MOLDX UPDATE Elaine K Jeter MD Mol DX





























- Slides: 36

MOLDX UPDATE Elaine K Jeter, MD

Mol. DX Program • Established to address: • Granular identification • Coding to accommodate to growth of molecular testing • Implement core clinical guidance for coverage • Molecular Diagnostic Testing Policy: • Labs must register their test & obtain unique ID • Applies to all molecular codes – except micro • Test ID is required claim element • New test TA for coverage 11/4/2015 2

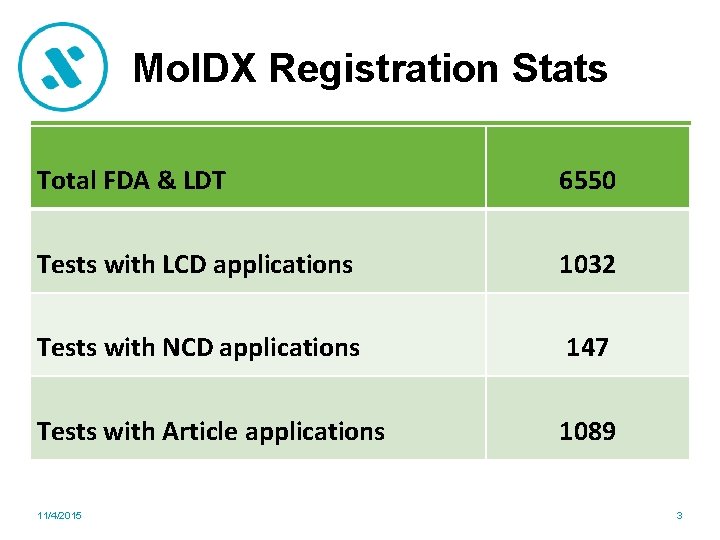
Mol. DX Registration Stats Total FDA & LDT 6550 Tests with LCD applications 1032 Tests with NCD applications 147 Tests with Article applications 1089 11/4/2015 3

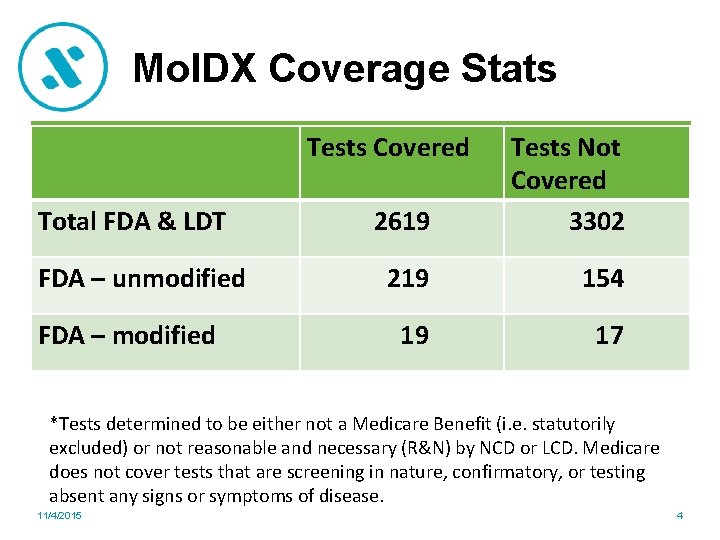
Mol. DX Coverage Stats Tests Covered Total FDA & LDT FDA – unmodified FDA – modified 2619 Tests Not Covered 3302 219 154 19 17 *Tests determined to be either not a Medicare Benefit (i. e. statutorily excluded) or not reasonable and necessary (R&N) by NCD or LCD. Medicare does not cover tests that are screening in nature, confirmatory, or testing absent any signs or symptoms of disease. 11/4/2015 4

Reasons for Non-coverage • No benefit category • • Screening Confirmatory testing when dx is known Quality measure Test doesn’t apply to Medicare population • SSA §(a)(1)(A) – “reasonable & necessary” • Lack of or insufficient clinical utility physician survey • Lack of analytical validation 11/4/2015 5

