Enoxaparin in primary PCI From FINESSE to ATOLL





- Slides: 18

Enoxaparin in primary PCI From FINESSE to ATOLL G. Montalescot Institut de Cardiologie Pitié-Salpêtrière Hospital Paris, France The FINESSE Trial is supported by Eli Lilly and Co and Centocor. G. Montalescot, disclosure: Institutional research grant, consulting and speaker fees from Daiichi Sankyo, Eli Lilly, Sanofi Aventis, BMS.

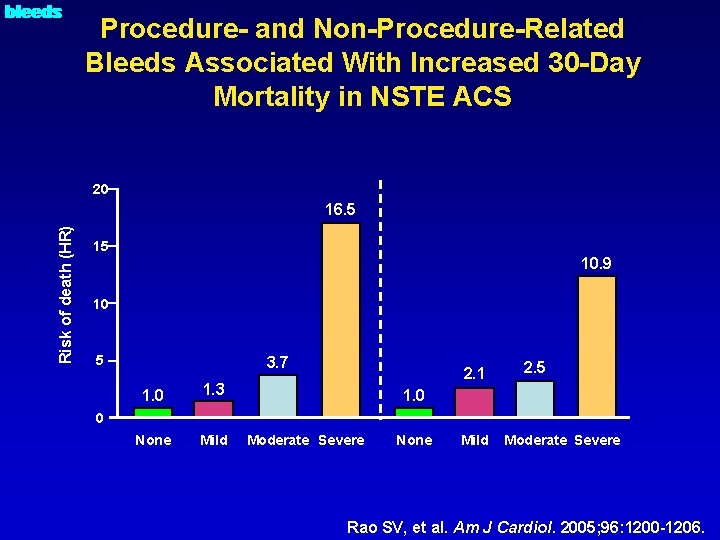
O bleeds Procedure- and Non-Procedure-Related Bleeds Associated With Increased 30 -Day Mortality in NSTE ACS 20 Risk of death (HR) 16. 5 15 10. 9 10 5 3. 7 1. 0 1. 3 None Mild 2. 1 2. 5 1. 0 0 Moderate Severe None Mild Moderate Severe Rao SV, et al. Am J Cardiol. 2005; 96: 1200 -1206.

Can we improve safety in PCI with enoxaparin?

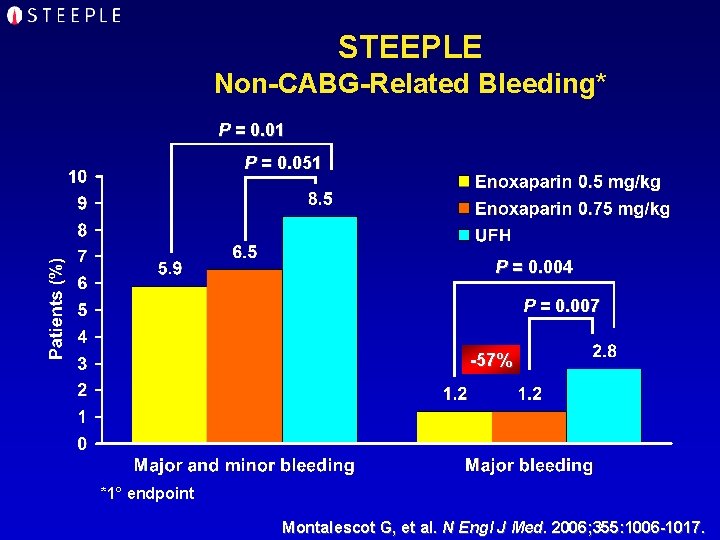
STEEPLE Non-CABG-Related Bleeding* P = 0. 01 P = 0. 051 P = 0. 004 P = 0. 007 -57% *1° endpoint Montalescot G, et al. N Engl J Med. 2006; 355: 1006 -1017.

