Chapter 15 Amines Structure Classification l Amines are







- Slides: 22

Chapter 15 Amines

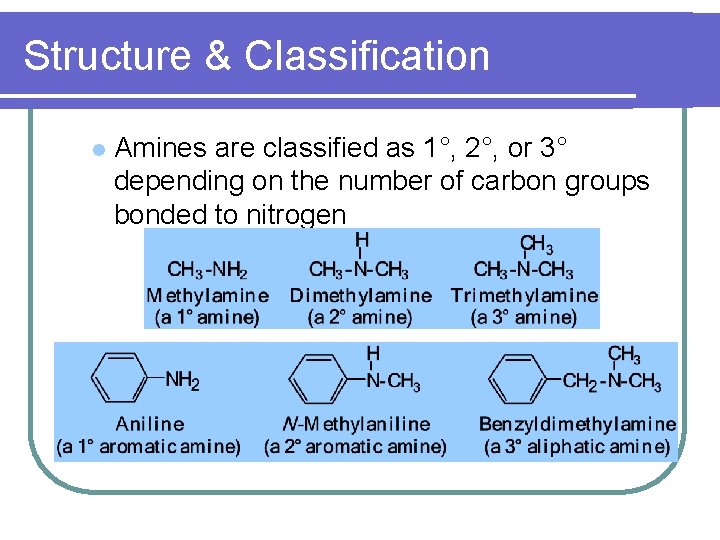
Structure & Classification l Amines are classified as 1°, 2°, or 3° depending on the number of carbon groups bonded to nitrogen

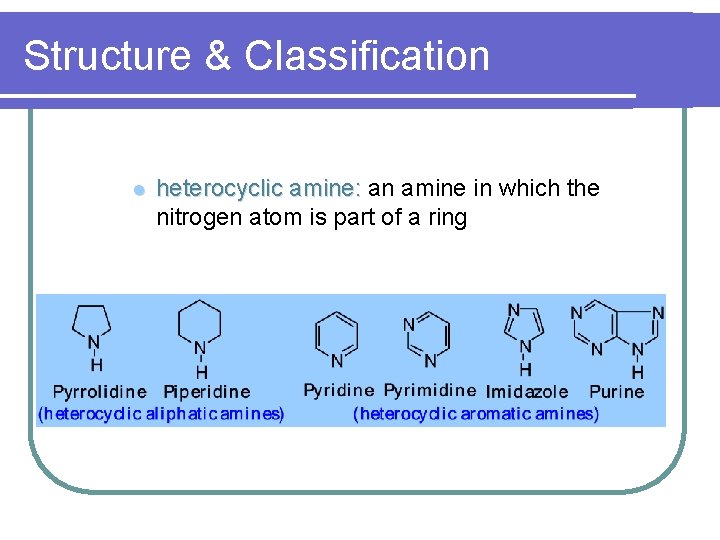
Structure & Classification l heterocyclic amine: an amine in which the nitrogen atom is part of a ring

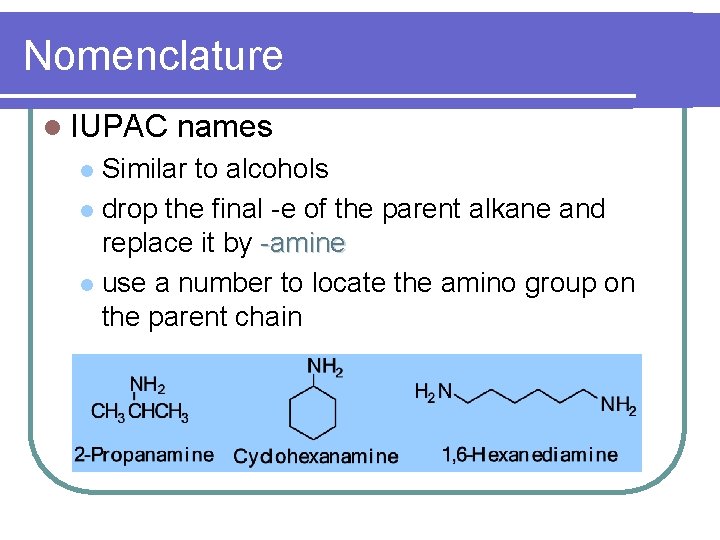
Nomenclature l IUPAC names Similar to alcohols l drop the final -e of the parent alkane and replace it by -amine l use a number to locate the amino group on the parent chain l

