Pure Substances and Mixtures Unit 2 Solution Solvent

Pure Substances and Mixtures Unit 2
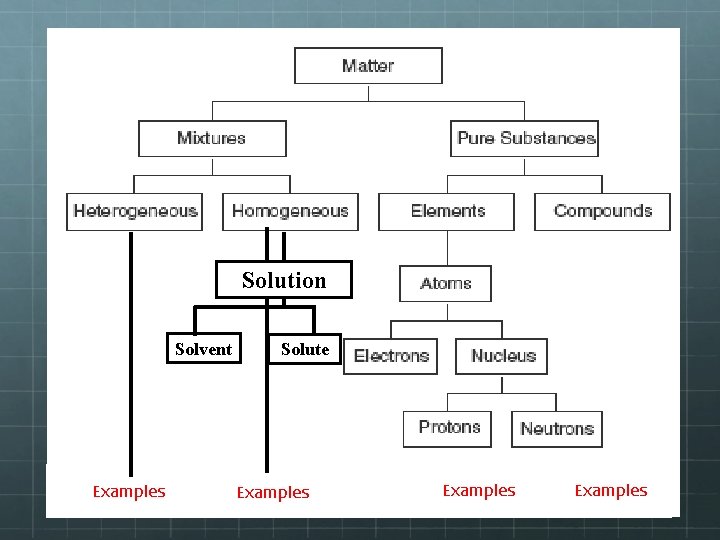
Solution Solvent Examples Solute Examples
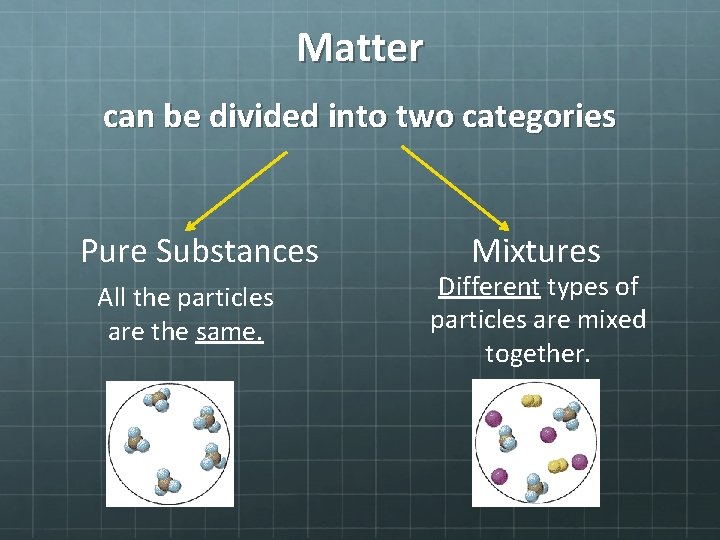
Matter can be divided into two categories Pure Substances All the particles are the same. Mixtures Different types of particles are mixed together.
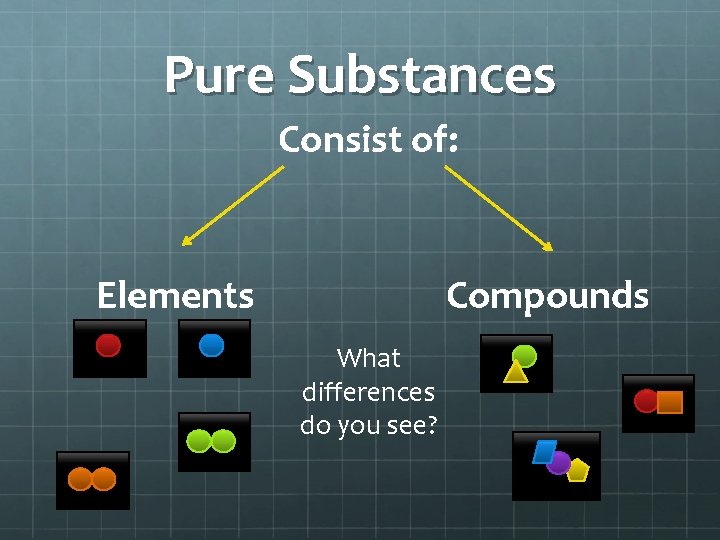
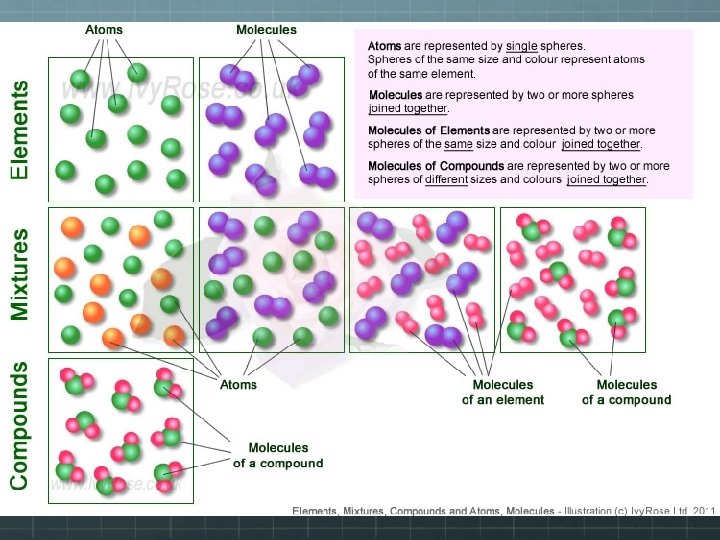
Pure Substances Consist of: Elements Compounds What differences do you see?