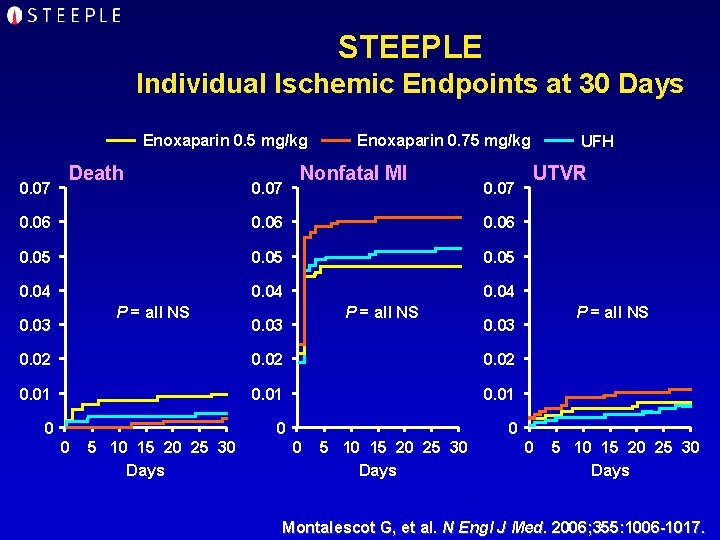
STEEPLE Individual Ischemic Endpoints at 30 Days Enoxaparin 0. 5 mg/kg 0. 07 Death 0. 07 Enoxaparin 0. 75 mg/kg Nonfatal MI 0. 07 0. 06 0. 05 0. 04 P = all NS 0. 03 0. 02 0. 01 0 0 0 5 10 15 20 25 30 Days UTVR P = all NS 0. 03 0. 02 0 UFH 0 5 10 15 20 25 30 Days Montalescot G, et al. N Engl J Med. 2006; 355: 1006 -1017.

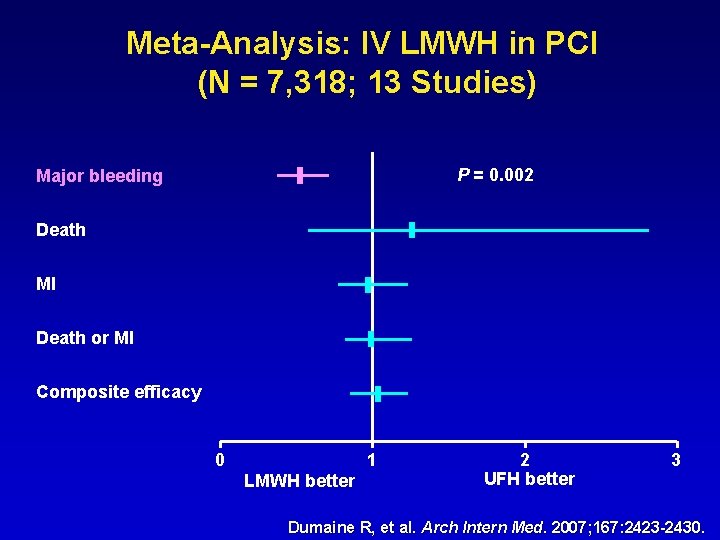
Meta-Analysis: IV LMWH in PCI (N = 7, 318; 13 Studies) P = 0. 002 Major bleeding Death MI Death or MI Composite efficacy 0 1 LMWH better 2 UFH better 3 Dumaine R, et al. Arch Intern Med. 2007; 167: 2423 -2430.

