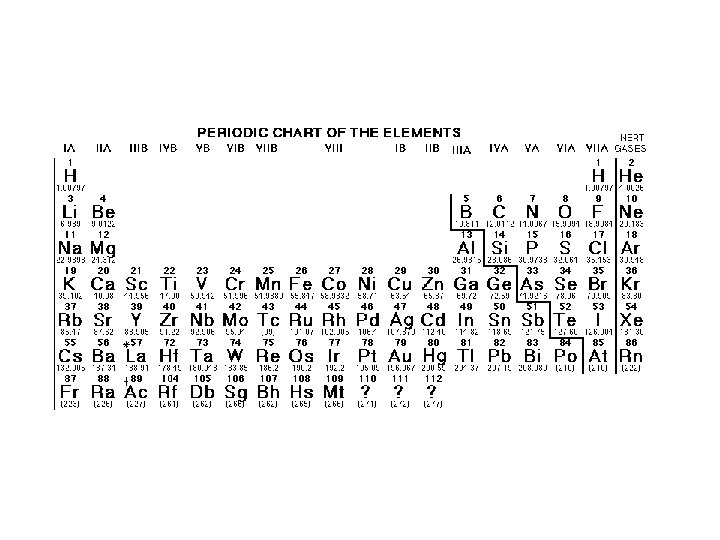
Elements Compounds and Mixtures Atom Basic unit of

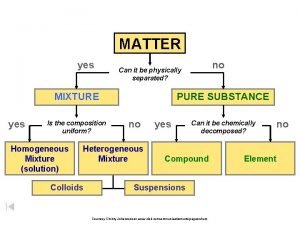
Elements Compounds and Mixtures
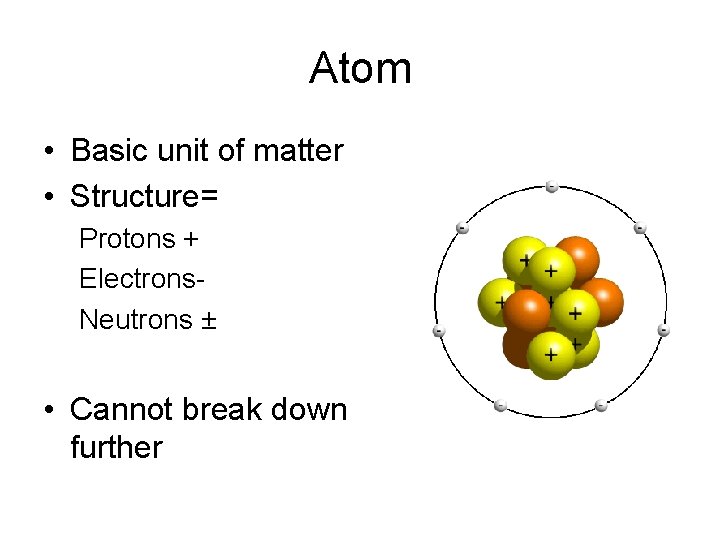
Atom • Basic unit of matter • Structure= Protons + Electrons. Neutrons ± • Cannot break down further

Atoms • Atoms are made up of electrons neutrons and protons.

Elements The Most Basic Form of Matter • Elements are made up of identical atoms. Elements are pure substances that cannot broken down in to simpler substances. Gold

