CHEMISTRY LESSON BASIC CHARACTER OF AMINES 1 objectives

CHEMISTRY LESSON: BASIC CHARACTER OF AMINES 1

objectives: Fundamental concepts on basic character of amines. ü Applications of these concepts in comparing the relative basic strengths of NH 3, various alkyl amines, C 6 H 5 NH 2 and other substituted aromatic amines. ü 2

FUNDAMENTAL CONCEPTS ON BASIC CHARACTER OF AMINES � Amines are derivatives of ammonia: NH 3 -H R – NH 2 +R � Various types of amines are primary (10), secondary (20) and tertiary (30): RNH 2 R 2 NH R 3 N (10) (20) (30) � The basic nature of an amine is due to the presence of a lone pair on N, and its ability to donate it; and depends on the stability of the conjugate acid. 3

� Steric hindrance plays an important role in deciding basic strength. � Electron releasing groups increase the basic strength , whereas electron withdrawing group decrease the basic strength of amines. � A higher value of p. Kb implies lower basic strength. 4

APPLICATION : COMPARISON OF NH 3 AND RNH 2 As the alkyl group has +I – effect, it increases the electron density on N. � Substituted ammonium ion is more stable due to charge dispersal by the alkyl group. � So, RNH 2 is more basic than NH 3. 5

APPLICATION : COMPARISON OF RNH 2, R 2 NH AND R 3 N IN GASEOUS STATE As the alkyl groups have +I – effect, it increases the electron density on N. � As these are in the gaseous phase, so only the +I – effect is considered. � So, the order of basic nature is: 30 > 20 > 10 > NH 3. 6

APPLICATION : COMPARISON OF RNH 2, R 2 NH AND R 3 N IN AQUEOUS STATE In aqueous solutions, in addition to +I – effect, the solvation effect and steric hindrance are also at work. � As 30 amines are least solvated, so their conjugate acids are least stable. So, we expect an order as : 10 > 20 > 30. � But, due to the combination of +I – and solvation effects, secondary amines become most basic. � So, the order of basic nature in methyl substituted amines is: 20 > 10 > 30 > NH 3. 7

But, this order applies to methyl substituted amines only. Ø If R– is ethyl (C 2 H 5–) group, then due to steric hindrance, solvation effect decreases in all the three types of amines. Hence, +I – effect of tertiary amines tends to dominate over the solvation effect of primary amines. Hence, the order of basic nature in ethyl substituted amines is: 20 > 30 > 10 > NH 3. Ø Similarly, in propyl substituted amines, though electron density is increased due to +I – effect, still, due to steric hindrance, ability to donate the electron pair is decreased and hence the order is changed again. The order of basic nature in propyl substituted amines is: 10 > NH 3 > 20 > 30. Similarly, the order of basic nature in butyl substituted amines is: NH 3 > 10 > 20 > 30. 8

APPLICATION : COMPARISON OF AROMATIC AMINES WITH ALIPHATIC AMINES p. Kb of aniline is very high, as compared to that of alkyl amines; i. e. , aniline is much less basic than alkyl amines. It is because the lone pair on nitrogen in aniline is in conjugation with the benzene ring. v Also, anilinium ion (conjugate acid) is less stable than aniline as it has lesser number of resonating structures. v 9
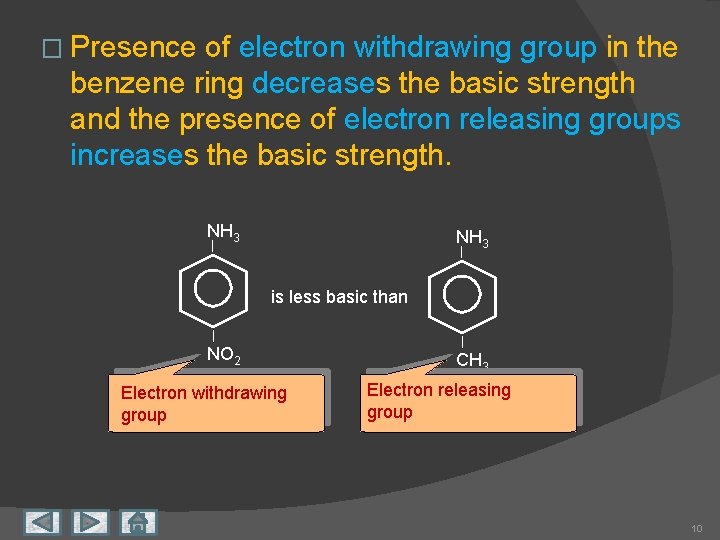
� Presence of electron withdrawing group in the benzene ring decreases the basic strength and the presence of electron releasing groups increases the basic strength. NH 3 is less basic than NO 2 Electron withdrawing group CH 3 Electron releasing group 10

THE ORTHO EFFECT IN AROMATIC AMINES � It has been observed that any group; whether electron withdrawing, or electron releasing, at the ortho position in aniline always decreases its basic strength. This is known as the ORTHO EFFECT. � The reason for the occurence of the ortho effect is steric hindrance. 11

QUESTIONS FOR PRACTICE (CLASSROOM EXERCISE) � Predict the increasing order of basic strength in each of the following cases: NH CH 3 – C – NH 2 (I) CH 3 – CH 2 – NH 2 (II) CH 3 O CH 3 – CH – NH 2 (III) CH 3 – C – NH 2 (IV) Answer: IV < III < I 12
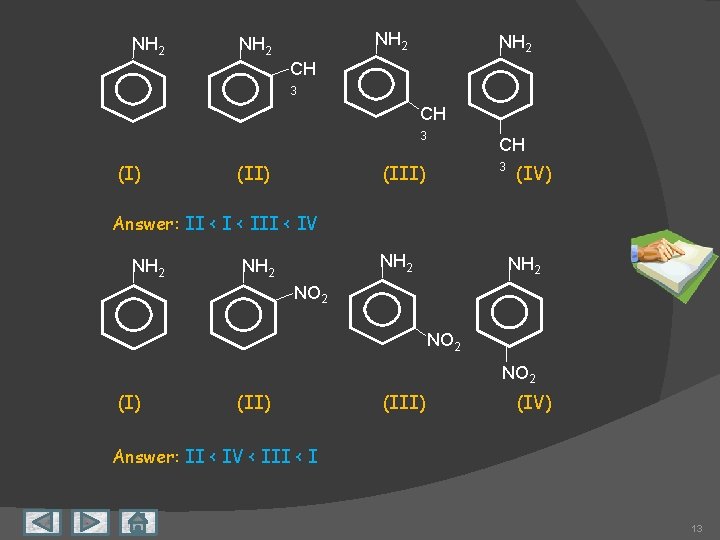
NH 2 CH 3 (I) (II) CH (III) 3 (IV) Answer: II < III < IV NH 2 NO 2 (I) (III) (IV) Answer: II < IV < III < I 13

N A TH U O KY 14
- Slides: 14