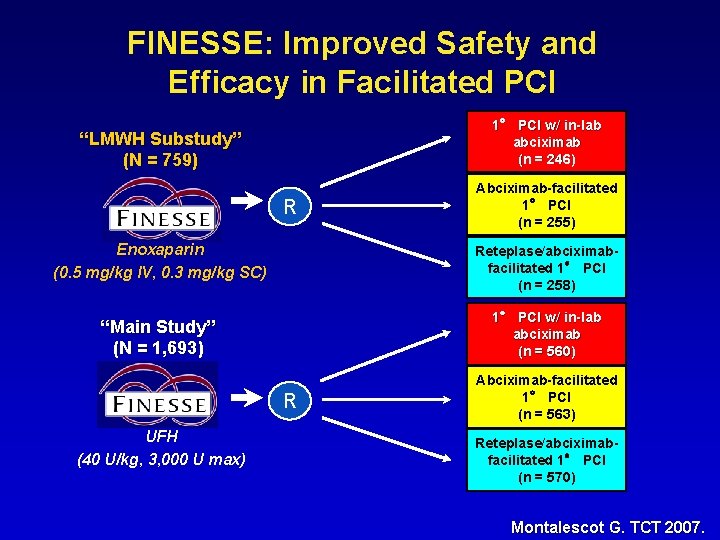
FINESSE: Improved Safety and Efficacy in Facilitated PCI 1° PCI w/ in-lab abciximab (n = 246) “LMWH Substudy” (N = 759) R Abciximab-facilitated 1° PCI (n = 255) Enoxaparin (0. 5 mg/kg IV, 0. 3 mg/kg SC) Reteplase/abciximabfacilitated 1° PCI (n = 258) “Main Study” (N = 1, 693) 1° PCI w/ in-lab abciximab (n = 560) R UFH (40 U/kg, 3, 000 U max) Abciximab-facilitated 1° PCI (n = 563) Reteplase/abciximabfacilitated 1° PCI (n = 570) Montalescot G. TCT 2007.

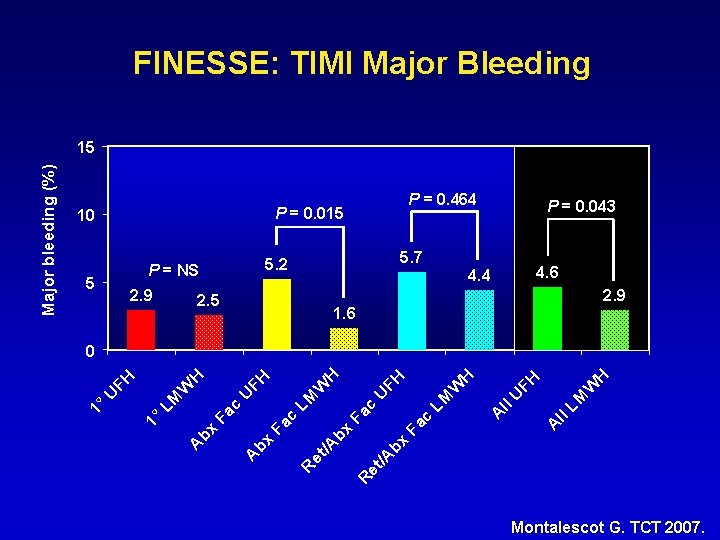
FINESSE: TIMI Major Bleeding P = 0. 464 P = 0. 015 10 5 2. 9 5. 7 5. 2 P = NS 2. 5 P = 0. 043 4. 6 4. 4 2. 9 1. 6 H W LM ll A A ll U FH H W c Fa bx bx /A /A R et et R LM U c Fa bx A FH H LM c Fa bx A W FH U W LM 1° U FH H 0 1° Major bleeding (%) 15 Montalescot G. TCT 2007.

Can we improve safety and efficacy in primary PCI?

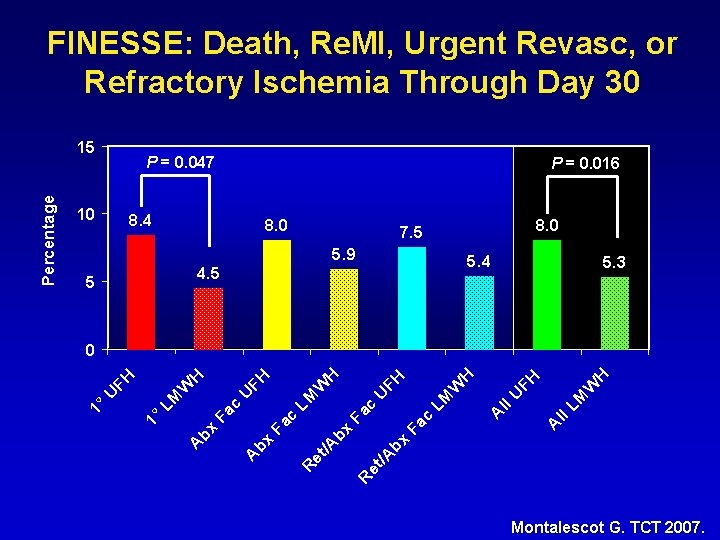
FINESSE: Death, Re. MI, Urgent Revasc, or Refractory Ischemia Through Day 30 P = 0. 047 10 P = 0. 016 8. 4 8. 0 7. 5 5. 9 5. 4 4. 5 5 5. 3 H W LM ll A A ll U FH H W c Fa bx bx /A /A R et et R LM U c Fa bx A FH H LM c Fa bx A W FH U W LM 1° U FH H 0 1° Percentage 15 Montalescot G. TCT 2007.

