Energy Pooling and Ionization in dense Alkali Metal








- Slides: 20

Energy Pooling and Ionization in dense Alkali Metal Vapor Sean Bresler, Joonbum Park, Michael Heaven ISMS 2017

Motivation • 1: Knize, R. J. , Zhdanov, B. V. , & Shaffer, M. K. (2011). Photoionization in alkali lasers. Optics Express, 19(8), 7894. https: //doi. org/10. 1364/OE. 19. 007894

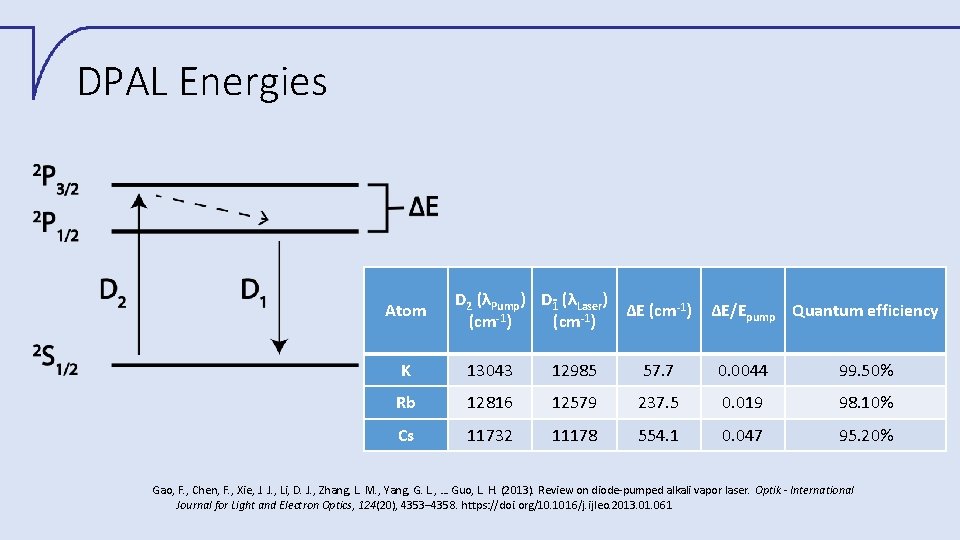
DPAL Energies Atom D 2 (λPump) D 1 (λLaser) ΔE (cm 1) 1 1 (cm ) ΔE/Epump Quantum efficiency K 13043 12985 57. 7 0. 0044 99. 50% Rb 12816 12579 237. 5 0. 019 98. 10% Cs 11732 11178 554. 1 0. 047 95. 20% Gao, F. , Chen, F. , Xie, J. J. , Li, D. J. , Zhang, L. M. , Yang, G. L. , … Guo, L. H. (2013). Review on diode-pumped alkali vapor laser. Optik - International Journal for Light and Electron Optics, 124(20), 4353– 4358. https: //doi. org/10. 1016/j. ijleo. 2013. 01. 061

