Composition of Matter Matter Everything in the universe











- Slides: 17

Composition of Matter • Matter - Everything in the universe is composed of matter – Matter is anything that occupies space and has mass • Mass – quantity of matter an object has • Volume – amount of space an object takes up.

Elements • Pure substances that cannot be broken down chemically into simpler kinds of matter • More than 100 elements (92 naturally occurring)

CHNOPS • 6 most common elements in living things are: – Carbon (C) – Hydrogen (H) – Nitrogen (N) – Oxygen (O) – Phosphorus (P) – Sulfur (S) • 90% or more of the mass of an organism is composed of C, H, N and O

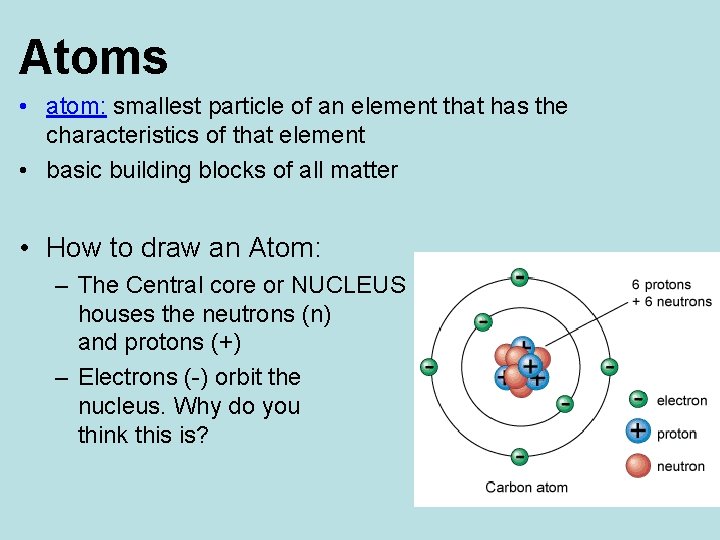
Atoms • atom: smallest particle of an element that has the characteristics of that element • basic building blocks of all matter • How to draw an Atom: – The Central core or NUCLEUS houses the neutrons (n) and protons (+) – Electrons (-) orbit the nucleus. Why do you think this is?

The Protons • All atoms of a given element have the same number of protons • Number of protons called the atomic number • Number of protons balanced by an equal number of negatively charged electrons

Atoms • around the nucleus are the energy levels where negative electrons spin around the nucleus – first energy level holds up to 2 electrons – second energy level holds up to 8 electrons – third energy level holds up to 8 electrons

How can you find the following? • What is the relationship between an atom and an element? Many atoms with the same # of protons make up elements • # of protons in an element? Atomic number • # of electrons in an element? # of protons • # of neutrons in an element? Atomic mass - # of protons

• A compound is a pure substance made up of atoms of two or more elements bonded together – The proportion of atoms are always fixed • Chemical formula shows the kind and proportion of atoms of each element that occurs in a particular compound

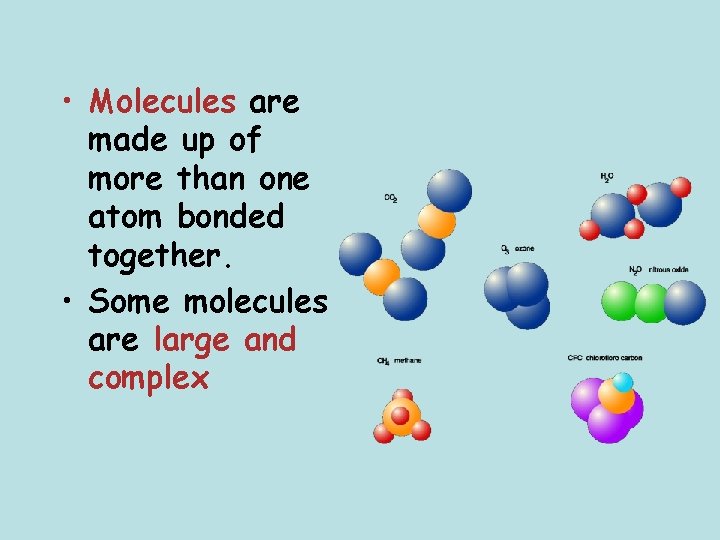
• Molecules are made up of more than one atom bonded together. • Some molecules are large and complex

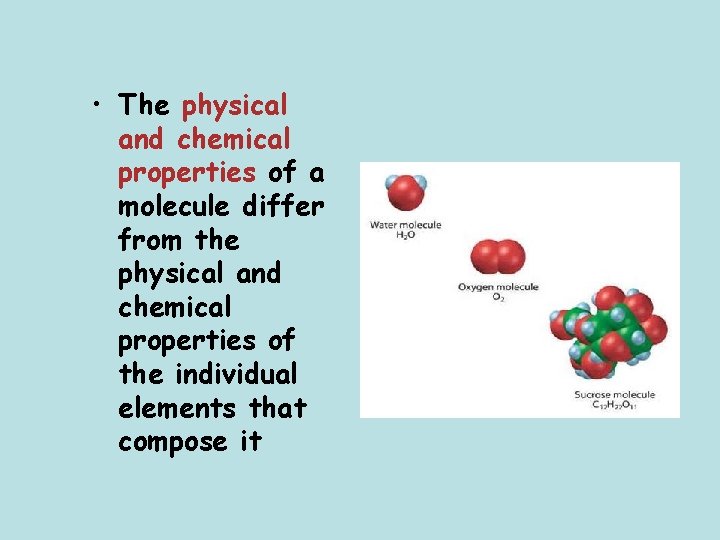
• The physical and chemical properties of a molecule differ from the physical and chemical properties of the individual elements that compose it