Pure Substances Element: Basic unit of substances which are made up of only ONE type of atom Cannot be separated by physical or chemical means Examples: Carbon (C) Oxygen (O) Gold (Ag) Helium (He) The entire periodic table of ELEMENTS
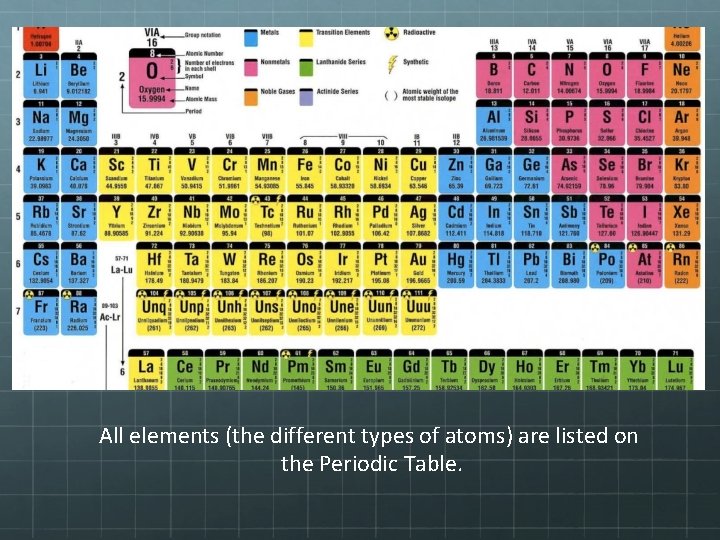
All elements (the different types of atoms) are listed on the Periodic Table.
![Pure Substances Compound: TWO or more DIFFERENT elements chemically combined [BONDED] CANNOT be separated Pure Substances Compound: TWO or more DIFFERENT elements chemically combined [BONDED] CANNOT be separated](http://slidetodoc.com/presentation_image/311fce656058fa703ab30a6ea923254a/image-7.jpg)
Pure Substances Compound: TWO or more DIFFERENT elements chemically combined [BONDED] CANNOT be separated by physical means (only a chemical reaction) Examples: Ammonia, salt, sugar, and formulas such as: Na. Cl CO 2 H 2 O Na. HCO 3

How to read a formula: H 2 0 This is a subscript. It tells us how many atoms of that element exist in one unit of that compound. Hydrogen is made of 2 H atoms and 1 O atom. No subscript is used when only one atom of an element is present.

Mixtures A mixture is a combination of two or more substances where there is NO chemical combination or reaction. They CAN be separated by physical means!

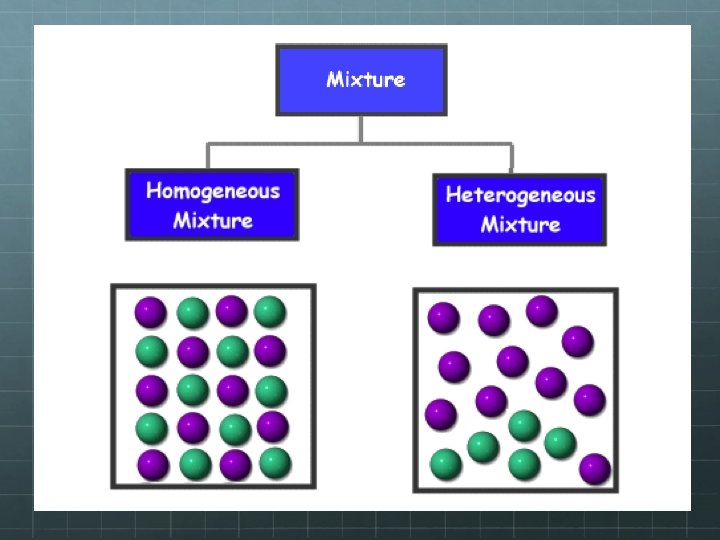
Mixtures are either Homogeneous ? Heterogeneous ?