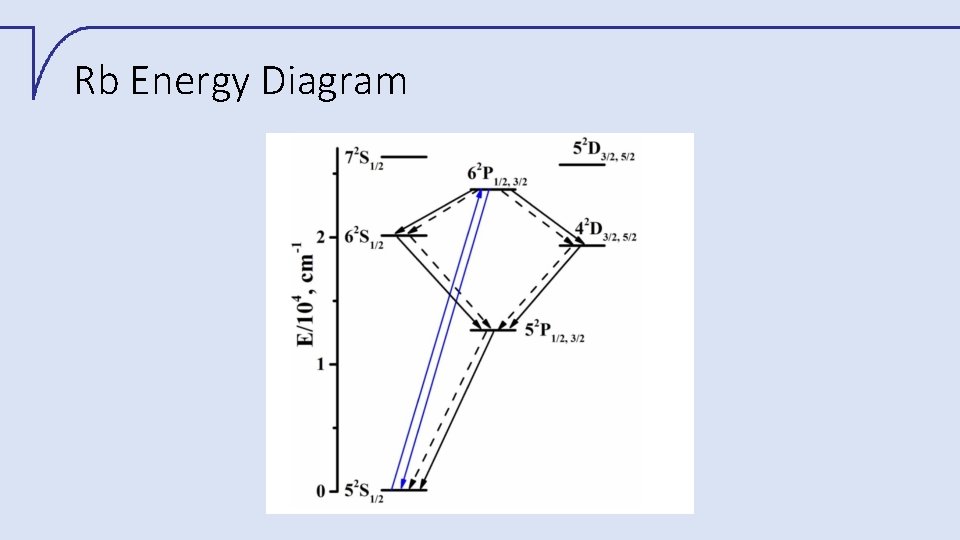
Rb Energy Diagram

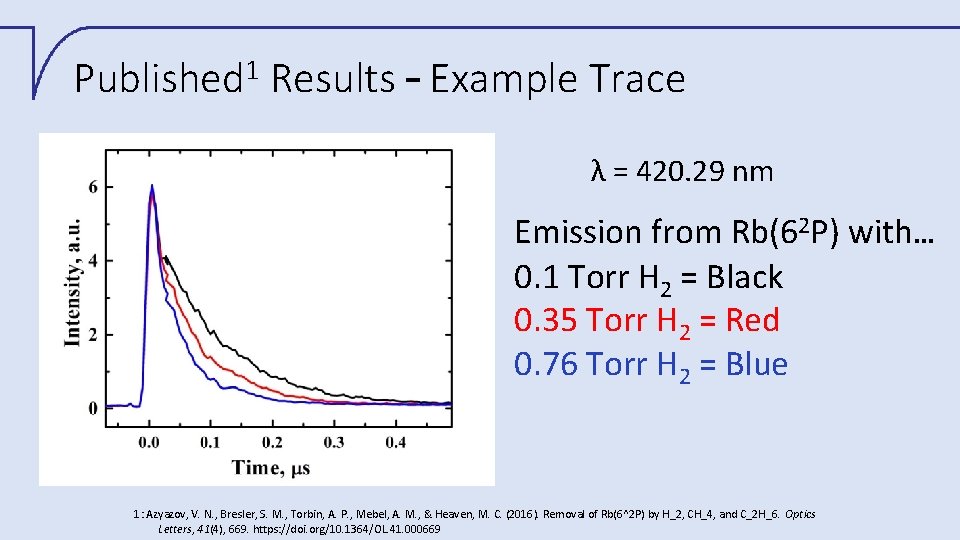
1 Published Results – Example Trace λ = 420. 29 nm Emission from Rb(62 P) with… 0. 1 Torr H 2 = Black 0. 35 Torr H 2 = Red 0. 76 Torr H 2 = Blue 1: Azyazov, V. N. , Bresler, S. M. , Torbin, A. P. , Mebel, A. M. , & Heaven, M. C. (2016). Removal of Rb(6^2 P) by H_2, CH_4, and C_2 H_6. Optics Letters, 41(4), 669. https: //doi. org/10. 1364/OL. 41. 000669

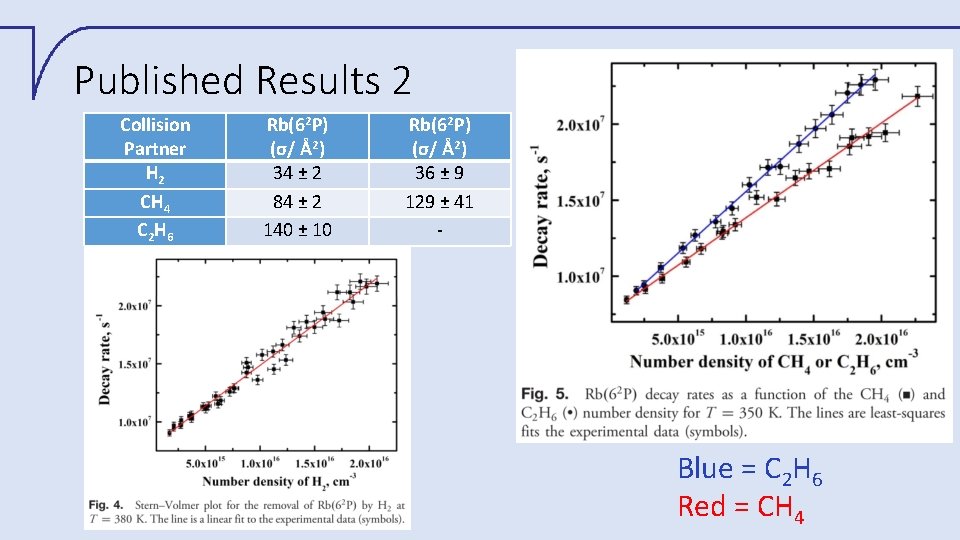
Published Results 2 Collision Partner H 2 CH 4 C 2 H 6 Rb(62 P) (σ/ Å2) 34 ± 2 84 ± 2 140 ± 10 Rb(62 P) (σ/ Å2) 36 ± 9 129 ± 41 - Blue = C 2 H 6 Red = CH 4