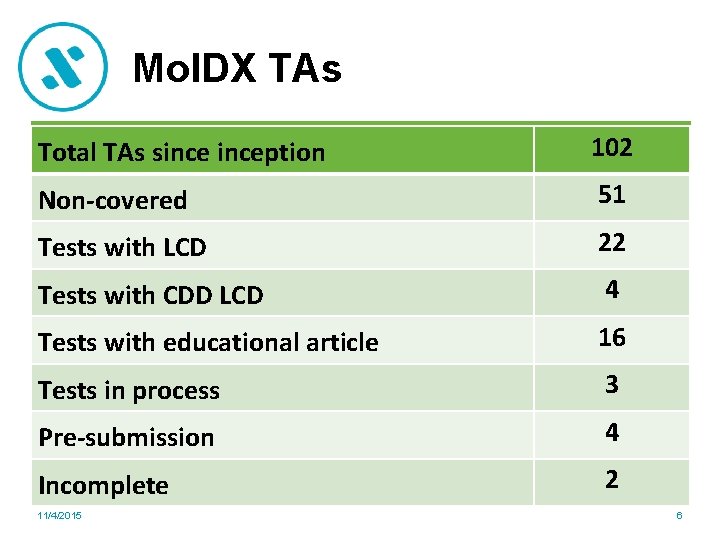
Mol. DX TAs Total TAs sinception 102 Non-covered 51 Tests with LCD 22 Tests with CDD LCD 4 Tests with educational article 16 Tests in process 3 Pre-submission 4 Incomplete 2 11/4/2015 6

Why so Many Policies? • Over-utilization/abuse • CYP testing • Hypercoagulability/Thrombophilia • Controlled Substance Monitoring and DOA Testing • CV Risk Assessment • Special stains and IHC 11/4/2015 7

Prostate CDD Policies • Decipher (Genome. Dx) – to determine which high-risk patients after RP can be closely followed rather than post-op XRT • Confirm. MDx (MDx. Health) – to determine which biopsy negative patients can avoid unnecessary repeat biopsies • Prolaris (Myriad) – to determine which low risk by biopsy prostate cancer patients can be managed by AS • Oncotype Prostate (Genomic Health) – to determine which low risk by biopsy prostate cancer patients can be managed by AS 11/4/2015 8

Mol. DX Roll Out • Operate under a JOA • • JM (SC, NC, VA, WV) JE (CA, NV, HI & Pacific islands) JF (WA, OR, ID, UT, AZ, MT, WY, SD, ND, AK) J 15 (Oh, KY) • Under discussion with other jurisdictions • PAMA & Proposed Rule – silent on nat’l roll out 11/4/2015 9

Hospital Labs • Incorrect info • Mol. DX identifiers • Does not apply to IP or OP labs billing Part A • Applies to all hospital outreach labs that bill Part B • CMS • Current system can’t accommodate narrative field • Developing work-around 11/4/2015 10

Next Generation Sequencing • Technology that analyzes hundreds of genes simultaneously • Two major types • Hot Spot Testing – looks at portions of genes and only specific types of mutations – identify SNVs and dup/dels • Comprehensive Genomic Profiling – looks at entire genes and all known mutations – identify SNVs, dup/dels, CNVs and translocations 11/4/2015 11

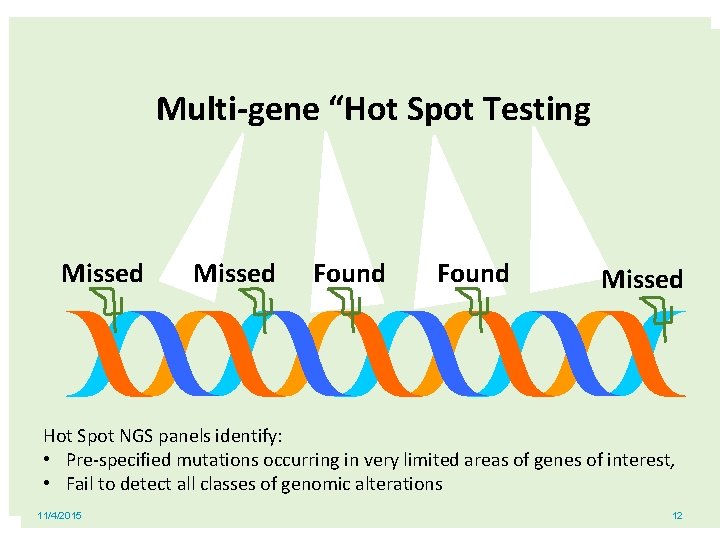
Multi-gene “Hot Spot Testing Missed Found Missed Hot Spot NGS panels identify: • Pre-specified mutations occurring in very limited areas of genes of interest, • Fail to detect all classes of genomic alterations 11/4/2015 12