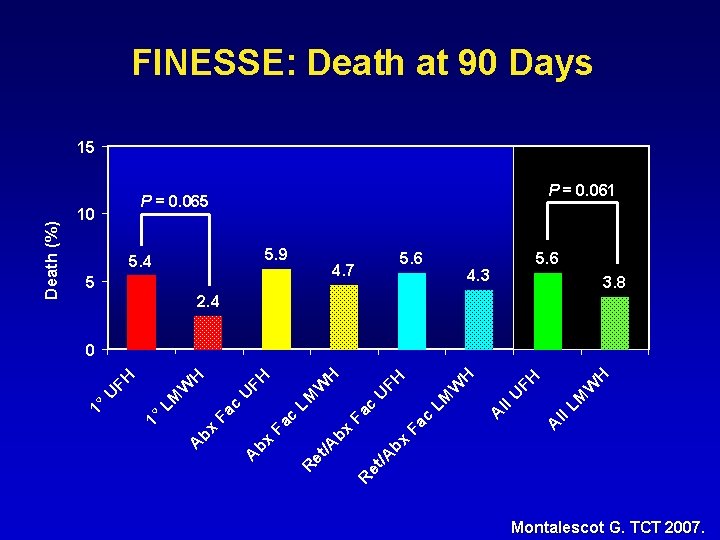
FINESSE: Death at 90 Days P = 0. 061 P = 0. 065 10 5. 9 5. 4 5 5. 6 4. 7 5. 6 4. 3 3. 8 2. 4 H W LM ll A A ll U FH H W c Fa bx bx /A /A R et et R LM U c Fa bx A FH H LM c Fa bx A W FH U W LM 1° U FH H 0 1° Death (%) 15 Montalescot G. TCT 2007.

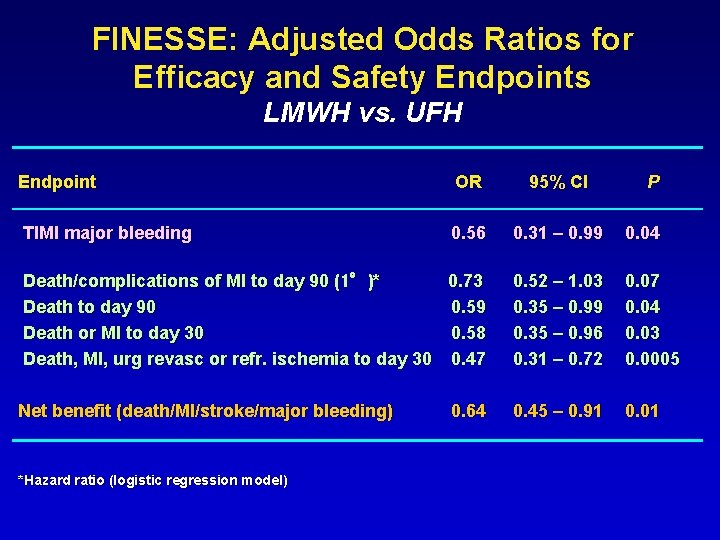
FINESSE: Adjusted Odds Ratios for Efficacy and Safety Endpoints LMWH vs. UFH Endpoint OR 95% CI TIMI major bleeding 0. 56 0. 31 – 0. 99 0. 04 Death/complications of MI to day 90 (1°)* 0. 73 Death to day 90 0. 59 Death or MI to day 30 0. 58 Death, MI, urg revasc or refr. ischemia to day 30 0. 47 0. 52 – 1. 03 0. 35 – 0. 99 0. 35 – 0. 96 0. 31 – 0. 72 0. 07 0. 04 0. 03 0. 0005 Net benefit (death/MI/stroke/major bleeding) 0. 45 – 0. 91 0. 01 *Hazard ratio (logistic regression model) 0. 64 P