Conclusions for Rb Experiment • Rb. H only detected for Rb(62 P) + H 2 • Rb(62 P) removal rates: • k. H 2 = (7. 0 ± 0. 2) x 10 -10 cm 3 s-1 for T = 380 K • k. CH 4 = (6. 2 ± 0. 2) x 10 -10 cm 3 s-1 for T = 350 K • k. C 2 H 6 = (8. 1 ± 0. 3) x 10 -10 cm 3 s-1 for T = 350 K • Deactivation is mostly a physical channel with a possible small chemical pathway. • Began investigating higher excited states; this was abandoned… or so we thought.

Apparatus (Re)selection Static Cell • Temperature easily Controlled • Sample lasts longer • Net flow is zero • Pressure is easily controlled • High number density – optical trapping Flow Cell • Net flow is controllable • Fresh sample every shot • Minimal Trapping • Pressure fluctuates • Temperature less well defined • High metal/gas consumption

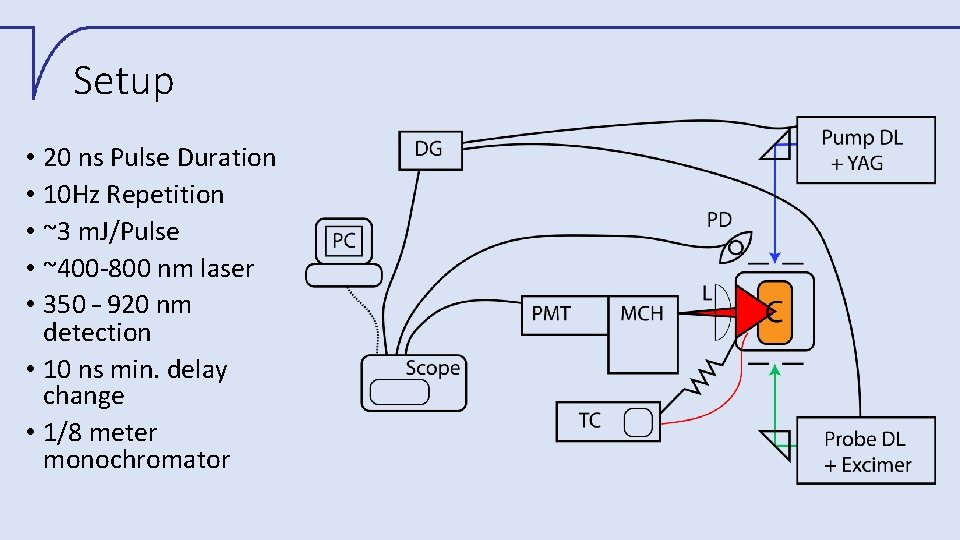
Setup • 20 ns Pulse Duration • 10 Hz Repetition • ~3 m. J/Pulse • ~400 -800 nm laser • 350 – 920 nm detection • 10 ns min. delay change • 1/8 meter monochromator

Overview • Cs, possibly mixed with He, H 2, CH 4, or C 2 H 6 • Single color/photon experiment • Two color experiment – Cs pump, Cs. H probe • Depletion measurements • Light may also be dispersed for state-to-state kinetics. • Observed signals are largely from saturated excitation (interaction volume driven to transparency) • All signals are time-resolved. • Where is the energy going, why are we getting snow, why can’t we see Cs. H with Methane in the flow cell?


