Elements Compounds and Mixtures Elements Mixture Compounds By

Elements, Compounds and Mixtures Elements Mixture Compounds By Miss Buicke.

What we must know from the syllabus: OC 3 Understand what an element is and recall that all known elements are listed in the Periodic Table; understand what a compound is and what a mixture is; recall that when elements combine to form compounds they may lose their individual properties OC 12 Compare the properties of the simple compounds to those of the constituent elements OC 13 Compare mixtures and compounds made from the same constituents,
Elements: An element is a substance made up of only one type of atom. There are over 100 different elements. Everything on earth is made from these elements. All the known elements are listed in a table called the periodic table. All elements are given symbols to identify them. We must know the name and symbol for the first twenty elements in the periodic table.

All the symbols have either one or two letters in them. The first letter is a capital letter. The second letter is always a small letter. One letter symbols Element Symbol Hydrogen H Nitrogen N Sulfur S Two letter symbols Element Symbol Zinc Zn Silver Ag Gold Au

Compounds: Compounds consist of two or more elements chemically combined Very often two or more elements from the periodic table join together and from new substances. These new substances are called compounds. Water is a compound made up of two elements: hydrogen and oxygen Water molecule
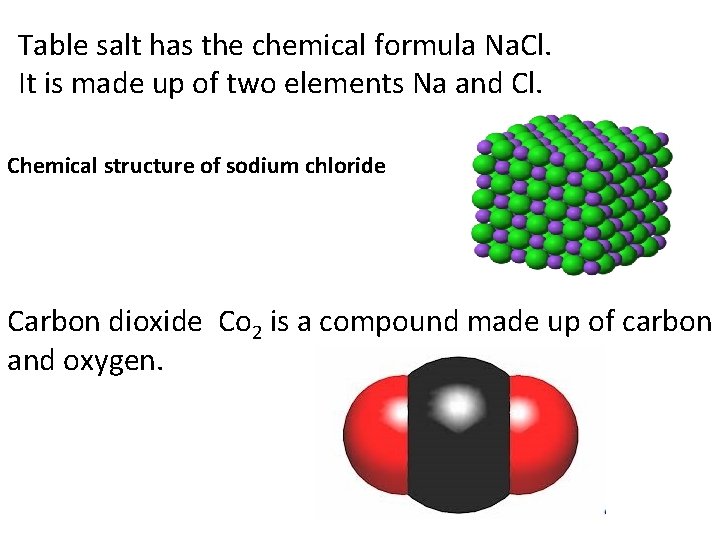
Table salt has the chemical formula Na. Cl. It is made up of two elements Na and Cl. Chemical structure of sodium chloride Carbon dioxide Co 2 is a compound made up of carbon and oxygen.

Mixtures: Mixtures are made up of two or more substances mingled together but not chemically combined. Therefore mixtures can be made up of elements or compounds but in no definite amounts. Air is a mixture of gases in no definite amounts. Sea water is a mixture of water and salt. Dolly mix is a mixture of different colored and shaped sweets.

Differences between compounds and mixtures Compounds Mixtures Made up of a single substance Made up of two or more substances Elements in a compound are Amounts of substances in a always in a fixed ratio mixture can vary Properties of a compound are different to those of the elements it contains Difficult to separate the elements of a compound Properties of a mixture are similar to those of the substances in the mixture Usually easy to separate the substances in a mixture There is usually a heat change when a compound is formed Very little heat change when a mixture is made.

To compare a mixture of iron and sulfur to the compound iron sulfide (Fe. S) Equipment: Mass balance , Spatula, Magnet, Test tube , Tongs, Bunsen burner, Pestel and mortar Chemicals: Iron filings, Sulfur

Method • 1 - using a pestel and mortor, grind up a mixture containing 6 g of iron and 4 g of sulfur. • 2 - wrap a magnet in paper and bring the magnet close to the mixture • 3 - remix the iron and sulfur, using a spatula, and place the mixture in a test tube. • 4 -Heat the test tube until the mixture glows red ( do this in a fume cupboard) • 5 -continue heating gently until the mixture stops glowing • 6 -Allow the test tube to cool and examine its contents. Remove the grey solid formed from the test tube • 7 - Bring a magnet close to the grey solid What do you notice?
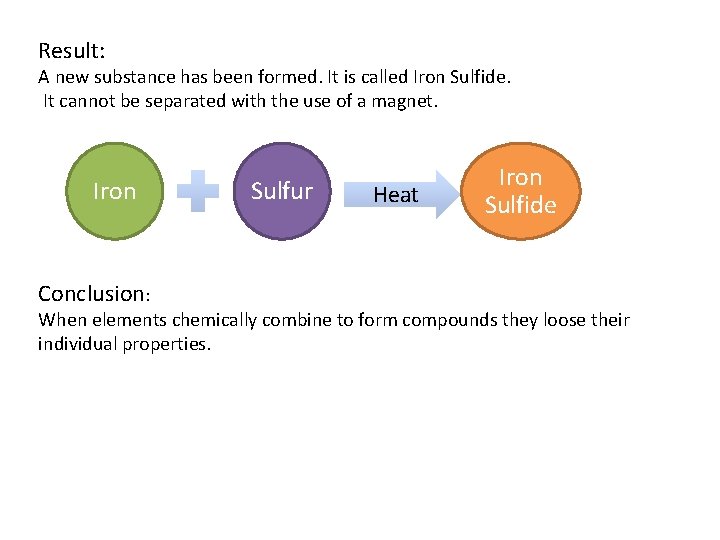
Result: A new substance has been formed. It is called Iron Sulfide. It cannot be separated with the use of a magnet. Iron Conclusion: Sulfur Heat Iron Sulfide When elements chemically combine to form compounds they loose their individual properties.

Properties of compounds: Compounds are made of different elements chemically combines. The properties of compounds are very often completely different from those of the elements from which they are made. compounds elements Sodium chloride (table salt) Sodium: very reactive metal, stored under oil Chlorine: poisonous green gas Carbon dioxide (used in Carbon: black solid fire extinguishers) Oxygen: needed for substances to burn Water (H 2 O) Hydrogen: Explosive gas Oxygen: needed to burn substances




- Slides: 16