Mixtures can be divided into two categories Homogenous The different parts are not visible. Heterogeneous The different parts are visible and can be separated mechanically.

Homogeneous Mixture The prefix: "homo"- indicates the same Have a uniform appearance and composition throughout

Homogeneous Mixture Examples: Vinegar Perfume/Cologne Honey Jell-O (Colloids) Mouthwash Vegetable Oil Coffee Water Blood

Mixtures Homogeneous Mixture Solution

Solution A Solution is a mixture of two or more substances. Solute: The substance that is being dissolved – the smallest amount of the two Solvent: The substance that is doing the dissolving – the larger amount of the two. Water is our universal solvent. Question: Kool-Aid and water – Which is the solvent? Which is the solute?

Heterogeneous Mixture The prefix: "hetero"- means different A heterogeneous mixture consists of visibly different substances or phases Two or more parts can be seen

Heterogeneous Mixture Examples: Lucky Charms Captain Crunch Granite/Marble Chocolate Chip Cookies Sand in water Muddy Water (Suspensions)
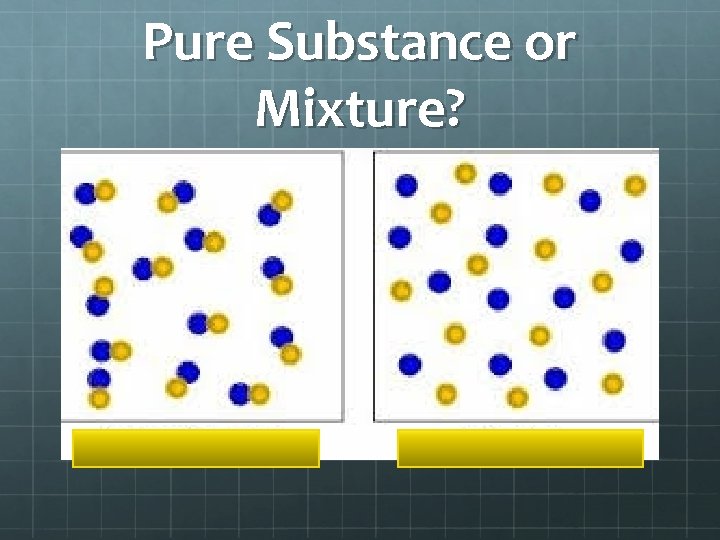
Pure Substance or Mixture?

So to review: Compound Mixture Element Fill in the squares with drawings of element/compound/mixture:

Which type of Pure Substance is each item? 3 T 1 2 EN EM PO D M 4 U D N M PO D N U PO M O C 5 O C E 6 O C M E L N U L E T N E U D N PO C O M T 7 N E EL M E
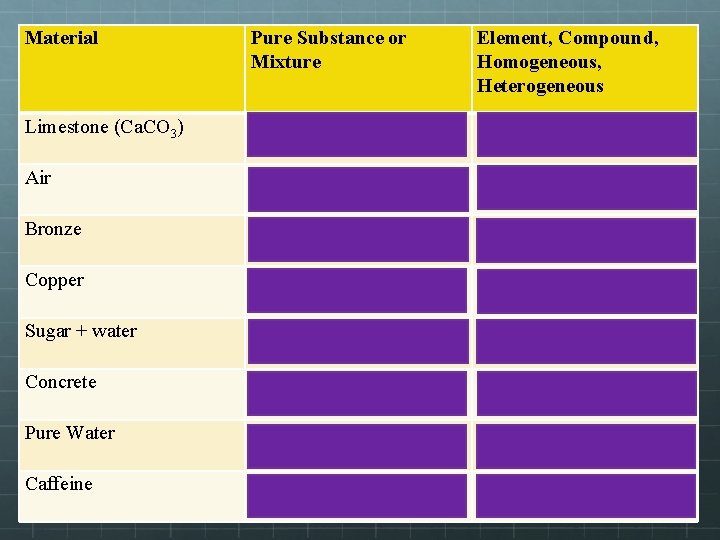
Material Pure Substance or Mixture Element, Compound, Homogeneous, Heterogeneous Limestone (Ca. CO 3) PURE SUBSTANCE COMPOUND Air MIXTURE HOMOGENEOUS Bronze MIXTURE HOMOGENEOUS Copper PURE SUBSTANCE ELEMENT Sugar + water MIXTURE HOMOGENEOUS Concrete MIXTURE HETEROGENEOUS Pure Water PURE SUBSTANCE COMPOUND Caffeine PURE SUBSTANCE COMPOUND
Create a synthesized tree map of your notes: Examples Word Bank: Element, Compound, Mixture, Matter, Pure Substance, Snow Globe, Homogeneous, Heterogeneous, H 2 O, Kool-Aid, O 2, Lemonade, H 2, Jell-O, CO 2
- Slides: 24