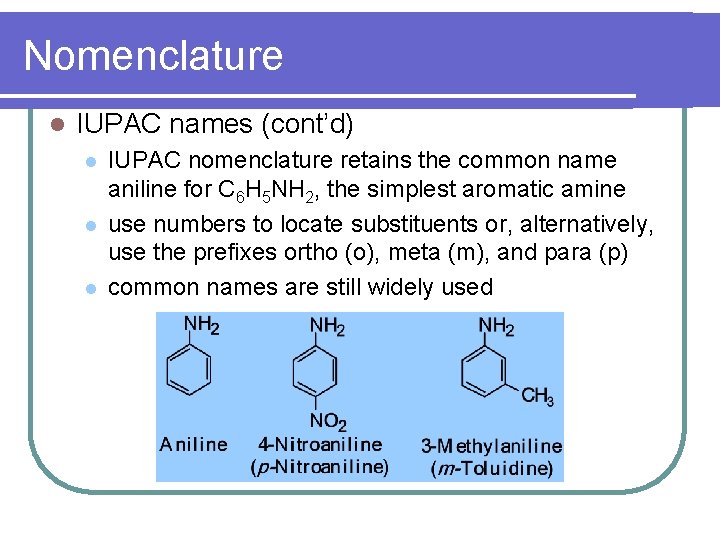
Nomenclature l IUPAC names (cont’d) l l l IUPAC nomenclature retains the common name aniline for C 6 H 5 NH 2, the simplest aromatic amine use numbers to locate substituents or, alternatively, use the prefixes ortho (o), meta (m), and para (p) common names are still widely used

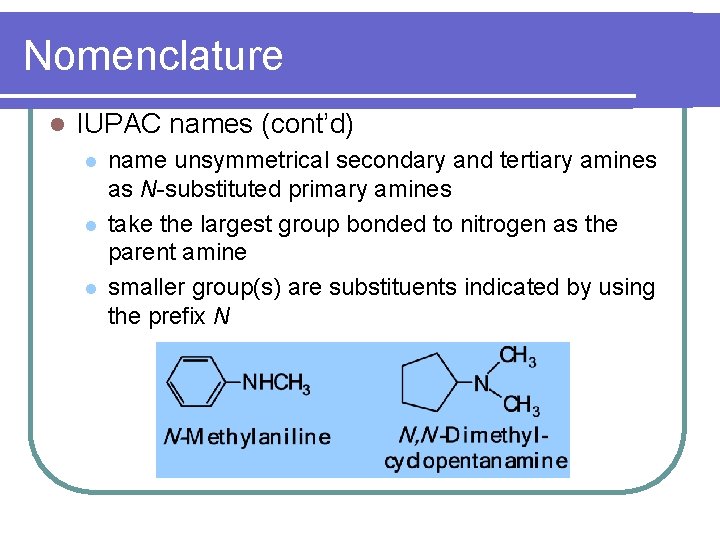
Nomenclature l IUPAC names (cont’d) l l l name unsymmetrical secondary and tertiary amines as N-substituted primary amines take the largest group bonded to nitrogen as the parent amine smaller group(s) are substituents indicated by using the prefix N