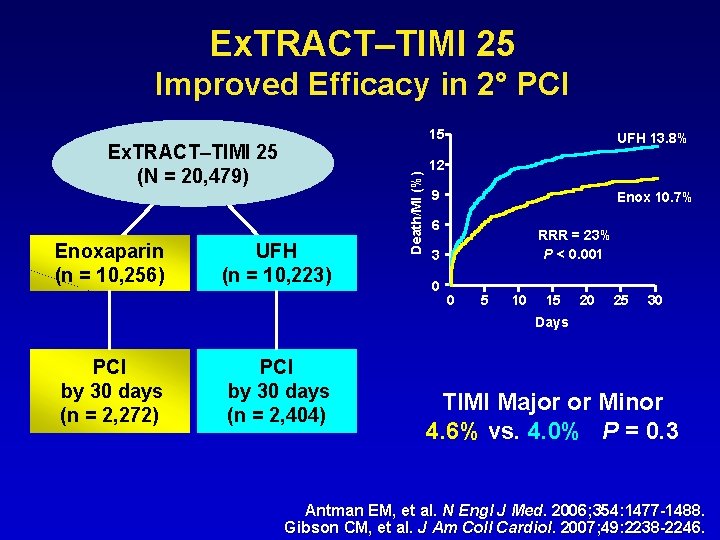
Ex. TRACT–TIMI 25 Improved Efficacy in 2° PCI Ex. TRACT–TIMI 25 (N = 20, 479) Enoxaparin (n = 10, 256) UFH (n = 10, 223) Death/MI (%) 15 UFH 13. 8% 12 9 Enox 10. 7% 6 RRR = 23% P < 0. 001 3 0 0 5 10 15 20 25 30 Days PCI by 30 days (n = 2, 272) PCI by 30 days (n = 2, 404) TIMI Major or Minor 4. 6% vs. 4. 0% P = 0. 3 Antman EM, et al. N Engl J Med. 2006; 354: 1477 -1488. Gibson CM, et al. J Am Coll Cardiol. 2007; 49: 2238 -2246.

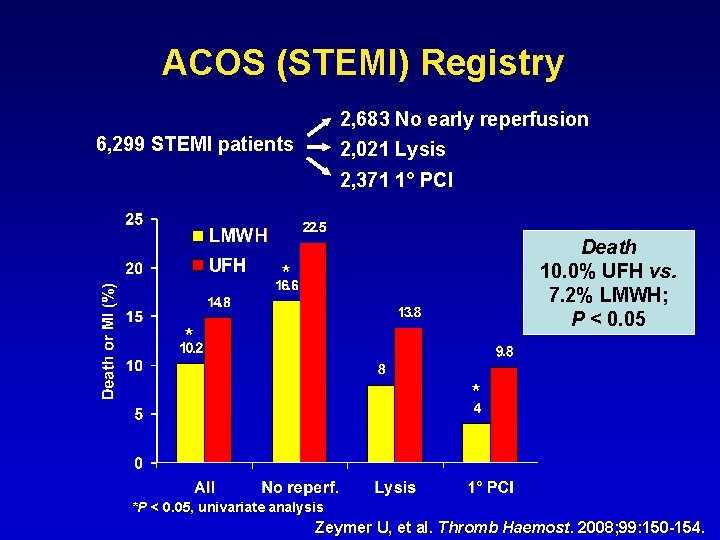
ACOS (STEMI) Registry 2, 683 No early reperfusion 6, 299 STEMI patients 2, 021 Lysis 2, 371 1° PCI Death 10. 0% UFH vs. 7. 2% LMWH; P < 0. 05 * *P < 0. 05, univariate analysis Zeymer U, et al. Thromb Haemost. 2008; 99: 150 -154.

