precursor) Serotonin Ø a neurotransmitter (mood control) Ø many antidepressant drugs increase the level of serotonin in the brain Melatonin Ø a hormone (produced in response to the light-dark cycle → regulates circadian rhythms) Ø an antioxidant → protective effects
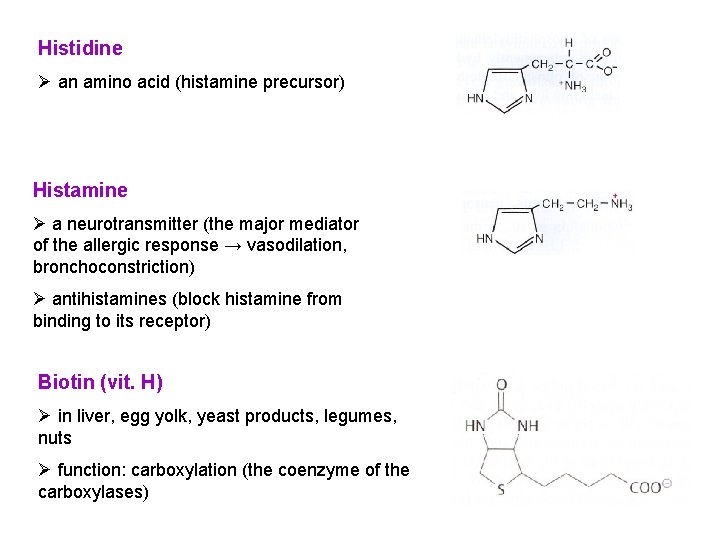
Histidine Ø an amino acid (histamine precursor) Histamine Ø a neurotransmitter (the major mediator of the allergic response → vasodilation, bronchoconstriction) Ø antihistamines (block histamine from binding to its receptor) Biotin (vit. H) Ø in liver, egg yolk, yeast products, legumes, nuts Ø function: carboxylation (the coenzyme of the carboxylases)
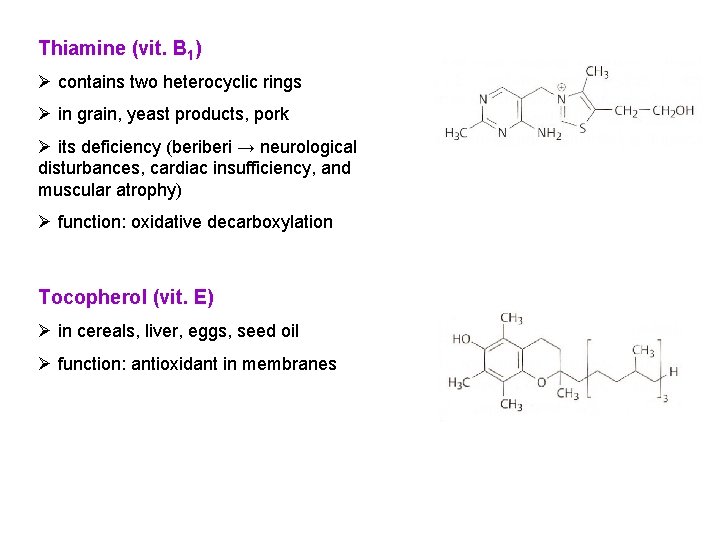
Thiamine (vit. B 1) Ø contains two heterocyclic rings Ø in grain, yeast products, pork Ø its deficiency (beri → neurological disturbances, cardiac insufficiency, and muscular atrophy) Ø function: oxidative decarboxylation Tocopherol (vit. E) Ø in cereals, liver, eggs, seed oil Ø function: antioxidant in membranes
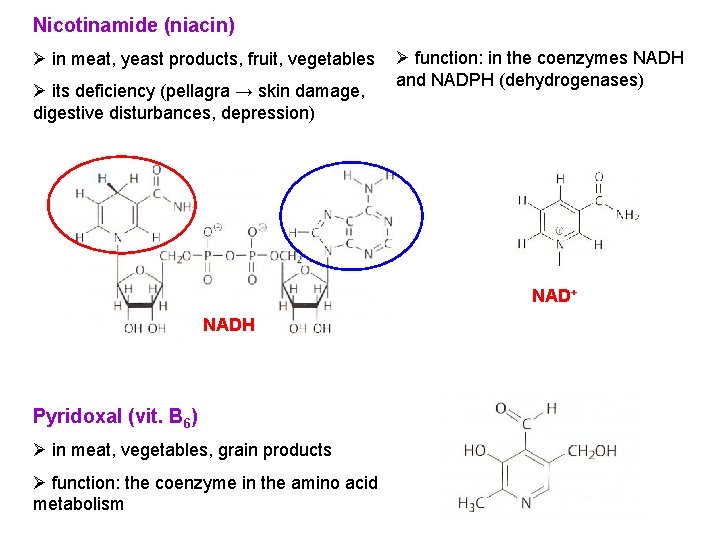