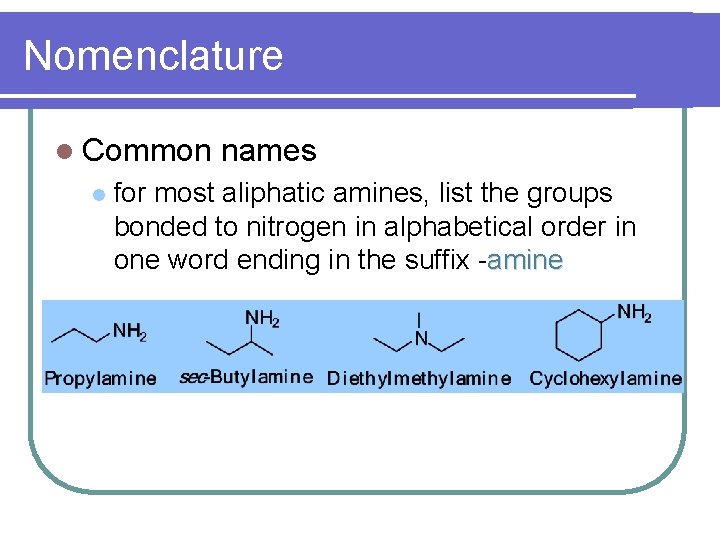
Nomenclature l Common l names for most aliphatic amines, list the groups bonded to nitrogen in alphabetical order in one word ending in the suffix -amine

Nomenclature l Amine salts when four things are bonded to a nitrogen atom, the nitrogen bears a positive charge l name the compound as a salt l replace the ending -amine (or aniline or pyridine or the like) by -ammonium (or anilinium or pyridinium or the like) and add the name of the anion l

Physical Properties l Like ammonia, low-molecular-weight amines have very sharp, penetrating odors trimethylamine, for example, is the pungent principle in the smell of rotting fish l two other particularly pungent amines are 1, 4 -butanediamine (putrescine) and 1, 5 pentanediamine (cadaverine) l

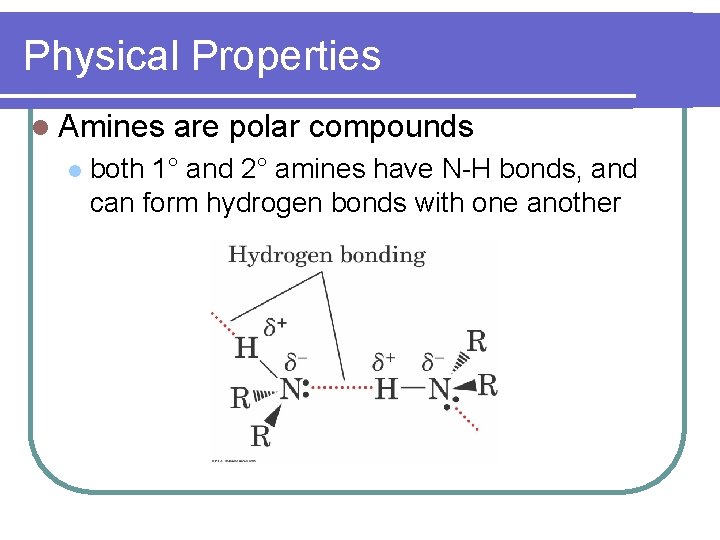
Physical Properties l Amines l are polar compounds both 1° and 2° amines have N-H bonds, and can form hydrogen bonds with one another

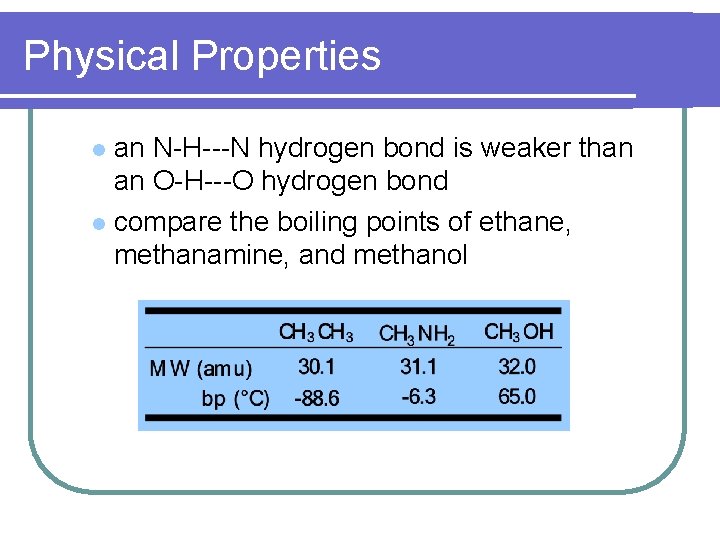
Physical Properties an N-H---N hydrogen bond is weaker than an O-H---O hydrogen bond l compare the boiling points of ethane, methanamine, and methanol l