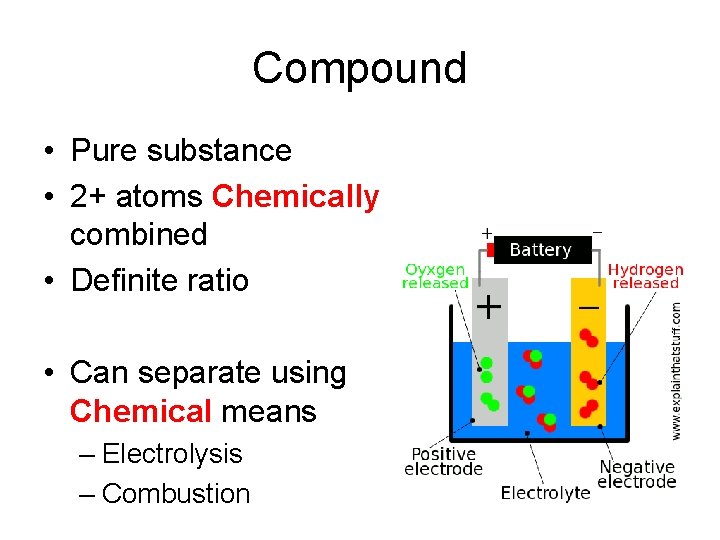
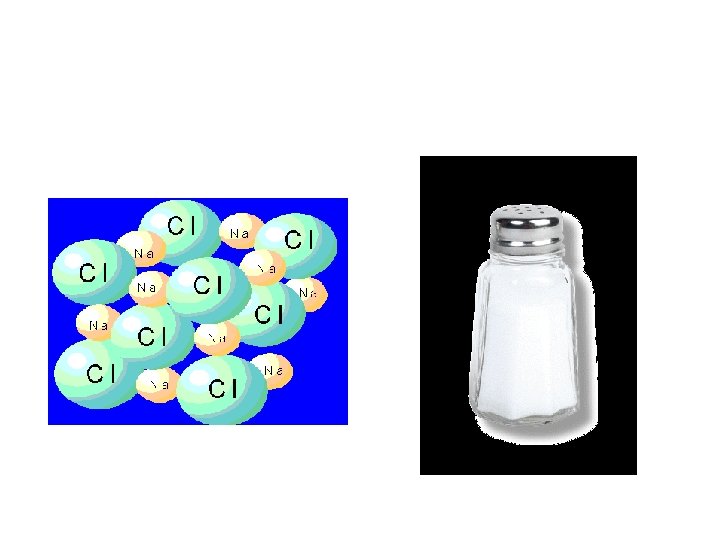
Compound • Pure substance • 2+ atoms Chemically combined • Definite ratio • Can separate using Chemical means – Electrolysis – Combustion
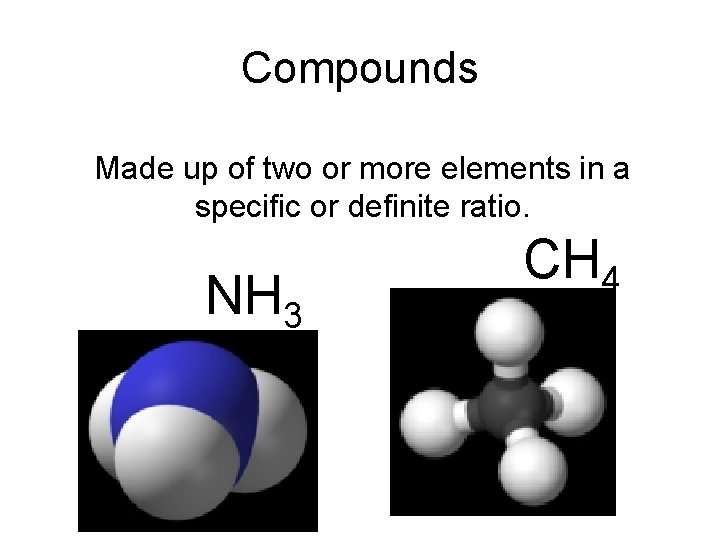
Compounds Made up of two or more elements in a specific or definite ratio. NH 3 CH 4
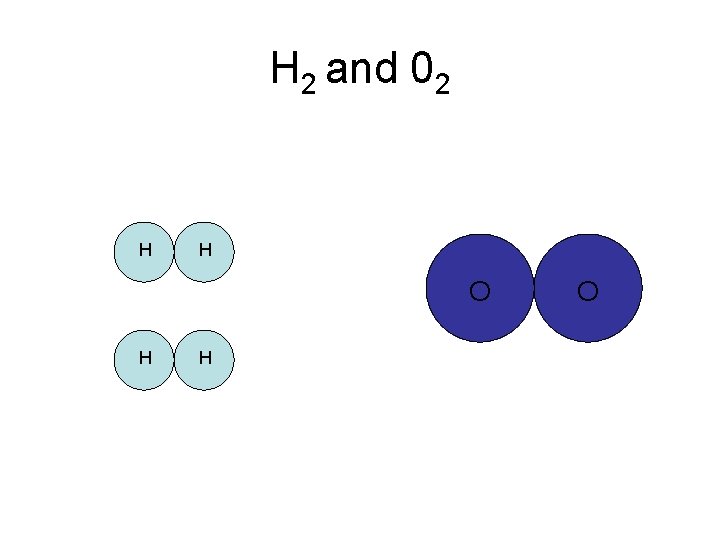
H 2 and 02 H H O
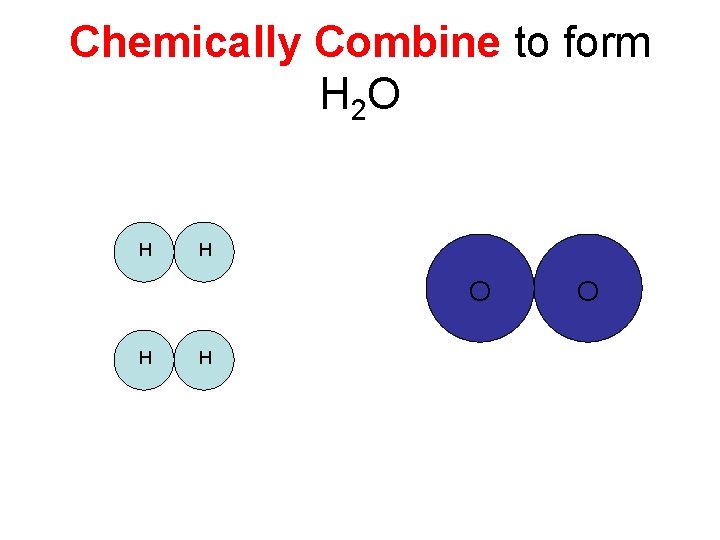
Chemically Combine to form H 2 O H H O
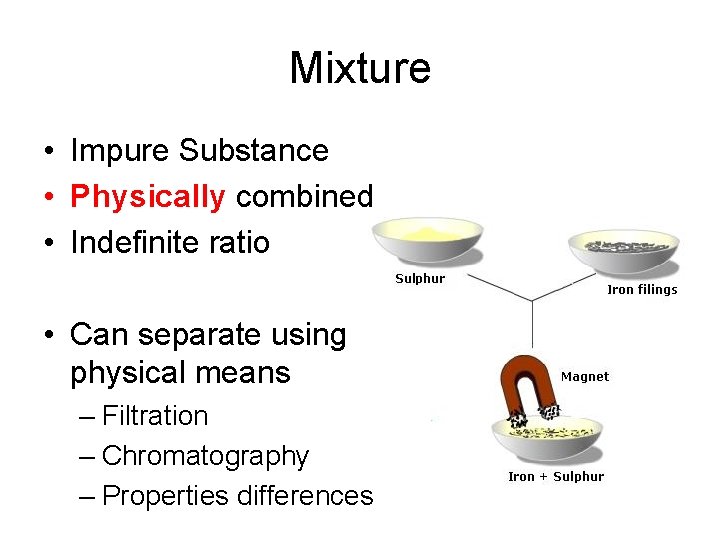
Mixture • Impure Substance • Physically combined • Indefinite ratio • Can separate using physical means – Filtration – Chromatography – Properties differences

