SECTION 1 COMPOSITION OF MATTER Warmup Think of




















- Slides: 23

SECTION 1: COMPOSITION OF MATTER

Warm-up: • Think of that air you are breathing in right now. • What are some components of air? • Why might air be referred to as a mixture?

Learning Goals • Define substances and mixtures. • Identify elements and compounds. • Compare and contrast solutions, colloids, and suspensions.

Matter • Matter: anything that has mass and takes up space • All matter can be divided into substances and mixtures.

Substances • Substances: type of matter with a fixed composition • can be either elements or compounds.

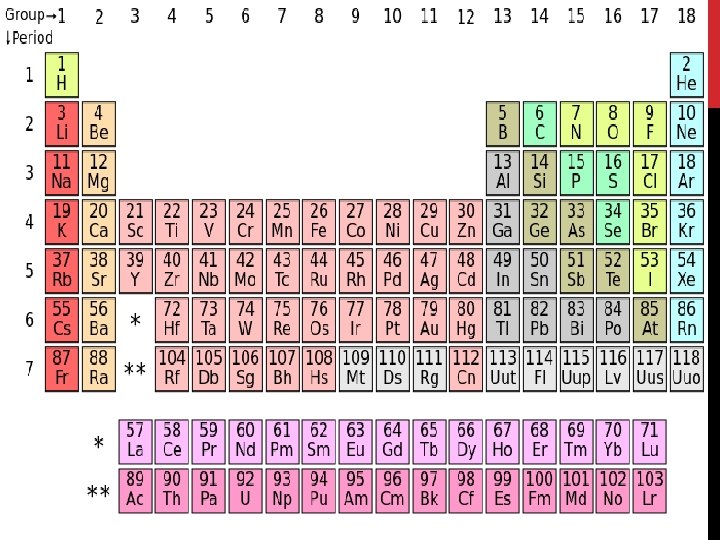
Elements • Elements: substance built from all atoms of the same identity; cannot be broken down into simpler parts • Examples: Hydrogen (H), oxygen (O), carbon (C)

Elements • All elements are listed on the periodic table. • There about 90 naturally occurring elements. • Approximately 20 have been made in laboratories.


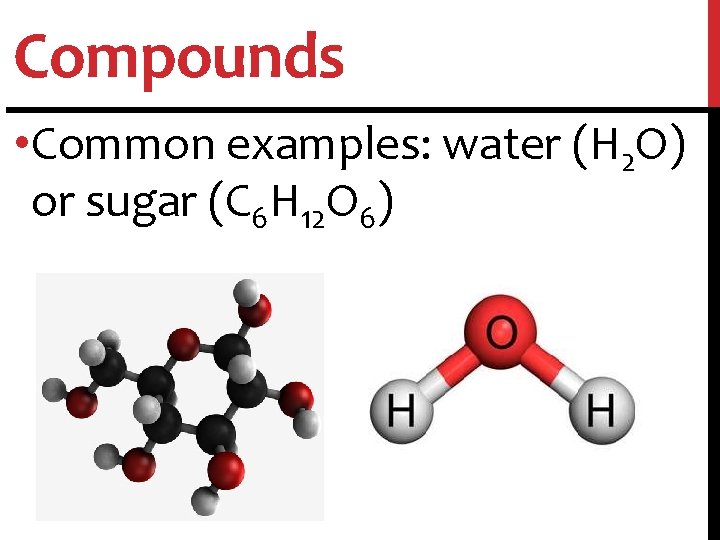
Compounds • Compounds: substance in which two or more atoms are combined in a fixed proportion

Compounds • Common examples: water (H 2 O) or sugar (C 6 H 12 O 6)

Mixtures • Mixture: material made of two or more substances that can be separated by physical means • Mixtures can be heterogeneous or homogeneous

Heterogeneous Mixtures • Heterogeneous mixture: a mixture in which the different materials can be distinguished easily

Heterogeneous Mixtures • Examples: salad, granite, dry soup mix

Homogeneous Mixtures • Homogeneous mixture: contains two or more gaseous, liquid, or solid substances blended evenly throughout • Examples: vinegar (acetic acid and water), iced tea, fog, smoke

Homogenous Mixtures • Homogeneous mixtures can be described as solutions or colloids

Solutions • Solution: homogeneous mixture with particles so small that they cannot be seen with a microscope and will never settle to the bottom of the container • Examples: lemonade, salt water

Solutions • Made up of two parts: • solute (what is dissolved) • solvent (what does the dissolving)

Colloids • Colloids: type of mixture with particles that are larger than those in solutions but are not heavy enough to settle out • Examples: paint, fog, smoke

Colloids • Colloids are detected using the Tyndall effect where you pass light through the substance. The particles in the colloid will scatter the light.

Suspensions • Some mixtures are neither solutions nor colloids • Suspension: heterogeneous mixture containing a liquid in which visible particles settle. • Examples: pond water, Italian dressing

Suspensions


Check-in: • Why might it be easy to confuse a compound a homogeneous mixture?