Physical Properties all classes of amines form hydrogen bonds with water and are more soluble in water than are hydrocarbons of comparable molecular weight l most low-molecular-weight amines are completely soluble in water l higher-molecular-weight amines are only moderately soluble in water or are insoluble l

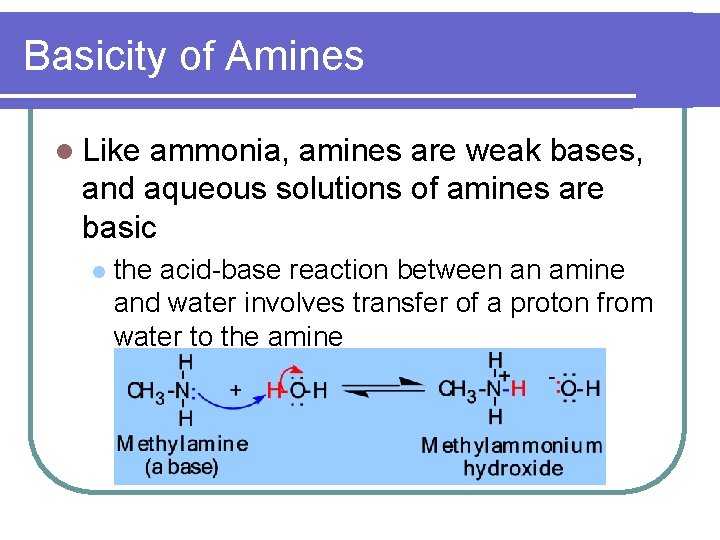
Basicity of Amines l Like ammonia, amines are weak bases, and aqueous solutions of amines are basic l the acid-base reaction between an amine and water involves transfer of a proton from water to the amine

Basicity of Amines l The base dissociation constant, Kb, for the reaction of an amine with water has the following form, illustrated for the reaction of methylamine with water to give methylammonium hydroxide l p. Kb is defined as the negative logarithm of Kb

Basicity of Amines aliphatic amines SS bases than NH 3 l aromatic amines are weaker bases l

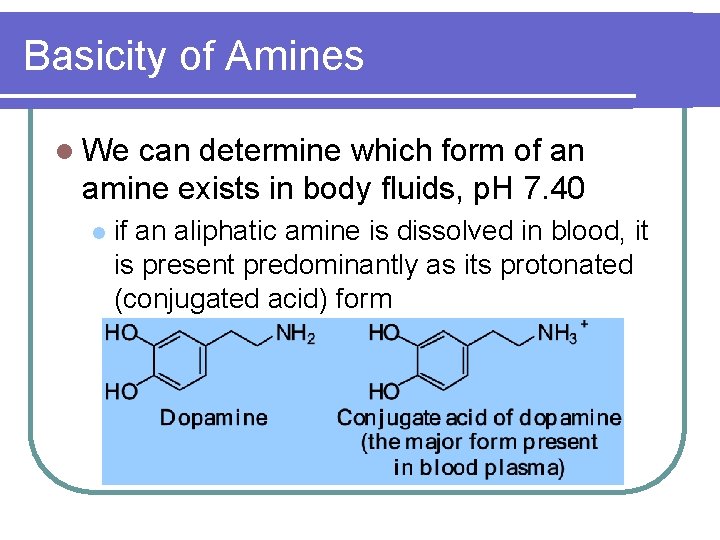
Basicity of Amines l We can determine which form of an amine exists in body fluids, p. H 7. 40 l if an aliphatic amine is dissolved in blood, it is present predominantly as its protonated (conjugated acid) form