Mixture • Mixture is two or more substances that retain their identity.


Tyndall Effect No! John Tyndall Yes!

Tyndall= DON’T see light particles= colloid
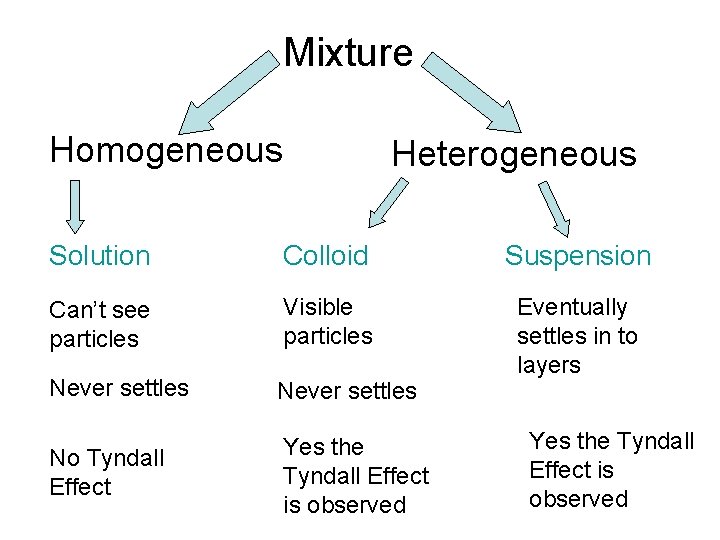
Mixture Homogeneous Heterogeneous Solution Colloid Suspension Can’t see particles Visible particles Never settles Eventually settles in to layers No Tyndall Effect Yes the Tyndall Effect is observed

Quick Quiz: which would be the element? Compound? Mixture?
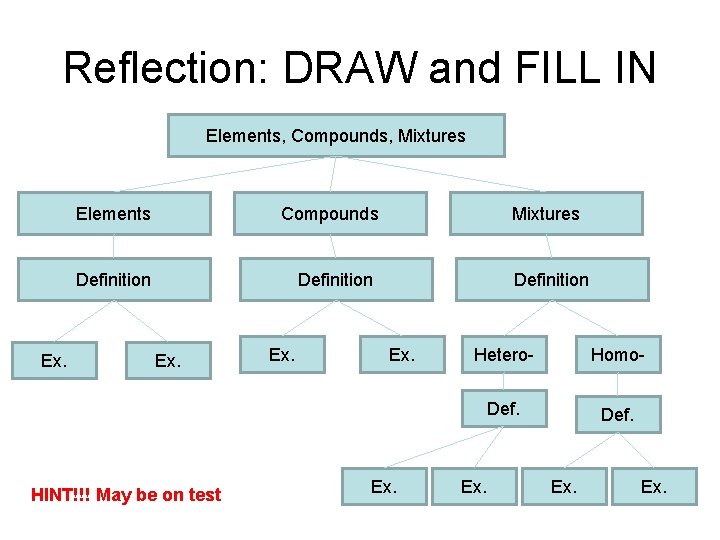
Reflection: DRAW and FILL IN Elements, Compounds, Mixtures Ex. Elements Compounds Definition Ex. HINT!!! May be on test Ex. Mixtures Definition Ex. Hetero- Homo- Def. Ex.
- Slides: 19