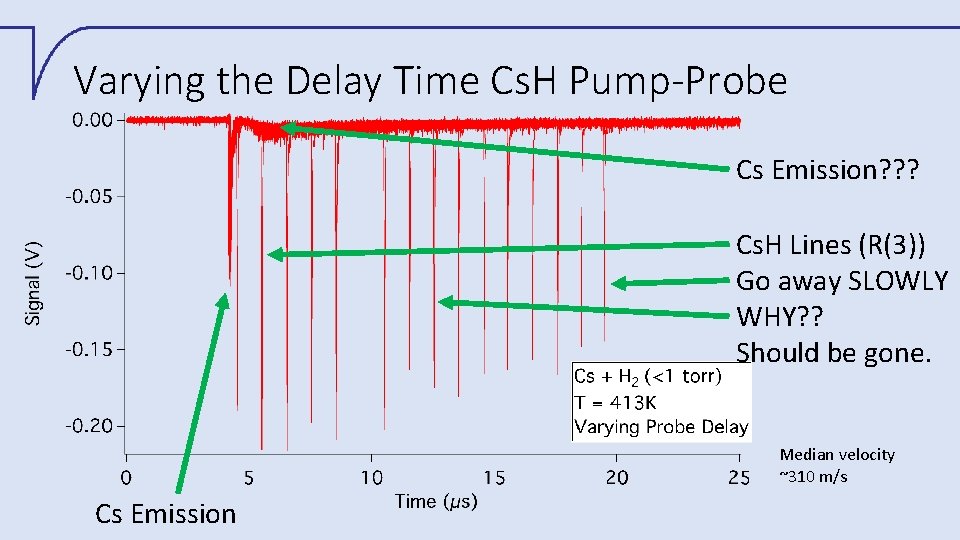
Varying the Delay Time Cs. H Pump-Probe Cs Emission? ? ? Cs. H Lines (R(3)) Go away SLOWLY WHY? ? Should be gone. Median velocity ~310 m/s Cs Emission

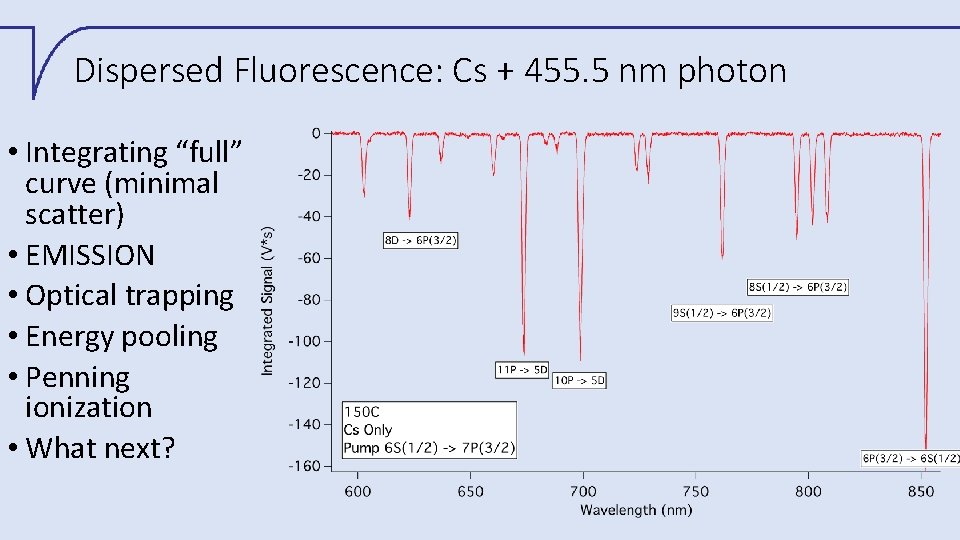
Dispersed Fluorescence: Cs + 455. 5 nm photon • Integrating “full” curve (minimal scatter) • EMISSION • Optical trapping • Energy pooling • Penning ionization • What next?

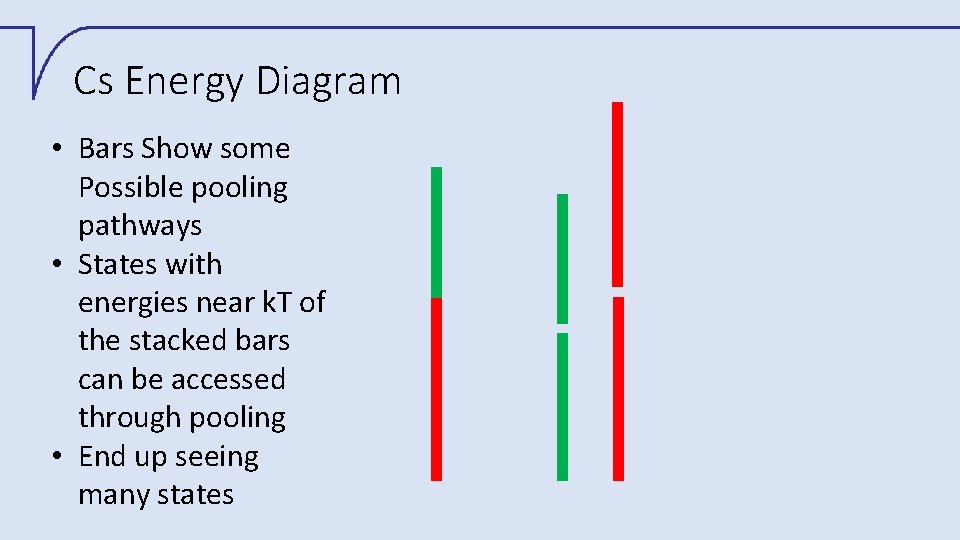
Cs Energy Diagram • Bars Show some Possible pooling pathways • States with energies near k. T of the stacked bars can be accessed through pooling • End up seeing many states