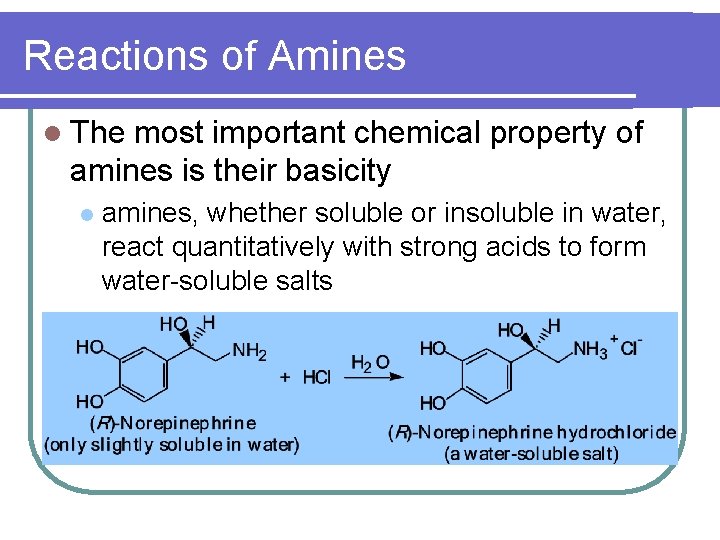
Reactions of Amines l The most important chemical property of amines is their basicity l amines, whether soluble or insoluble in water, react quantitatively with strong acids to form water-soluble salts

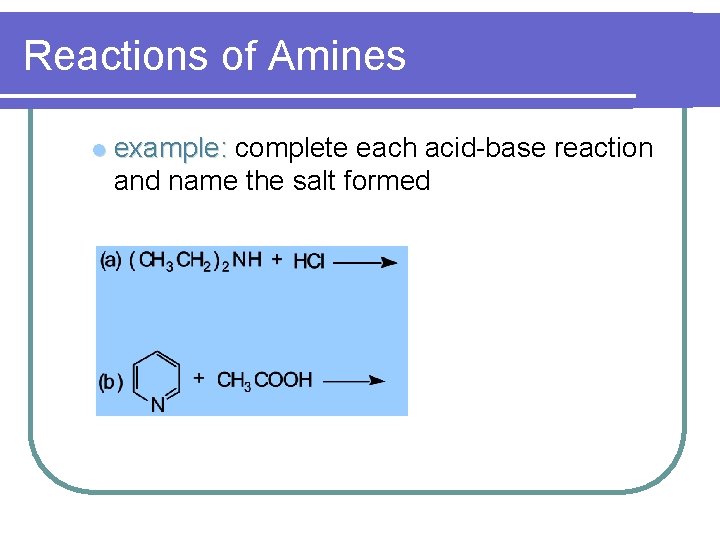
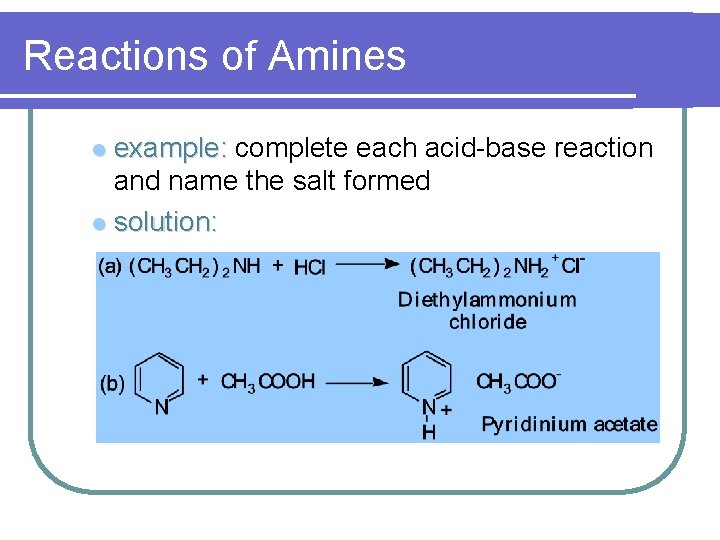
Reactions of Amines l example: complete each acid-base reaction and name the salt formed

Reactions of Amines example: complete each acid-base reaction and name the salt formed l solution: l

Synthesis of Amines l 1) Alkyl halides react with ammonia to make primary amines l l l This can lead to multiple amination products Example on board 2) Alkyl halides may be reacted with cyanide and then hydrogenated to for the primary amine exclusively. l Example on board

Reactions of Amines l 3) Aldehydes and ketones can condense with primary amines and then be hydrogenated to for secondary amines l Example on the board

Amines End Chapter 16