Pure Substances and Mixtures Pure Substances A pure

Pure Substances and Mixtures

Pure Substances: A ‘pure’ substance is any substance that is made up of just one element or one type of compound. Examples include: - Gold - Pure water (but not mineral water!) - Iron But what do all these new terms mean?

Elements, Compounds and Mixtures Elements are made up of one type of atom. Compounds are made up of two or three different atoms that are chemically bonded to each other. Mixtures are made up of two or more atoms or compounds that are not chemically bonded to each other.

Copy and complete the sentences below: Word bank: 1: 2, only, elements, 100, compounds, 1: 1 atoms, gases, combine, mixtures 1) The smallest building blocks of matter are called a____. 2) All elements are made of o____ type of atom. 3) There are just over 1____ elements. 4) Most _____ are metals. 5) Some elements are g_____, for example. 6) When two or more different elements c_____ a compound is formed. 8) The proportions of atoms in a particular compound are always the same. For example ___: ___ or ___: ___ 9) Atoms of the same element or of different elements can combine to form c_______. 10) M_______ can be of different elements (example) or of elements and compounds (example) or even of different compounds (example)
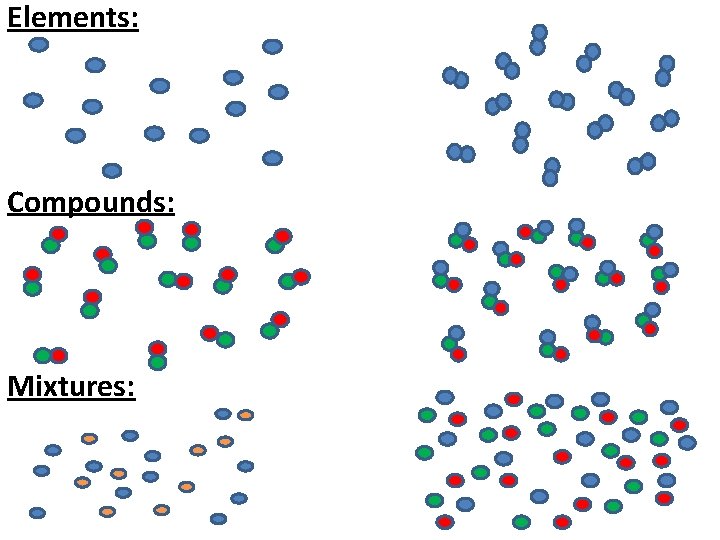
Elements: Compounds: Mixtures:
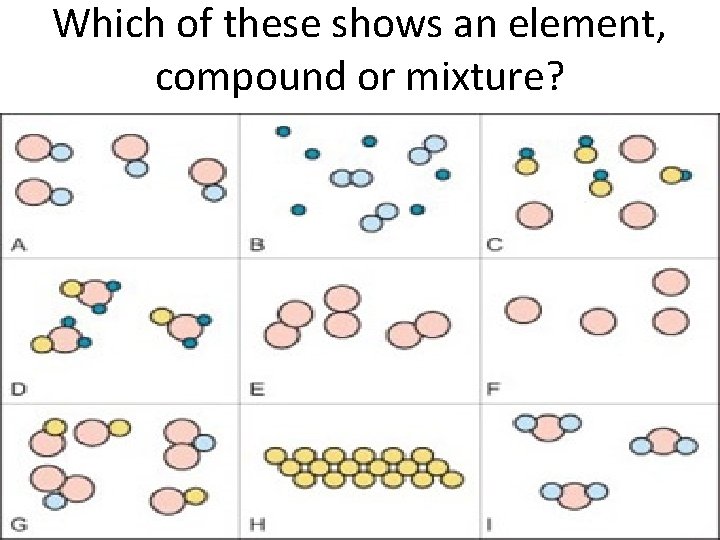
Which of these shows an element, compound or mixture?
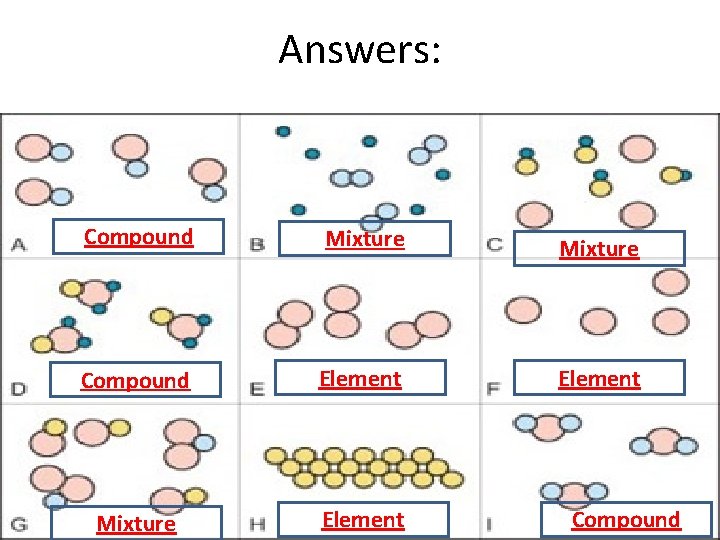
Answers: Compound Mixture Compound Element Mixture Element Compound

Have a go at the worksheet titled ‘Elements compounds and mixtures worksheet’ Answers: 1) A, D, E 2) B, C, F 3) A 4) D, E 5) B, F 6) B, One large hydrogen atom chemically bonded to two smaller oxygen atoms 7) iii – Carbon monoxide, One carbon atom chemically bonded to one oxygen atom
- Slides: 8