How do you draw an Atom to show its electrons will bond? Bohr Diagram: shows ALL electrons with a + (#) and n (#) in the nucleus Ex: Lewis Dot Structure: shows only the VALENCE electrons around the element symbol Ex:

States of Matter • Atoms are in constant motion • The rate at which atoms or molecules in a substance move determines its state

Why do atoms bond? • For the same reasons WE do! – An Atom is typically unstable by itself – It will bond to other atoms so that it can fill up its outside shell of electrons. – Review: When an element bonds to become more stable it is then called a: Molecule

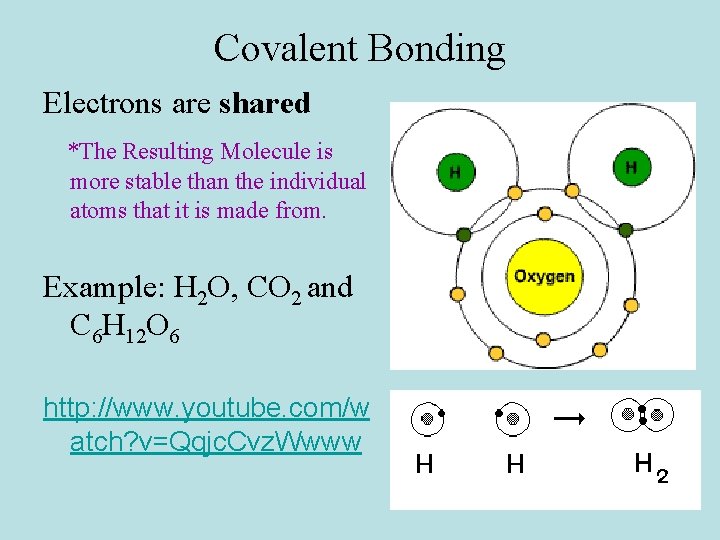
Covalent Bonding Electrons are shared *The Resulting Molecule is more stable than the individual atoms that it is made from. Example: H 2 O, CO 2 and C 6 H 12 O 6 http: //www. youtube. com/w atch? v=Qqjc. Cvz. Wwww

Polarity • Molecules with partial charges on opposite ends ex: H 2 O • Polar Covalent Bonding: electrons are shared unequally, so the electrical charge is not balanced (ex: sugar) • Non- Polar Covalent Bonding: electrons are shared equally by united atoms with a balanced electrical charge (ex: oil, grease, wax)

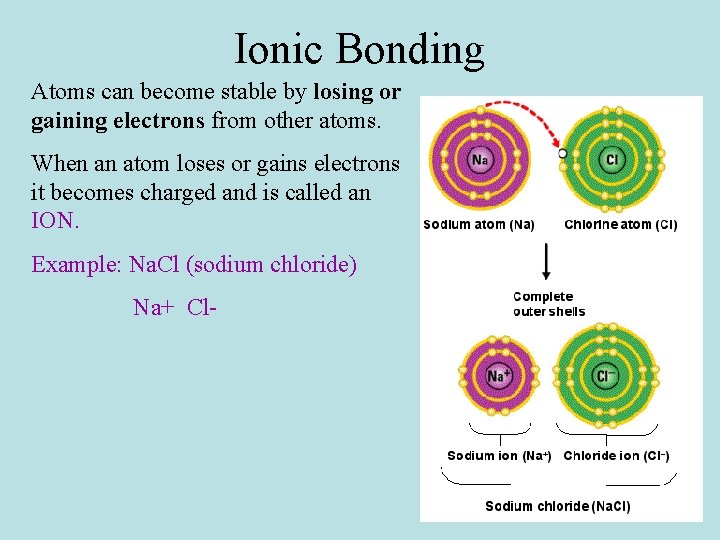
Ionic Bonding Atoms can become stable by losing or gaining electrons from other atoms. When an atom loses or gains electrons it becomes charged and is called an ION. Example: Na. Cl (sodium chloride) Na+ Cl-

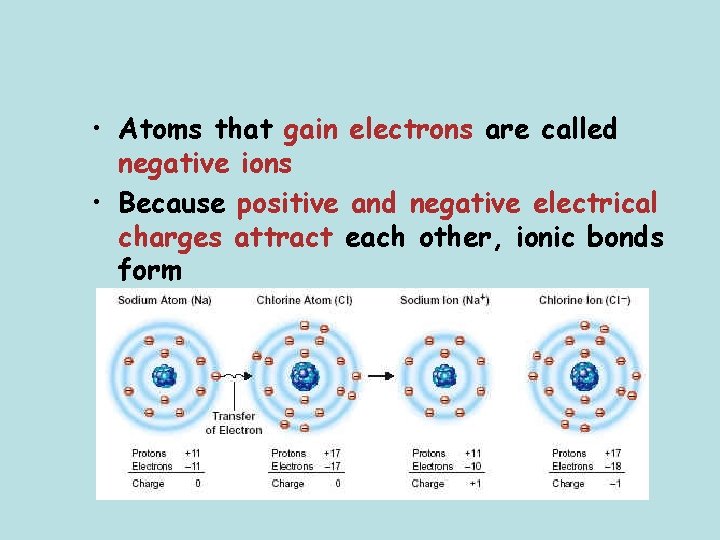
• Atoms that gain electrons are called negative ions • Because positive and negative electrical charges attract each other, ionic bonds form