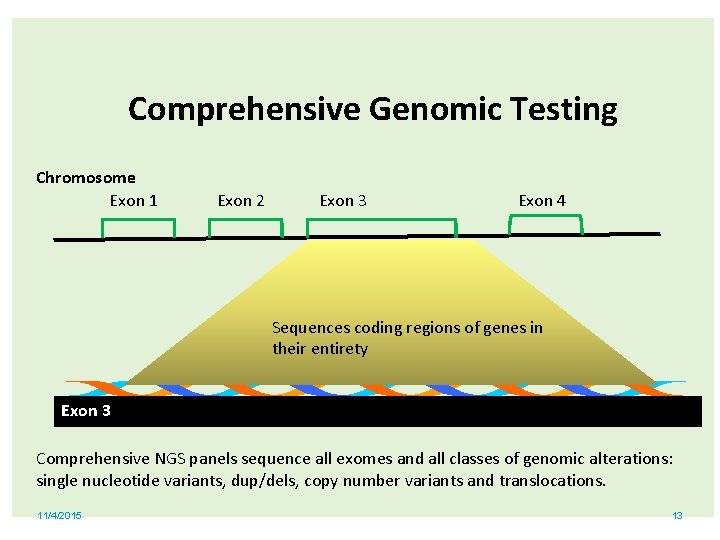
Comprehensive Genomic Testing Chromosome Exon 1 Exon 2 Exon 3 Exon 4 Sequences coding regions of genes in their entirety Exon 3 Comprehensive NGS panels sequence all exomes and all classes of genomic alterations: single nucleotide variants, dup/dels, copy number variants and translocations. 11/4/2015 13

NGS is Here • May replace several individual tests & provide potential healthcare cost savings • Decrease number biopsies • Decrease aggregate cost • Possible decrease ‘shot gun’ use of chemotherapy • May possibly improve patient outcomes • Identify patients for targeted therapy missed by Companion Dx or LDT • Identify patients for clinical trials & investigational therapies 11/4/2015 14

NGS High Points • No standardization or cross validation of platforms • Generally more sensitive than companion dx methodologies – unclear if better outcomes • Less tissue required for analysis • Pressure to cover this technology • Academic centers • Industry – vocal high profile companies & political ears 11/4/2015 15

Non-standardized Testing • Three components • Pre-analytical • Sequencing • Data interpretation using complex algorithms • No comparison between labs or platforms, and disagree on methods to determine components • 80% concordance – simplest DNA alterations • 20% concordance – complicated alterations 11/4/2015 16

Low Allele Frequency • NGS – high assay sensitivity & tumor heterogeneity • Detect alterations in very small number of cells • Report positive alteration – directly guide tx • 100% therapeutic response to cells, but the vast majority of tumor remains untreated and the patient experiences a poor outcome 11/4/2015 17

False Results • High false positives • ~50% of reported alterations are erroneous • Driver mutations vs passenger mutations • Many patients will receive tx they will not respond to • Patients will be shunted from other treatments they could have received while chasing the erroneous result • False negatives • Most “hotspot” labs must perform FISH testing to rule out false negatives 11/4/2015 18

Lack Data Collection • NGS panels place patients in harms way by allowing patients to: • Receive testing & tx that compete with existing trials • Receive ineffective treatments • Miss publication of key toxicities that lead to better understanding of disease • Potentially hamper, rather than advance science, by use of off-label drugs without a body of literature 11/4/2015 19

Solutions to NGS Pitfalls • Standardize Testing • CAP/AMP developing standards • FDA assessment of LDTs • Compare testing with existing literature established with targeted therapy – does it improve outcomes • Attach high quality testing as inclusion criteria in current and future clinical trials • Collect outcomes and aggregate nationally 11/4/2015 20

Example of Problem • NSCLC NCCN Guidelines – Version 7. 2015 • ALK and EGFR – adequate literature for coverage • Adenocarcinoma – test all patients • Squamous histology – if never smokers, small bx used in testing, or mixed histology • ROS 1 – adequate literature for coverage after NCCN review 11/4/2015 21

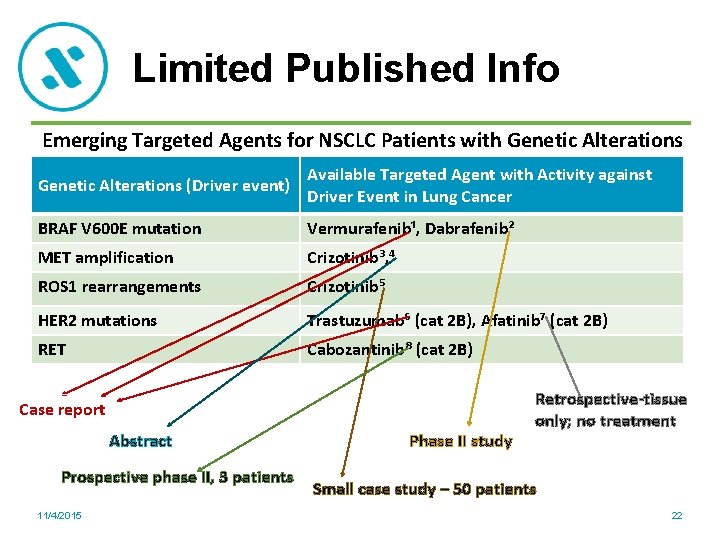
Limited Published Info Emerging Targeted Agents for NSCLC Patients with Genetic Alterations (Driver event) Available Targeted Agent with Activity against Driver Event in Lung Cancer BRAF V 600 E mutation Vermurafenib¹, Dabrafenib² MET amplification Crizotinib³, ⁴ ROS 1 rearrangements Crizotinib⁵ HER 2 mutations Trastuzumab⁶ (cat 2 B), Afatinib⁷ (cat 2 B) RET Cabozantinib⁸ (cat 2 B) Case report Abstract Prospective phase II, 3 patients 11/4/2015 Phase II study Retrospective-tissue only; no treatment Small case study – 50 patients 22