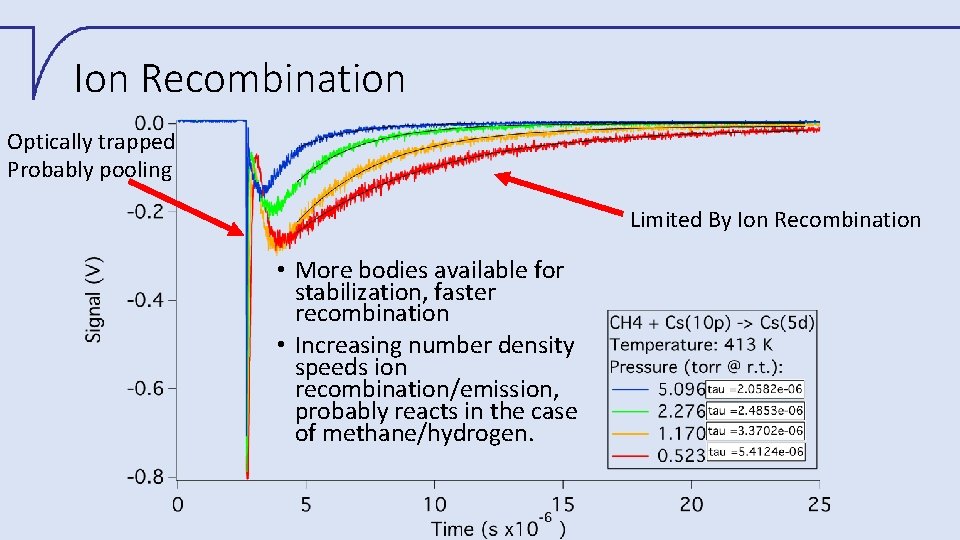
Ion Recombination Optically trapped Probably pooling Limited By Ion Recombination • More bodies available for stabilization, faster recombination • Increasing number density speeds ion recombination/emission, probably reacts in the case of methane/hydrogen.

1. 2. 3. 4. 5. 6. • Collisions also assist in spin orbit mixing Suggested Mechanism • 3+ body collisions necessary to stabilize 2 2 Cs + 2 hν → 2 Cs(7 P 3/2) ion recombination. Cs(72 P 3/2) + Cs(72 P 3/2) → Cs+ + Cs(62 S 1/2) • Time between steps 1 Cs+ + Cs → Cs 2+ and 2 allows for radiative relaxation to Cs 2+ + e- → Cs 2* lower states, so step 2 * * 2 Cs 2 → Cs + Cs(6 S 1/2) need not be specifically Cs* → → Cs(62 S 1/2) + nhν Cs(72 P 3/2) • Intermediate states collide to make higher neutral states