Nicotinamide (niacin) Ø in meat, yeast products, fruit, vegetables Ø its deficiency (pellagra → skin damage, digestive disturbances, depression) Ø function: in the coenzymes NADH and NADPH (dehydrogenases) NAD+ NADH Pyridoxal (vit. B 6) Ø in meat, vegetables, grain products Ø function: the coenzyme in the amino acid metabolism
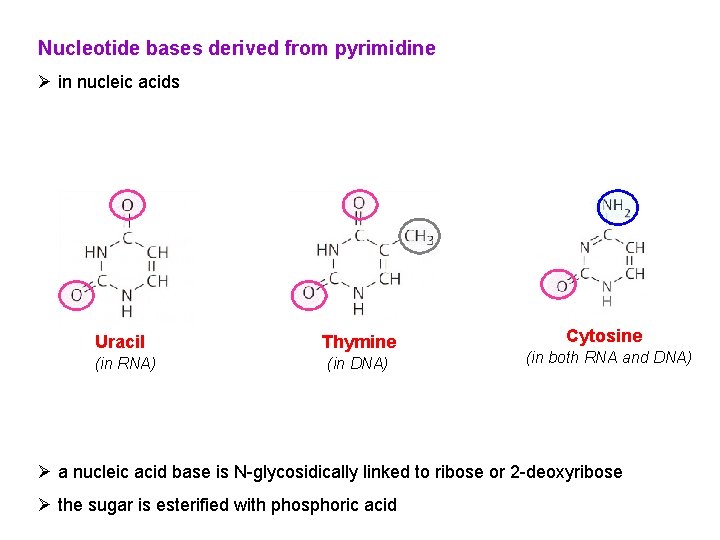
Nucleotide bases derived from pyrimidine Ø in nucleic acids Uracil (in RNA) Thymine (in DNA) Cytosine (in both RNA and DNA) Ø a nucleic acid base is N-glycosidically linked to ribose or 2 -deoxyribose Ø the sugar is esterified with phosphoric acid
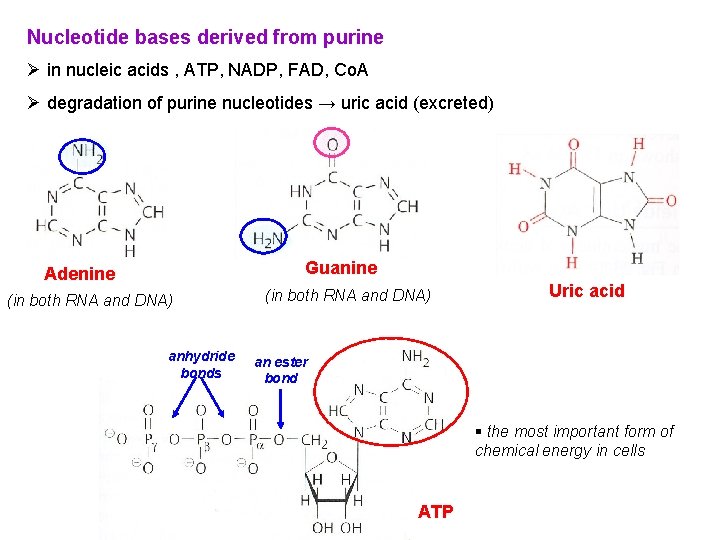
Nucleotide bases derived from purine Ø in nucleic acids , ATP, NADP, FAD, Co. A Ø degradation of purine nucleotides → uric acid (excreted) Guanine Adenine (in both RNA and DNA) anhydride bonds (in both RNA and DNA) Uric acid an ester bond § the most important form of chemical energy in cells ATP
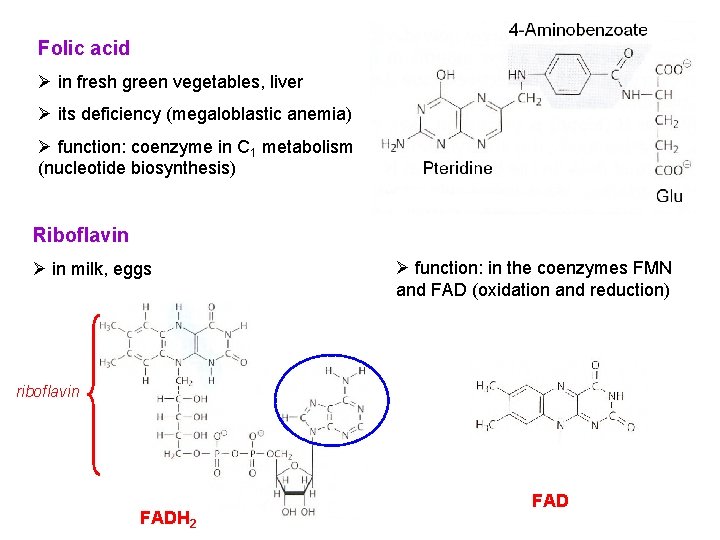
Folic acid Ø in fresh green vegetables, liver Ø its deficiency (megaloblastic anemia) Ø function: coenzyme in C 1 metabolism (nucleotide biosynthesis) Riboflavin Ø in milk, eggs Ø function: in the coenzymes FMN and FAD (oxidation and reduction) riboflavin FADH 2 FAD

Thank you for your attention
- Slides: 70