Emerging Targeted Agents • Evidence is inadequate – case reports, abstracts, small trials • Multiple ongoing trials waiting final reporting on these agents • Insufficient evidence to allow wholesale coverage • Unclear where to include these other agents in biologic pathways 11/4/2015 23

Coverage & Coding Issues • Genes in NGS panels must meet R&N determination • AMA acknowledges error to “unbundle” – currently correcting their error 11/4/2015 24

NGS in Mol. DX • NGS panel must submit new DEX registration • End date prior test registrations • Must include genes in NGS panel • Coverage remains the same – based on R&N • Reimbursement will change • Working with AMA 11/4/2015 25

ICD-10 • Unspecified codes were intentionally removed from 9 to 10 crosswalk – specificity is the purpose of ICD-10 • LCDs back to CAC to remove ICD-10 s that were erroneously included • Request addition of specific ICD-10 s where needed with reasoning – add as needed 11/4/2015 26

CSM & DOA Policy Goals • For pain & substance use disorder patients: • Define appropriate indications • Testing frequency for prescribed substances • Define documentation requirements • Tests ordered & medical indication for specific test selection • Provide education-testing methods & limitations 11/4/2015 27

11/4/2015 28

11/4/2015 29

11/4/2015 30

11/4/2015 31

Drug Testing Claims Data • Utilization spikes • Physician owned labs (POLs) – self-referral • Billing semi-quant testing with Quantitative codes • Using methodology codes (83542 – LC-MS) to bill for opioid metabolites; 83925 – opiate(s), drug & metabolites • Billing schemes to induce referrals • Labs charges physician below market flat fee, physician marks up to private payers 11/4/2015 32

Testing for Substance Use 11/4/2015 33

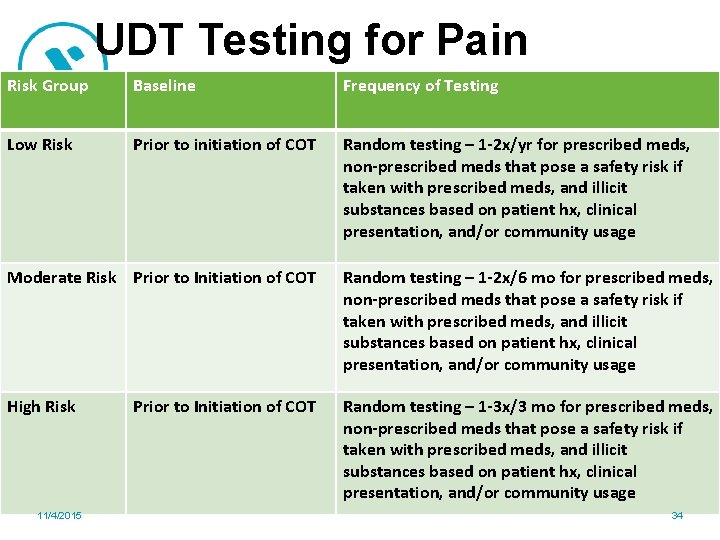
UDT Testing for Pain Risk Group Baseline Frequency of Testing Low Risk Prior to initiation of COT Random testing – 1 -2 x/yr for prescribed meds, non-prescribed meds that pose a safety risk if taken with prescribed meds, and illicit substances based on patient hx, clinical presentation, and/or community usage Moderate Risk Prior to Initiation of COT Random testing – 1 -2 x/6 mo for prescribed meds, non-prescribed meds that pose a safety risk if taken with prescribed meds, and illicit substances based on patient hx, clinical presentation, and/or community usage High Risk Random testing – 1 -3 x/3 mo for prescribed meds, non-prescribed meds that pose a safety risk if taken with prescribed meds, and illicit substances based on patient hx, clinical presentation, and/or community usage 11/4/2015 Prior to Initiation of COT 34

UDT Coding/Billing Claim • One drug per claim line • Each drug must use short string text in SV 101 -7 field of claim • Up to 7 drugs reimbursed per CPT • Tiered panel reimbursement: • 8 -15 • 16 -34 • >35 11/4/2015 35

Questions? Elaine. jeter@palmettogba. com 11/4/2015 36