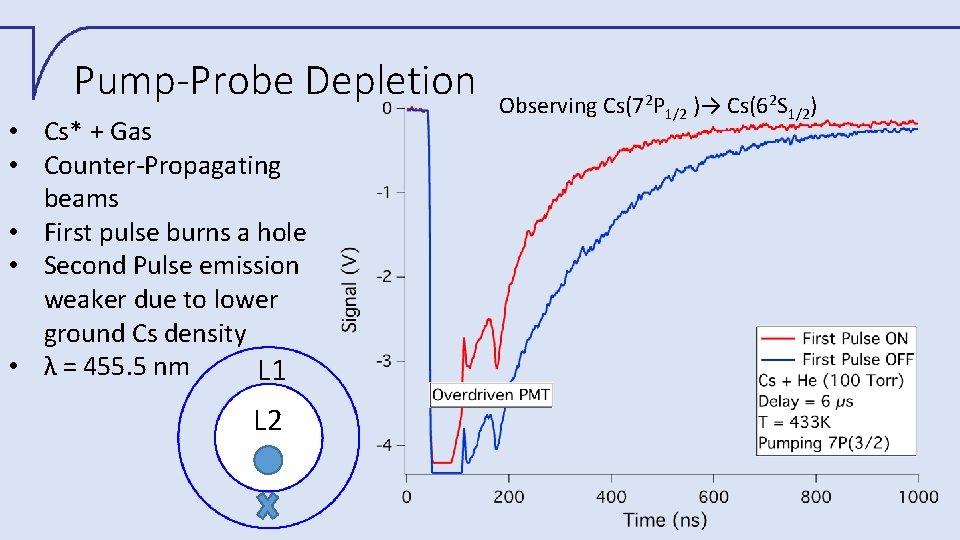
Pump-Probe Depletion • Cs* + Gas • Counter-Propagating beams • First pulse burns a hole • Second Pulse emission weaker due to lower ground Cs density • λ = 455. 5 nm L 1 L 2 Observing Cs(72 P 1/2 )→ Cs(62 S 1/2)

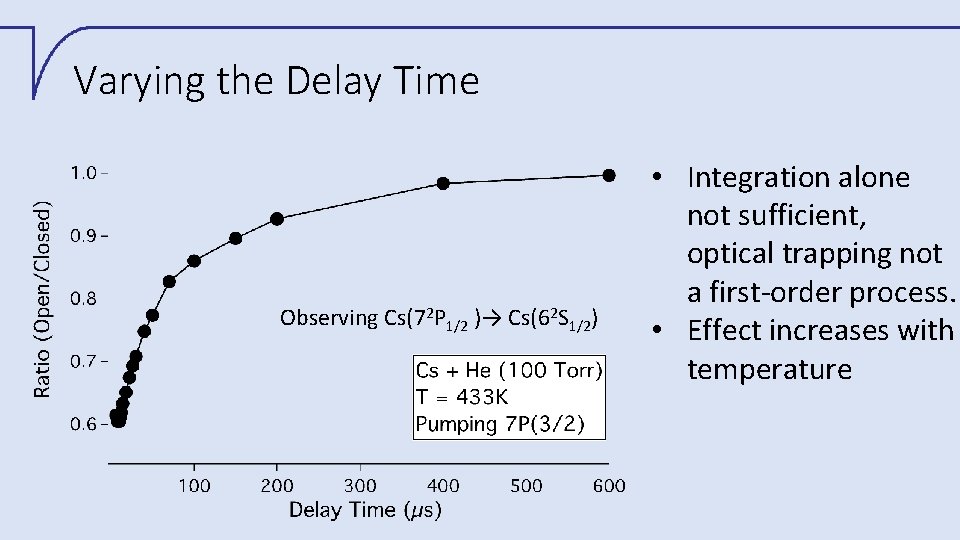
Varying the Delay Time Observing Cs(72 P 1/2 )→ Cs(62 S 1/2) • Integration alone not sufficient, optical trapping not a first-order process. • Effect increases with temperature

Cs Conclusions and Future work • Rb(62 P) removal in the presence of H 2, CH 4, C 2 H 6 has been quantified • Energy Pooling is extremely efficient in the Cs Case, and is an easy way to make Cs+ Ions. REMPI type schemes or UV photodeatchment has been attempted, doesn’t work. • Cs* DOES react with H 2, CH 4 to form Cs. H. Cannot Rule out the possibility that Cs(72 P 3/2) is NOT the primary reactive partner with CH 4 • Goal is to get branching ratios. Complicated, need model. working on differential equations. Want repeatability. • Cs* + C 2 H 6 will be determined soon (ethane shortage)

Thanks • • • Michael C. Heaven, Ph. D (PI) Joonbum Park, Ph. D (partner) Jiande Han, Ph. D Daniel Frohman, Ph. D Kyle Mascaritolo, Ph. D (gone) Joshua Bartlett, Ph. D (gone) Michael Sullivan, Ph. D (going) Robert Van. Gundy Amanda Dermer Mallory Theis Jessica Cifuentes (undergrad) • Wafaa Fawzy, Ph. D (Visiting Prof. Murray State) • Jacob Stewart, Ph. D (taught me how to use an oscilloscope) • Ian Finneran, Ph. D (Inspired me on the bus back last year, Cal. Tech) • Joint Technology Office; Air Force Office of Scientific Research (AFOSR) (FA 9550 -13 -1 -0002)