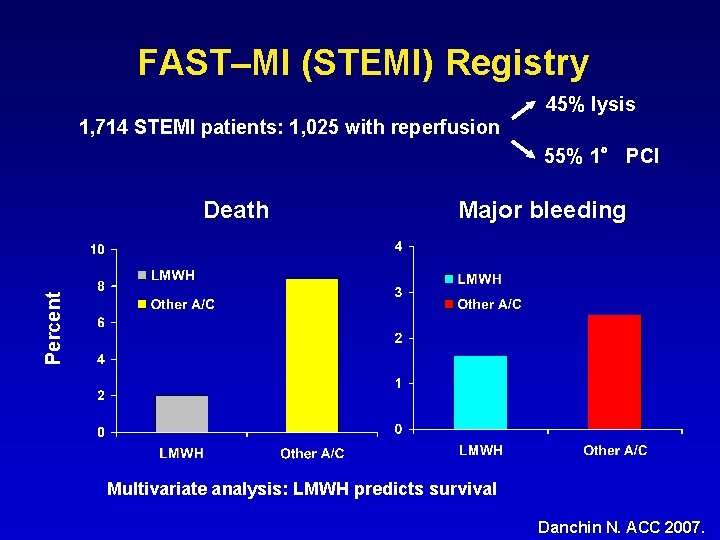
FAST–MI (STEMI) Registry 45% lysis 1, 714 STEMI patients: 1, 025 with reperfusion 55% 1° PCI Major bleeding Percent Death Multivariate analysis: LMWH predicts survival Danchin N. ACC 2007.

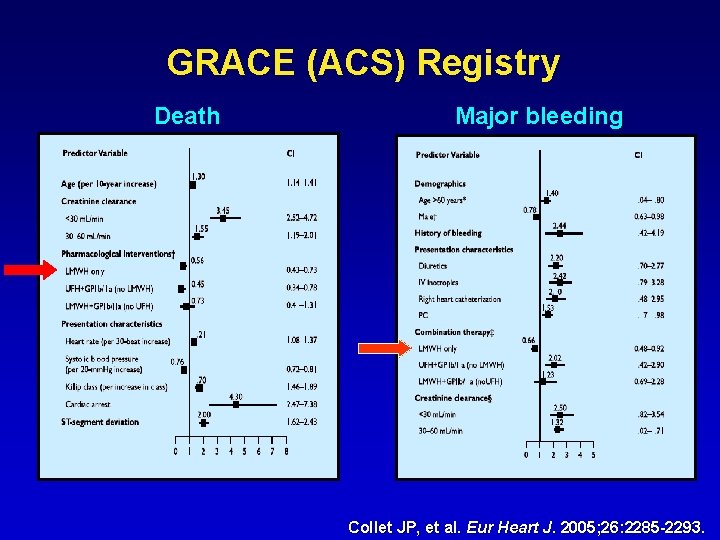
GRACE (ACS) Registry Death Major bleeding Collet JP, et al. Eur Heart J. 2005; 26: 2285 -2293.

ATOLL: Study Design STEMI planned for 1° PCI Randomization (N = 850) ENOXAPARIN IV 0. 5 mg/kg (± GP IIb/IIIa inhibitor) UFH IV 50 – 70 IU/kg if GP IIb/IIIa 70 – 100 IU/kg if no GP IIb/IIIa Dose adjusted to ACT 1° PCI and stenting 1° EP: Death, complication of MI, procedure failure or non-CABG major bleeding at 30 days Main 2° EP: Death, MI, refractory ischemia, urgent revasc. 6 -month follow-up Patients who have already received UFH or LMWH or any other anticoagulant are excluded. All concomitant drugs accepted, except lytics; cross-over to other anticoagulant NOT accepted.

Conclusions • Time has come for a change of anticoagulation in p. PCI • Favorable data with IV enoxaparin in expeditive care of ACS, elective PCI, p. PCI • Same dose regimen of enoxaparin whatever the antiplatelet regimen • Optimal cost: benefit ratio